Advanced Manufacturing of Hydrocortisone: A Breakthrough in Purity and Process Efficiency
The pharmaceutical landscape for corticosteroid manufacturing is undergoing a significant transformation driven by the urgent need for greener, more efficient synthetic pathways. Patent CN114075258B introduces a groundbreaking preparation method for hydrocortisone that fundamentally alters the traditional production paradigm by eliminating the reliance on toxic halogenating agents. This innovation addresses critical bottlenecks in the synthesis of this vital adrenocortical hormone, which is essential for treating a wide spectrum of conditions ranging from adrenal insufficiency to severe allergic reactions. By shifting away from hazardous iodine-based chemistry toward a controlled hypochlorite oxidation and resin-mediated hydrolysis strategy, this technology offers a robust solution for achieving ultra-high purity standards required by modern regulatory bodies. The method not only simplifies the operational workflow but also drastically reduces the environmental footprint associated with heavy metal and halogen waste disposal. For industry stakeholders, this represents a pivotal opportunity to upgrade manufacturing capabilities while ensuring a stable supply of high-quality active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of hydrocortisone has been plagued by complex multi-step sequences that rely heavily on toxic and environmentally unfriendly reagents such as molecular iodine. Traditional routes, often starting from precursors like 17-hydroxy-4,9-diene-pregna-3,20-dione, necessitate bromination, debromination, and subsequent iodination steps that introduce significant safety risks and waste management challenges. A major technical hurdle in these legacy processes is the formation of structurally similar impurities, specifically anecortave acetate derivatives, which share nearly identical physical properties with the target molecule. As illustrated in the reaction pathways below, these impurities are notoriously difficult to separate using standard purification techniques, leading to compromised product quality and reduced overall yields. The persistence of these contaminants not only threatens patient safety but also creates substantial downstream processing costs for manufacturers attempting to meet stringent pharmacopeial specifications.
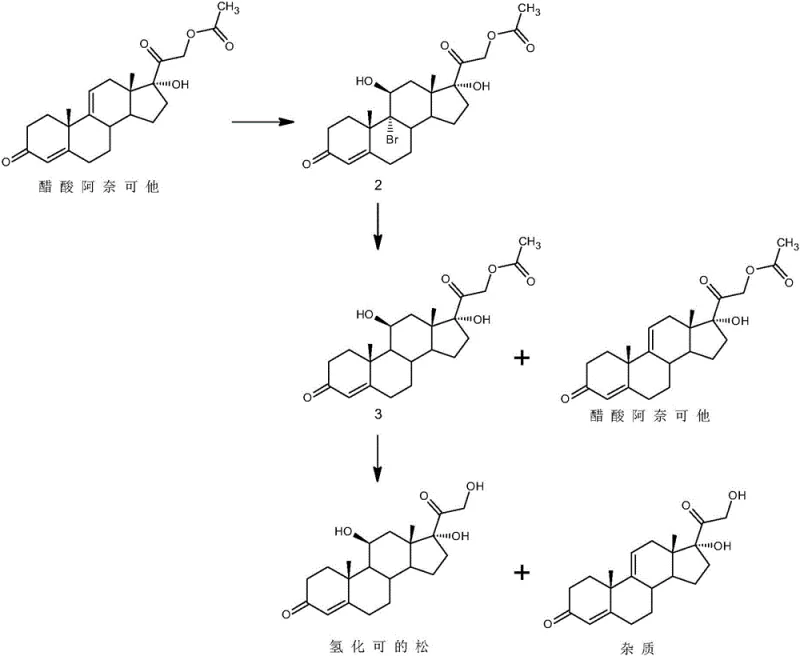
The Novel Approach
In stark contrast to these cumbersome legacy methods, the novel approach detailed in the patent utilizes a streamlined two-step protocol that prioritizes selectivity and operational simplicity. The core innovation lies in the strategic use of sodium hypochlorite under weakly acidic conditions to selectively oxidize and modify the problematic impurities without degrading the desired hydrocortisone acetate backbone. This chemical modification transforms the stubborn contaminants into polar derivatives that can be effortlessly removed during a subsequent recrystallization phase. Furthermore, the replacement of harsh alkaline hydrolysis with a mild IRA-400 resin-catalyzed process prevents the formation of secondary degradation products often seen in base-mediated reactions. This methodology ensures that the final product achieves exceptional purity levels while maintaining a high mass recovery rate, effectively solving the long-standing issue of impurity control in corticosteroid manufacturing.
