Advanced Peptide Coupling Strategy for Scalable Quinazoline Derivative Production
Advanced Peptide Coupling Strategy for Scalable Quinazoline Derivative Production
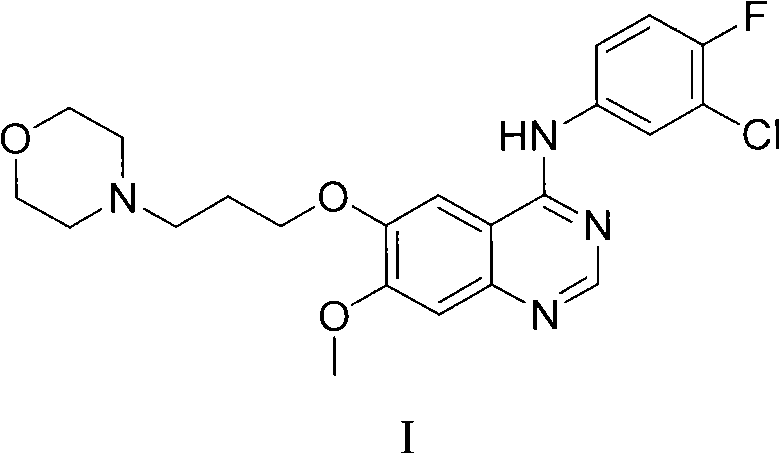
The pharmaceutical industry continuously seeks robust synthetic pathways for critical oncology therapeutics, particularly tyrosine kinase inhibitors targeting the epidermal growth factor receptor (EGFR). ZD1839, widely known as Gefitinib, represents a cornerstone in the treatment of non-small cell lung cancer, functioning by suppressing the ATP binding site of the EGFR tyrosine kinase domain. The preparation of such high-value quinazoline derivatives has historically faced significant challenges regarding environmental impact and process efficiency. Patent CN102153519B discloses a transformative preparation method that addresses these longstanding issues by introducing a novel peptide coupling strategy. This technical breakthrough offers a viable alternative to traditional chlorination routes, providing a pathway characterized by mild reaction conditions, reduced side reactions, and superior yield profiles. For global procurement and R&D teams, understanding this shift from harsh halogenation to catalytic coupling is essential for securing a reliable quinazoline derivative supplier capable of meeting stringent regulatory and volume demands.
The Limitations of Conventional Methods vs. The Novel Approach
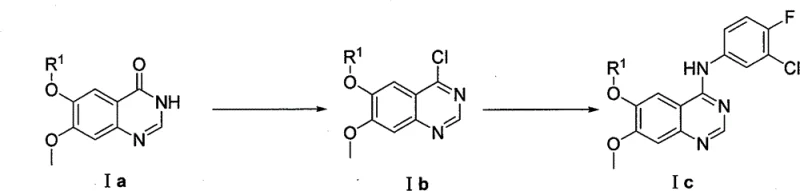
The Limitations of Conventional Methods
Historically, the synthesis of key quinazoline intermediates, specifically the conversion of quinazolinones to 4-anilinoquinazolines, has relied heavily on activation via chlorination. As illustrated in prior art references such as EP566226 and WO9633980, the standard protocol involves treating the quinazolinone precursor (Compound Ia) with aggressive chlorinating agents like phosphorus pentachloride, phosphorus oxychloride (POCl3), thionyl chloride, or oxalyl chloride. This approach presents severe drawbacks for modern green chemistry standards and industrial safety. The reaction conditions are inherently harsh, often requiring elevated temperatures and generating substantial quantities of corrosive acidic waste that necessitates complex neutralization and disposal procedures. Furthermore, the subsequent nucleophilic substitution with aniline derivatives frequently suffers from long reaction times and the formation of numerous by-products, leading to lower overall yields and challenging purification processes. These factors collectively increase the cost of goods sold (COGS) and introduce significant supply chain risks related to waste management compliance.
