Revolutionizing Miriplatin Manufacturing: Aqueous Synthesis and Commercial Scalability for Global Oncology Markets
The pharmaceutical industry is constantly seeking robust manufacturing pathways for complex oncology agents, and patent CN101402655A presents a transformative approach to the production of Miriplatin, a lipophilic platinum-based anticancer drug. This specific intellectual property details a novel preparation method that fundamentally shifts the synthesis paradigm from hazardous organic solvent systems to a benign aqueous environment. By utilizing Cis-dinitrato((1R,2R)-1,2-cyclohexanediamine)platinum(II) as the starting material and reacting it with myristate salts in purified water, the process achieves exceptional product quality while mitigating significant environmental and operational risks. The strategic importance of this technology lies in its ability to deliver high-purity intermediates suitable for sensitive therapeutic applications, addressing the critical needs of R&D directors who prioritize impurity profiles and process safety. As a leading entity in fine chemical manufacturing, understanding the nuances of this patent allows us to offer superior supply chain solutions for global pharmaceutical partners seeking reliable Miriplatin supplier capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of lipophilic platinum complexes like Miriplatin relied heavily on methods disclosed in earlier patents such as WO94/14470, which necessitated the use of chloroform as a primary solvent and silver myristate as a reagent. These conventional approaches suffer from severe inherent drawbacks, primarily stemming from the extremely low solubility of the reactants in chloroform, which forces reaction times to extend beyond twenty-four hours to achieve completion. The reliance on chloroform introduces substantial regulatory and safety burdens, as it is a toxic halogenated solvent that requires rigorous containment, recovery, and disposal protocols to protect operator health and comply with environmental standards. Furthermore, the use of silver salts not only escalates raw material costs significantly but also generates silver chloride precipitates that complicate downstream processing and filtration steps. These factors collectively result in a manufacturing process that is inefficient, costly, and environmentally unsustainable, creating bottlenecks for procurement managers aiming to reduce the overall cost of goods sold for anticancer drug manufacturing.
The Novel Approach
In stark contrast, the methodology outlined in CN101402655A introduces a groundbreaking aqueous-phase synthesis that eliminates the need for toxic organic solvents entirely by leveraging the solubility properties of alkali metal myristate salts. By reacting the platinum precursor with sodium, potassium, or ammonium myristate in purified water, the process achieves rapid conversion within a timeframe of merely 0.5 to 8 hours, representing a drastic improvement in throughput efficiency. The product precipitates directly from the aqueous solution upon cooling, simplifying isolation to basic filtration and drying operations without the need for complex extraction or distillation units. This innovation not only removes the hazards associated with chloroform handling but also significantly lowers the barrier for commercial scale-up of complex pharmaceutical intermediates by utilizing water as the universal reaction medium. The result is a streamlined, cost-effective pathway that aligns perfectly with modern green chemistry principles and supply chain sustainability goals.
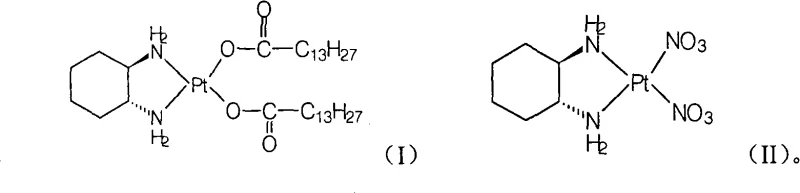
Mechanistic Insights into Aqueous Ligand Exchange and Crystallization
The core chemical transformation in this novel process involves a ligand exchange reaction where the nitrate groups on the platinum center are displaced by myristate anions in an aqueous environment. This mechanism is facilitated by the high solubility of the alkali metal myristate salts in water, which ensures a homogeneous reaction phase that promotes efficient molecular collisions between the platinum complex and the fatty acid ligands. The reaction temperature, controllable between 10°C and 80°C, provides the necessary thermal energy to overcome activation barriers without compromising the stability of the coordination sphere around the platinum atom. Crucially, the use of water as a solvent leverages the hydrophobic effect; as the lipophilic Miriplatin molecule forms, it becomes insoluble in the aqueous medium and spontaneously crystallizes out of the solution. This self-separation mechanism acts as a driving force for the reaction equilibrium, pushing the conversion towards completion and minimizing the presence of unreacted starting materials in the final cake.
From a quality control perspective, this mechanistic pathway offers superior impurity management compared to organic solvent routes. The aqueous environment allows for the effective washing of inorganic byproducts, such as sodium nitrate or potassium nitrate, directly from the filter cake using cold water, thereby achieving high chemical purity levels exceeding 99.0% without extensive recrystallization steps. Moreover, the mild conditions preserve the stereochemical integrity of the chiral (1R,2R)-cyclohexanediamine ligand, ensuring that the optical purity remains above 99.9% as confirmed by chiral HPLC analysis. This level of stereochemical control is paramount for R&D directors evaluating the biological consistency of the API, as any racemization could lead to inactive or toxic isomers. The structural fidelity of the final product is rigorously maintained, ensuring that the therapeutic profile matches the reference standards required for oncology treatments.