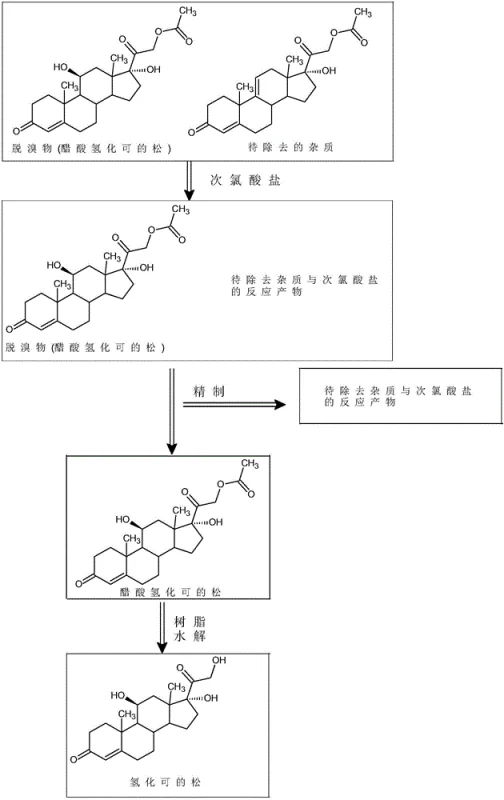
Mechanistic Insights into Hypochlorite-Mediated Impurity Removal
The chemical elegance of this process stems from the precise control of oxidative potential generated in situ. By introducing glacial acetic acid prior to the addition of sodium hypochlorite, the reaction system establishes a buffered weak acid environment that favors the formation of hypochlorous acid rather than free chlorine gas or highly reactive hypochlorite ions. This specific species acts as a selective oxidant that targets the electron-rich double bonds or specific functional groups present in the impurity molecules, such as anecortave acetate, converting them into more polar epoxides or chlorohydrins. The reaction kinetics are carefully managed by maintaining temperatures between 15°C and 40°C, ensuring that the oxidation is complete without inducing over-oxidation of the steroid nucleus. Once the conversion is verified via HPLC, the residual oxidizing power is immediately quenched using a sodium metabisulfite solution, which neutralizes any remaining hypochlorous acid and prevents further unwanted side reactions during the workup phase.
Following the oxidative purification, the transition to the final hydrocortisone product is achieved through a sophisticated solid-phase hydrolysis mechanism. Unlike traditional liquid-base hydrolysis which can lead to epimerization or dehydration of the sensitive steroid skeleton, the use of IRA-400 anion exchange resin provides a heterogeneous catalytic surface that facilitates the cleavage of the acetate ester group under mild conditions. The resin acts as a proton acceptor, promoting nucleophilic attack by water molecules on the carbonyl carbon of the ester linkage while sequestering the resulting acetate ions. This heterogeneous nature allows for easy separation of the catalyst via simple filtration, eliminating the need for complex neutralization and extraction steps that typically generate large volumes of saline wastewater. The result is a cleaner reaction profile that preserves the stereochemical integrity of the 11-beta-hydroxyl and 17-alpha-hydroxyl groups, which are critical for the biological activity of the final drug substance.
How to Synthesize Hydrocortisone Efficiently
The implementation of this advanced synthesis route requires careful attention to solvent selection and stoichiometric ratios to maximize the efficiency of the impurity removal step. The process begins by dissolving the crude hydrocortisone acetate in a binary solvent system comprising dichloromethane and a lower alcohol such as methanol or isopropanol, which ensures complete solubility of the steroid substrate. Following the addition of the acid buffer and the controlled dosing of the hypochlorite solution, the reaction mixture is monitored until the disappearance of the starting impurities is confirmed.