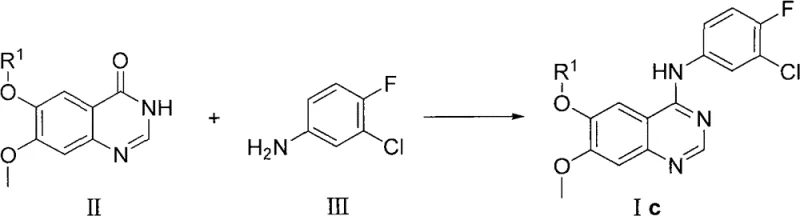
The Novel Approach
In stark contrast to the legacy chlorination methodologies, the invention described in CN102153519B proposes a direct condensation strategy that bypasses the isolation of unstable chloro-intermediates. The novel approach utilizes a peptide coupling mechanism to directly link the quinazolinone core (Compound II) with the aniline nucleophile (Compound III). This method operates under significantly milder conditions, typically employing aprotic polar solvents such as acetonitrile, DMSO, or DMF. By leveraging peptide coupling agents, the reaction activates the carbonyl functionality of the quinazolinone in situ, facilitating a smooth nucleophilic attack by the aniline without the need for prior halogenation. This paradigm shift not only simplifies the operational workflow by reducing the number of unit operations but also drastically minimizes the generation of hazardous inorganic waste. The result is a cleaner reaction profile with fewer impurities, directly translating to easier downstream processing and higher quality final products suitable for pharmaceutical applications.
Mechanistic Insights into Peptide Coupling Cyclization
The core of this technological advancement lies in the activation of the lactam carbonyl group within the quinazolinone ring system. In the presence of a peptide coupling agent, such as benzotriazole-1-yloxytris(dimethylamino)phosphonium hexafluorophosphate (BOP), the oxygen atom of the carbonyl group is activated to form a highly reactive intermediate species. This activation lowers the energy barrier for nucleophilic attack, allowing the amine group of the 3-chloro-4-fluoroaniline (Compound III) to displace the activated leaving group efficiently. The reaction is further promoted by the presence of a non-nucleophilic base, such as 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU) or triethylamine, which serves to deprotonate the incoming aniline and neutralize the acid by-products generated during the coupling process. This mechanistic pathway avoids the formation of highly reactive and toxic chloro-species, thereby enhancing the safety profile of the manufacturing process while maintaining high chemoselectivity for the desired 4-position substitution.
From an impurity control perspective, this mechanism offers distinct advantages over the traditional route. In chlorination-based syntheses, over-chlorination or incomplete substitution often leads to difficult-to-remove halogenated impurities that can persist through multiple crystallization steps. The peptide coupling route, however, generates by-products that are typically organic phosphorous or urea derivatives, which are more soluble in aqueous washes or can be easily separated during the extraction phase. The patent data indicates that reaction temperatures can be finely tuned between 40°C and 120°C to optimize kinetics without triggering thermal degradation of the sensitive quinazoline scaffold. This precise control over reaction parameters ensures a consistent impurity profile, which is critical for meeting the rigorous specifications required by regulatory bodies for active pharmaceutical ingredients (APIs) and their advanced intermediates.
How to Synthesize Quinazoline Derivatives Efficiently
The practical implementation of this synthesis involves a straightforward one-pot procedure that is highly amenable to standard reactor setups found in fine chemical facilities. The process begins with the suspension of the quinazolinone substrate in a dry aprotic solvent, followed by the sequential addition of the coupling agent, base, and aniline partner. The reaction mixture is then agitated under an inert nitrogen atmosphere to prevent moisture interference, which could hydrolyze the activated intermediate. Monitoring is typically conducted via HPLC or TLC until the starting material is fully consumed. Following the reaction, the workup involves solvent removal, liquid-liquid extraction using ethyl acetate and aqueous base to remove acidic residues, and final purification via recrystallization from ethanol. This streamlined protocol reduces the operational complexity typically associated with multi-step heterocycle functionalization.
- Combine the quinazolinone substrate (Compound II), aniline derivative (Compound III), organic solvent, base, and peptide coupling agent in a reaction vessel under inert atmosphere.
- Stir the reaction mixture at a temperature ranging from room temperature to 120°C, monitoring progress until completion.