How to Synthesize Miriplatin Efficiently
The implementation of this aqueous synthesis route requires precise control over stoichiometry and thermal parameters to maximize yield and purity. The patent specifies a molar ratio of the platinum precursor to the myristate salt between 1:2.0 and 1:3.0, ensuring an excess of the fatty acid ligand to drive the reaction to completion. Detailed standard operating procedures regarding mixing speeds, addition rates, and drying temperatures are critical for reproducing the high yields observed in the experimental examples, which range from approximately 68% to over 88% depending on specific conditions. For technical teams looking to adopt this methodology, adherence to the specified water-to-solid ratios is essential to maintain proper slurry density and heat transfer during the exothermic neutralization and complexation phases. The standardized synthesis steps below outline the critical operational milestones required to achieve consistent commercial-grade output.
- Dissolve Cis-dinitrato((1R,2R)-1,2-cyclohexanediamine)platinum(II) in purified water under heating to ensure complete solubility.
- Add alkali metal myristate salt (Sodium, Potassium, or Ammonium myristate) to the aqueous solution while maintaining temperature between 10°C and 80°C.
- Stir the reaction mixture for 0.5 to 8 hours, then cool to room temperature to precipitate the product, followed by filtration and vacuum drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this aqueous manufacturing process represents a significant opportunity to optimize cost structures and mitigate supply risks associated with volatile organic solvent markets. By eliminating the dependency on chloroform and silver salts, manufacturers can achieve substantial cost savings in raw material procurement and waste disposal fees, directly impacting the bottom line of the production budget. The simplified workflow reduces the requirement for specialized explosion-proof equipment and solvent recovery systems, lowering capital expenditure requirements for facility upgrades. Additionally, the reduced reaction time from days to hours enhances asset utilization, allowing existing reactor trains to produce significantly more batches per year without additional infrastructure investment. These efficiencies translate into a more resilient supply chain capable of meeting fluctuating market demands for high-purity oncology intermediates with greater agility and reliability.
- Cost Reduction in Manufacturing: The replacement of expensive silver myristate with readily available alkali metal myristate salts drastically reduces the direct material cost per kilogram of the final product. Furthermore, the elimination of chloroform removes the need for costly solvent recovery distillation columns and hazardous waste incineration services, leading to significant operational expenditure reductions. The simplified isolation process, which relies on filtration rather than extraction and evaporation, also reduces energy consumption related to heating and vacuum generation. These cumulative factors create a highly competitive cost position for manufacturers adopting this technology, enabling them to offer more attractive pricing models to downstream pharmaceutical clients while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: Utilizing water as the primary solvent removes the supply chain vulnerabilities associated with regulated halogenated solvents, which are often subject to strict quotas and environmental restrictions. The raw materials required, such as sodium myristate and the platinum precursor, are commodity chemicals with stable global availability, ensuring continuous production capability even during market disruptions. The robustness of the aqueous process also minimizes the risk of batch failures due to solvent quality variations, as purified water is easily generated onsite to consistent pharmacopeial standards. This stability is crucial for supply chain heads responsible for guaranteeing uninterrupted delivery of critical cancer treatment intermediates to global formulation partners.
- Scalability and Environmental Compliance: The inherent safety of the water-based system facilitates easier regulatory approval for new manufacturing sites, as it aligns with increasingly stringent global environmental, health, and safety (EHS) regulations. The absence of toxic solvent emissions simplifies the permitting process for plant expansions and reduces the liability footprint of the manufacturing facility. From a scalability perspective, the exothermic nature of the reaction is easily managed in large-scale reactors using standard cooling jackets, avoiding the thermal runaway risks often associated with organic solvent reactions. This makes the technology ideal for commercial scale-up of complex pharmaceutical intermediates, allowing producers to seamlessly transition from pilot plant quantities to multi-ton annual production capacities without re-engineering the core chemistry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and sourcing of Miriplatin using this advanced aqueous methodology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this intermediate into their broader drug development pipelines. The answers reflect the consensus on process performance, quality attributes, and regulatory considerations relevant to the fine chemical industry.
Q: How does the aqueous synthesis method improve impurity control compared to traditional chloroform-based routes?
A: The aqueous method avoids the use of chloroform and silver salts, which are sources of residual solvent and heavy metal impurities. By utilizing water-soluble myristate salts, the reaction proceeds homogeneously, allowing for easier removal of inorganic byproducts through simple water washing, resulting in chemical purity exceeding 99.0%.
Q: What are the scalability advantages of using myristate salts instead of silver myristate?
A: Replacing expensive silver myristate with sodium or potassium myristate drastically reduces raw material costs. Furthermore, the reaction kinetics in water are significantly faster (0.5-8 hours vs. 24+ hours), enabling higher throughput and more efficient use of reactor capacity for commercial scale-up.
Q: Is the optical purity of Miriplatin maintained during the aqueous ligand exchange process?
A: Yes, the mild reaction conditions in aqueous media (10°C to 80°C) preserve the stereochemical integrity of the (1R,2R)-cyclohexanediamine ligand. Patent data confirms optical purity remains above 99.9%, ensuring the biological efficacy required for anticancer applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Miriplatin Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to leverage this advanced aqueous synthesis technology for the commercial production of Miriplatin. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of major pharmaceutical contracts. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest international standards. Our commitment to quality assurance means that every gram of Miriplatin we supply is backed by comprehensive documentation and traceability, giving R&D and procurement teams the confidence they need to proceed with clinical and commercial programs.
We invite global partners to collaborate with us to unlock the full potential of this cost-effective manufacturing route. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized process can enhance your supply chain efficiency. Let us be your trusted partner in delivering high-quality anticancer intermediates that drive innovation and improve patient outcomes worldwide.