- Dissolve crude hydrocortisone acetate in a mixed organic solvent, add glacial acetic acid to create a weak acid buffer, and treat with sodium hypochlorite to oxidize and remove structurally similar impurities.
- Neutralize excess oxidant with sodium metabisulfite, recrystallize the purified acetate, and subsequently perform hydrolysis using IRA-400 resin at low temperatures to yield the final hydrocortisone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iodine-free methodology translates into tangible strategic benefits regarding cost stability and operational reliability. By eliminating the dependency on volatile iodine markets and the specialized equipment required for handling corrosive halogens, manufacturers can significantly reduce their exposure to raw material price fluctuations and regulatory compliance costs. The simplified workflow, which consolidates multiple purification stages into a single oxidative treatment, inherently lowers the consumption of utilities such as steam and cooling water, thereby driving down the overall cost of goods sold. Furthermore, the reduction in hazardous waste generation simplifies the environmental permitting process and minimizes the liabilities associated with the disposal of toxic effluents, making the facility more sustainable and resilient against tightening environmental regulations.
- Cost Reduction in Manufacturing: The elimination of expensive iodine reagents and the associated complex purification steps leads to a direct reduction in raw material expenditures. Additionally, the use of common, commodity-grade chemicals like sodium hypochlorite and acetic acid ensures a stable and low-cost supply chain for reagents. The simplified downstream processing also reduces the labor hours and solvent volumes required for isolation, contributing to substantial overall cost savings in the production of high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Relying on widely available industrial chemicals rather than specialized halogenating agents mitigates the risk of supply disruptions caused by geopolitical factors or manufacturer shortages. The robustness of the resin-catalyzed hydrolysis step ensures consistent batch-to-batch quality, reducing the frequency of failed batches and the need for re-processing. This reliability allows for more accurate production planning and shorter lead times for delivering critical hormone intermediates to downstream formulation partners.
- Scalability and Environmental Compliance: The mild reaction conditions, operating primarily at ambient or slightly elevated temperatures, make this process highly scalable from pilot plant to commercial tonnage without requiring exotic high-pressure or cryogenic equipment. The significant reduction in toxic waste streams aligns with green chemistry principles, facilitating easier compliance with increasingly strict environmental discharge standards. This environmental compatibility enhances the corporate social responsibility profile of the manufacturing site and future-proofs the operation against potential carbon taxes or waste levies.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and validation of this novel hydrocortisone synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of the method's superiority over existing state-of-the-art processes.
Q: Why is the traditional iodine-based synthesis route being replaced?
A: Traditional routes rely on toxic iodine for substitution reactions, which generates difficult-to-remove impurities and poses significant environmental and safety hazards. The new method eliminates iodine entirely.
Q: How does the hypochlorite step improve product purity?
A: By generating hypochlorous acid in a weakly acidic environment, the process selectively oxidizes specific structural impurities that are chemically similar to the target molecule, converting them into forms that are easily separated during recrystallization.
Q: What are the expected purity and yield specifications for this method?
A: This patented process consistently achieves a hydrocortisone acetate purity exceeding 99.2% and a final hydrocortisone purity greater than 99.5%, with total mass yields surpassing 75%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydrocortisone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the one described in CN114075258B can be seamlessly transferred to full-scale manufacturing. We are committed to delivering products with stringent purity specifications, utilizing our rigorous QC labs to verify that every batch meets the highest international standards for hormonal intermediates. Our infrastructure is designed to handle complex organic syntheses with a focus on safety, quality, and environmental stewardship.
We invite potential partners to engage with our technical procurement team to discuss how this advanced hydrocortisone manufacturing route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will enhance the efficiency and sustainability of your API production capabilities.
