Breakthrough Purification Technology for High-Purity Miriplatin: Enhancing API Quality and Commercial Scalability
The pharmaceutical landscape for oncology treatments demands uncompromising standards of purity, particularly for platinum-based antitumor agents like Miriplatin (also known as Miboplatin). Patent CN112094298A introduces a transformative preparation method that addresses the longstanding challenge of removing homologous impurities from crude Miriplatin. Historically, the synthesis of this critical liver cancer therapeutic has been plagued by the persistence of structurally similar byproducts that evade standard purification techniques. This new technical disclosure outlines a sophisticated solvent engineering approach that leverages specific solubility differentials to achieve purity levels exceeding 99.87%. For global procurement and R&D teams, this represents a significant leap forward in process reliability, offering a pathway to produce high-purity pharmaceutical intermediates with reduced regulatory risk and enhanced batch-to-batch consistency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for Miriplatin have struggled with two distinct but interrelated bottlenecks: the introduction of toxic heavy metals and the inability to effectively purge homologous impurities. Earlier methodologies, such as those disclosed in CN101402655A, relied on silver ions to facilitate the formation of intermediate platinum complexes. While chemically effective, this approach inevitably introduces silver residues, necessitating expensive and technically demanding removal steps to meet stringent safety specifications for parenteral drugs. Furthermore, alternative purification strategies involving mixed solvents like n-butanol and ethanol, or dichloromethane and methanol-water systems, have demonstrated limited efficacy. These conventional recrystallization protocols often fail to distinguish between the target molecule and its homologous counterparts, leaving behind impurities that compromise the safety profile of the final Active Pharmaceutical Ingredient (API).
The Novel Approach
The innovative method described in the patent circumvents these historical deficiencies by employing a ternary solvent system comprising an alkyl halide, an ester solvent, and acetone. Instead of attempting to crystallize the product directly from a messy mixture, this process ingeniously induces the selective crystallization of the impurities themselves. By dissolving the crude material in an alkyl halide such as dichloromethane and subsequently introducing a mixed solution of ethyl acetate and acetone, the solubility parameters of the system are shifted. This shift forces the homologous impurities to precipitate out of the solution while the desired Miriplatin remains in the filtrate. Subsequent concentration of the filtrate yields a white solid of exceptional purity. This inversion of the typical purification logic—removing impurities via crystallization rather than the product—provides a robust mechanism for achieving the ultra-low impurity profiles required for modern oncology therapeutics.
Mechanistic Insights into Solvent-Driven Impurity Rejection
The core of this technological advancement lies in the precise manipulation of intermolecular forces and solubility equilibria. Miriplatin, chemically defined as a platinum complex with a tetracosanoic acid ligand, possesses a long hydrophobic chain that dictates its solvation behavior. The homologous impurities, which differ only by the length of their fatty acid chains (ranging from shorter C8/C10 chains to slightly longer variants), exhibit subtly different lattice energies and solvation shells. 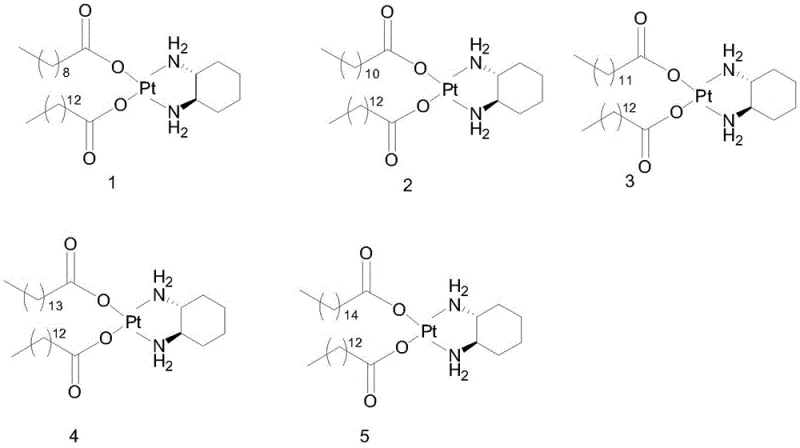
As depicted in the structural analysis, Impurities 1 through 5 represent variants where the fatty acid chain length deviates from the target specification. In a standard solvent system, these molecules are too similar to separate efficiently. However, the specific combination of an ester solvent (like ethyl acetate) and acetone creates a polarity environment where these specific homologous variants become thermodynamically unstable in the solution phase at lower temperatures. The alkyl halide acts as a primary solubilizer for the bulk material, while the anti-solvent effect of the acetone/ester mixture is tuned to exceed the saturation point of the impurities first. This selective precipitation ensures that when the filtration step occurs, the solid waste contains the unwanted homologous species, leaving the mother liquor enriched with the target Miriplatin. This mechanism effectively decouples the purification efficiency from the initial crude quality, providing a powerful buffer against upstream process variations.
