Advanced Green Synthesis of Hydrocortisone Acetate for Commercial Scale-Up
Advanced Green Synthesis of Hydrocortisone Acetate for Commercial Scale-Up
The pharmaceutical industry is constantly seeking robust, environmentally sustainable pathways for the production of critical corticosteroids like Hydrocortisone Acetate, a vital anti-inflammatory agent used globally. Patent CN102827230A introduces a groundbreaking five-step synthetic methodology that transforms Prednisolone Oxide (Pu Shi oxide) into high-purity Hydrocortisone Acetate through a sequence of ketalation, reduction, hydrolysis, iodination, and acetylation. This novel approach strategically bypasses the use of highly toxic pyridine and carcinogenic reagents often found in legacy manufacturing protocols, offering a cleaner alternative that aligns with modern green chemistry principles. By leveraging common auxiliary materials and accessible raw materials, this process not only mitigates environmental pollution but also stabilizes product quality against the color deviations and instability typical of older methods. For R&D directors and procurement specialists, this technology represents a significant leap forward in achieving consistent, high-yield production while adhering to increasingly strict regulatory standards for API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for Hydrocortisone Acetate have long been plagued by significant operational and environmental drawbacks that hinder efficient large-scale production. Historically, the acetylation step relied heavily on pyridine acting as both a solvent and a catalyst, a practice that introduces severe toxicity risks, unpleasant odors, and substantial challenges in solvent recovery and waste management. Furthermore, existing technologies often necessitate the use of hazardous reagents such as bromine water and Nickel-Aluminum catalysts, which generate heavy metal-containing waste streams that are costly and difficult to dispose of safely. The reliance on these dangerous chemicals not only elevates production costs due to rigorous safety and disposal requirements but also compromises the stability of the final product, often resulting in yellowish discoloration and variable quality that fails to meet stringent pharmaceutical specifications. These inherent inefficiencies make conventional methods unsuitable for the demands of modern, high-volume industrial manufacturing where safety and consistency are paramount.
The Novel Approach
In stark contrast, the innovative process detailed in the patent utilizes a sophisticated multi-step sequence starting from Prednisolone Oxide to achieve superior results without the baggage of toxic legacy reagents. The method employs a mild ketalation step using boron trifluoride-ether followed by a precise reduction with lithium aluminum hydride, ensuring selective transformation of the steroid backbone under controlled thermal conditions between 20°C and 40°C. Crucially, the final acetylation is achieved using potassium acetate and glacial acetic acid in DMF, completely eliminating the need for pyridine and its associated hazards. This strategic shift not only simplifies the downstream processing by removing the need for complex heavy metal scavenging but also enhances the overall yield to over 80% with an HPLC purity exceeding 98.5%. By substituting dangerous inputs with common, manageable chemicals, this approach offers a scalable, cost-effective solution that significantly lowers the barrier for compliant commercial production.
Mechanistic Insights into Steroid Functionalization and Side-Chain Modification
The core of this synthetic strategy lies in the precise manipulation of the steroid nucleus, specifically the protection and selective reduction of the ketone functionalities to establish the correct stereochemistry at the C11 and C17 positions. The process initiates with the formation of a 3,20-diethylene glycol ketal, which serves as a robust protecting group to shield the diketone system during the subsequent vigorous reduction with lithium aluminum hydride. This protection is vital as it prevents unwanted side reactions at the C3 and C20 positions, allowing the hydride reagent to selectively target the C11 keto group to form the crucial 11β-hydroxyl moiety. Following reduction, a controlled acidic hydrolysis step removes the ketal protecting groups to regenerate the 3,20-diketone system, yielding the intermediate 11β,17α-dihydroxy-4-pregnene-3,20-diketone with high fidelity. This sequence ensures that the delicate balance of functional groups required for biological activity is maintained throughout the synthesis, minimizing the formation of regio-isomeric impurities.
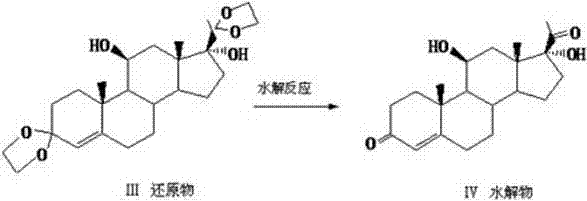
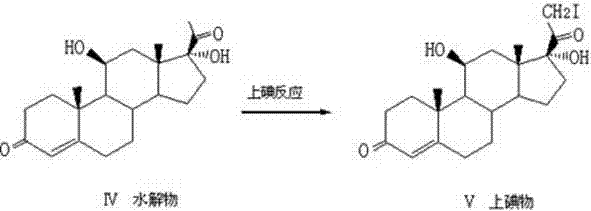
Following the establishment of the core hydroxyl pattern, the synthesis proceeds to the critical functionalization of the C21 side chain, which distinguishes Hydrocortisone Acetate from its precursors. The intermediate undergoes a specialized iodination reaction utilizing iodine and calcium chloride in a methanol-chloroform system at low temperatures of 0°C to 5°C, introducing an iodine atom at the C21 position to create a reactive leaving group. This 21-iodo intermediate is then subjected to a nucleophilic substitution reaction where potassium acetate in DMF displaces the iodine atom, effectively installing the acetate ester group essential for the drug's pharmacokinetic profile. This two-step functionalization via an iodo-intermediate is superior to direct acetylation methods as it offers better control over reaction kinetics and minimizes the formation of di-acetylated byproducts. The entire pathway is designed to maximize atom economy and minimize waste, resulting in a final product that is chemically pure and structurally identical to the reference standard.