How to Synthesize High-Purity Miriplatin Efficiently
The operational execution of this purification protocol is designed for simplicity and reproducibility, making it highly attractive for process chemistry teams aiming to scale up production. The procedure begins with the dissolution of crude Miriplatin in a chlorinated solvent under inert atmosphere to prevent oxidation or degradation. The critical step involves the controlled dropwise addition of the ester-acetone mixture, which must be managed to ensure uniform nucleation of the impurities. Following a period of temperature-controlled stirring to maximize impurity crystal growth, the suspension is filtered. The true value of this method is realized in the final step: the filtrate, now stripped of the difficult-to-remove homologous impurities, is concentrated under reduced pressure to recover the purified product.
- Dissolve crude Miriplatin in an alkyl halide solvent (e.g., dichloromethane) under nitrogen protection and heat until fully solubilized.
- Dropwise add a precise mixed solution of an ester solvent (such as ethyl acetate) and acetone to the reaction vessel while maintaining controlled temperatures.
- Cool the mixture to induce selective crystallization of impurities, filter them off, and concentrate the filtrate under reduced pressure to isolate high-purity Miriplatin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this purification technology translates into tangible strategic advantages beyond mere chemical purity. The elimination of silver-based reagents removes a significant cost driver and supply chain vulnerability, as silver prices can be volatile and the handling of heavy metal waste incurs substantial environmental compliance costs. Furthermore, the simplified workflow reduces the overall cycle time of the manufacturing process. By avoiding complex multi-step extractions or the use of toxic solvents like toluene in the final crystallization stages, the facility can operate with greater throughput and reduced downtime for cleaning and validation.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from the drastic simplification of the purification train. By removing the necessity for silver salts, the direct material costs are lowered, and the downstream burden of heavy metal scavenging is entirely eliminated. Additionally, the high recovery yield reported in the examples suggests that less raw material is wasted during the purification stage. The use of common, recoverable solvents like dichloromethane and ethyl acetate further enhances the cost-efficiency profile, allowing for solvent recycling loops that minimize waste disposal expenses and reduce the overall cost of goods sold (COGS) for the final API.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on specialized reagents or complex purification steps that are prone to failure. This method utilizes commodity chemicals that are readily available in the global market, mitigating the risk of raw material shortages. The robustness of the crystallization process means that even if the quality of the incoming crude Miriplatin fluctuates slightly, the purification step is capable of consistently delivering a product that meets tight specifications. This resilience ensures that production schedules can be maintained without unexpected delays caused by out-of-specification batches, securing a steady flow of high-purity pharmaceutical intermediates to downstream formulation partners.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process offers a cleaner manufacturing footprint. The avoidance of toluene and the reduction of heavy metal usage align with modern green chemistry principles and increasingly stringent global environmental regulations. The simplicity of the operation—essentially a dissolve-filter-concentrate sequence—is inherently easier to scale from pilot plant to commercial tonnage than multi-stage extraction protocols. This scalability ensures that as demand for Miriplatin grows, the manufacturing capacity can be expanded rapidly without requiring fundamental re-engineering of the process, thereby supporting long-term supply security for the global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-purity Miriplatin synthesis route. These insights are derived directly from the experimental data and process descriptions provided in the patent literature, offering clarity on how this technology resolves specific industry pain points.
Q: How does this new method address the issue of heavy metal residues in Miriplatin?
A: Unlike prior art methods (e.g., CN101402655A) that utilize silver salts for nitrate exchange—introducing risks of silver ion contamination—this novel purification protocol relies solely on organic solvent engineering. By eliminating the need for inorganic salt metathesis involving heavy metals, the process inherently prevents heavy metal residues, significantly simplifying the downstream purification burden and ensuring compliance with strict ICH Q3D guidelines for elemental impurities.
Q: What specific impurities are targeted by this crystallization technique?
A: The primary challenge in Miriplatin synthesis is the presence of homologous impurities arising from fatty acid chain length variations. As illustrated in the structural analysis, these impurities (labeled Impurity 1 through 5) possess carboxylate chains varying from C8 to C14, which are structurally analogous to the target tetracosanoic acid ligand. Traditional recrystallization often fails to separate these close analogs. This method specifically targets these homologous species, reducing their collective content to below 0.02%.
Q: Is this purification process suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for scalability. It avoids complex operations such as activated carbon filtration at high temperatures or multi-step extractions that lead to yield loss. The use of common industrial solvents like dichloromethane, ethyl acetate, and acetone ensures that the process can be easily transferred from laboratory bench scale to multi-ton production without requiring exotic equipment or hazardous conditions, thereby supporting robust supply chain continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Miriplatin Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and rigorous quality control. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of advanced purification technologies are fully realized in practice. Our state-of-the-art facilities are equipped with rigorous QC labs capable of detecting trace impurities at ppm levels, guaranteeing that every batch of Miriplatin we supply meets the most stringent purity specifications required for oncology applications. We are committed to delivering not just a chemical product, but a validated, reliable supply solution that supports your drug development and commercialization timelines.
We invite you to leverage our technical capabilities to optimize your supply chain for Miriplatin and related platinum-based intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing processes can enhance the quality and economics of your pharmaceutical portfolio.