How to Synthesize Hydrocortisone Acetate Efficiently
The synthesis of Hydrocortisone Acetate via this patented route involves a carefully orchestrated series of reactions that balance reactivity with selectivity to ensure high yields and purity. The process begins with the ketal protection of the starting material, followed by reduction and hydrolysis to set the stereochemistry, and concludes with side-chain iodination and acetylation. Each step requires precise control of temperature and stoichiometry, particularly the low-temperature iodination and the reflux conditions for acetylation, to prevent degradation of the sensitive steroid skeleton. While the general workflow is straightforward, the specific ratios of reagents such as the 1:2:2 ratio of starting material to ethylene glycol and orthoformate in the first step are critical for success. For a comprehensive understanding of the exact operational parameters and workup procedures required to replicate this high-efficiency process, please refer to the detailed standardized synthesis guide provided below.
- Perform ketal protection of the 3,20-diketone using ethylene glycol and triethyl orthoformate with BF3-ether catalyst.
- Execute selective reduction of the 11-keto group using Lithium Aluminum Hydride in THF followed by acidic hydrolysis to restore ketones.
- Conduct C21 iodination using iodine and calcium chloride, followed by nucleophilic substitution with potassium acetate to finalize the acetate group.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers profound advantages in terms of cost structure, risk mitigation, and operational continuity. By eliminating the need for pyridine and heavy metal catalysts like Nickel-Aluminum, manufacturers can drastically reduce the costs associated with hazardous waste disposal, solvent recovery systems, and specialized safety equipment. The reliance on common, commercially available reagents such as calcium chloride, iodine, and potassium acetate ensures a stable supply chain that is less susceptible to the volatility and sourcing difficulties often associated with exotic or highly regulated chemicals. Furthermore, the avoidance of toxic substances simplifies the regulatory compliance landscape, reducing the administrative burden and potential liabilities related to environmental discharge and worker safety. This streamlined chemical profile translates directly into a more resilient manufacturing operation capable of sustaining long-term production schedules without interruption.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents such as pyridine and nickel-aluminum catalysts removes the necessity for complex and costly waste treatment infrastructure. Without the need to recover toxic solvents or remove heavy metal residues from the final product, the downstream processing becomes significantly simpler and cheaper, leading to substantial overall cost savings. Additionally, the high yield reported in the patent means that less raw material is wasted per unit of product, further optimizing the cost of goods sold and improving profit margins for large-scale producers.
- Enhanced Supply Chain Reliability: The process utilizes basic industrial chemicals like ethylene glycol, hydrochloric acid, and acetic acid, which are produced in massive volumes globally and are unlikely to face supply shortages. This reliance on commodity chemicals rather than specialized, single-source reagents diversifies the supply base and reduces the risk of production stoppages due to raw material unavailability. Consequently, manufacturers can maintain consistent inventory levels and meet delivery commitments with greater confidence, ensuring a steady flow of high-quality intermediates to downstream API formulators.
- Scalability and Environmental Compliance: The reaction conditions, ranging from ambient temperatures to mild heating, are inherently safe and easily scalable from pilot plants to multi-ton commercial reactors without requiring exotic high-pressure or cryogenic equipment. The absence of carcinogenic reagents and heavy metals aligns perfectly with modern environmental regulations, facilitating easier permitting and reducing the risk of fines or shutdowns due to non-compliance. This environmental compatibility makes the process future-proof, allowing companies to expand capacity sustainably while maintaining a positive corporate social responsibility profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology for Hydrocortisone Acetate. These answers are derived directly from the experimental data and comparative analysis presented in the patent documentation to provide clarity on the process benefits. Understanding these specifics is crucial for technical teams evaluating the feasibility of adopting this route for their own manufacturing portfolios.
Q: How does this process improve upon traditional pyridine-based acetylation methods?
A: This method eliminates the use of pyridine, a toxic solvent with a noxious odor that is difficult to recover and poses significant environmental hazards. By replacing it with safer solvents and reagents like glacial acetic acid and potassium acetate, the process drastically reduces waste treatment costs and operator exposure risks.
Q: What represents the key advantage regarding heavy metal contamination?
A: Unlike conventional routes that may utilize Nickel-Aluminum catalysts requiring complex removal steps, this synthesis avoids transition metal catalysts in the critical functionalization steps. This simplifies the purification workflow and ensures the final API meets stringent heavy metal limits without expensive scavenging processes.
Q: What purity levels can be expected from this synthetic route?
A: The patent data indicates that the optimized process consistently yields Hydrocortisone Acetate with an HPLC content exceeding 98.5%. The overall yield is reported to be greater than 80%, demonstrating both high efficiency and excellent impurity control suitable for pharmaceutical grade production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydrocortisone Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced, environmentally responsible synthetic routes to meet the evolving demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex steroid syntheses like the one described in CN102827230A can be translated efficiently from the laboratory to full-scale manufacturing. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical techniques to verify every batch. By partnering with us, you gain access to a supply chain that prioritizes both quality and sustainability, mitigating the risks associated with traditional, polluting manufacturing methods.
We invite you to engage with our technical procurement team to discuss how this innovative process can be tailored to your specific volume requirements and cost targets. Please contact us to request a Customized Cost-Saving Analysis that details the potential economic benefits of switching to this greener protocol. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise can secure your supply of high-purity Hydrocortisone Acetate while optimizing your overall production economics.