- Perform workup by evaporating solvent, extracting with ethyl acetate, washing with aqueous base, drying, and recrystallizing to obtain the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this peptide coupling methodology represents a strategic opportunity to optimize the sourcing of critical oncology intermediates. The elimination of hazardous chlorinating reagents removes a major bottleneck in waste disposal logistics, significantly reducing the environmental compliance burden and associated costs. Furthermore, the use of commercially available coupling agents and bases ensures a stable supply of raw materials, mitigating the risk of production delays caused by the scarcity of specialized reagents. The mild reaction conditions also imply lower energy consumption compared to the high-temperature reflux often required for chlorination, contributing to a more sustainable and cost-effective manufacturing footprint. These factors collectively enhance the reliability of the supply chain, ensuring continuous availability of high-purity materials for downstream drug formulation.
- Cost Reduction in Manufacturing: The transition away from phosphorus-based chlorinating agents eliminates the need for expensive corrosion-resistant equipment and complex scrubbing systems required to handle toxic gases like HCl and SO2. By simplifying the synthetic route to a direct coupling reaction, manufacturers can reduce the number of processing steps, thereby lowering labor costs and increasing overall plant throughput. The higher yields reported in the patent examples suggest a more efficient utilization of raw materials, which directly impacts the variable cost per kilogram of the final intermediate. Additionally, the simplified purification process reduces solvent consumption and waste treatment expenses, driving down the total cost of ownership for the production line.
- Enhanced Supply Chain Reliability: The reliance on standard organic solvents like acetonitrile and DMF, along with common bases like DBU and triethylamine, ensures that the supply chain is not vulnerable to the volatility of specialized reagent markets. The robustness of the reaction conditions allows for flexible scheduling and easier scale-up from pilot to commercial batches without significant re-optimization. This stability is crucial for maintaining just-in-time delivery schedules for pharmaceutical clients who require consistent quality and quantity. The ability to source raw materials from multiple global vendors further diversifies supply risk, ensuring business continuity even in the face of regional disruptions or logistical challenges.
- Scalability and Environmental Compliance: The process is explicitly designed with industrial suitability in mind, avoiding the use of strong Lewis acids like aluminum chloride that generate massive amounts of solid waste difficult to dispose of safely. The liquid-phase nature of the reaction and workup facilitates automation and continuous processing potential, which are key drivers for modern scalable manufacturing. From an environmental standpoint, the reduction in hazardous waste generation aligns with increasingly strict global regulations on chemical emissions and effluent discharge. This compliance advantage not only protects the manufacturer from regulatory fines but also enhances the brand reputation of the supply chain partners as responsible stewards of environmental safety.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical benefits and operational parameters of the technology. Understanding these details is vital for technical teams evaluating the feasibility of integrating this method into existing production workflows or for procurement specialists assessing the long-term viability of suppliers utilizing this approach.
Q: What are the primary advantages of this peptide coupling method over traditional chlorination routes?
A: The primary advantage is the elimination of harsh chlorinating agents like phosphorus oxychloride or thionyl chloride, which generate significant hazardous waste. This new method utilizes mild peptide coupling conditions, resulting in fewer side reactions, higher purity, and reduced environmental impact suitable for industrial scale-up.
Q: Which coupling agents and bases are preferred for this synthesis?
A: The patent specifies benzotriazole-1-yloxytris(dimethylamino)phosphonium hexafluorophosphate (BOP) as a preferred coupling agent. Suitable bases include 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU), triethylamine, or sodium hydride, depending on the specific substrate reactivity and solvent system employed.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is explicitly designed for industrial suitability. It uses commercially available raw materials, operates under relatively mild temperatures (40-120°C), and avoids difficult post-treatment steps associated with strong Lewis acids, making it highly scalable for reliable supply chain integration.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinazoline Derivative Supplier
As the demand for targeted cancer therapies continues to grow, the need for efficient and sustainable manufacturing processes for key intermediates like quinazoline derivatives becomes paramount. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging advanced synthetic methodologies such as the peptide coupling route described in CN102153519B to deliver superior value to our global partners. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of multinational pharmaceutical companies. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of intermediate meets the highest standards for safety and efficacy required for clinical and commercial applications.
We invite you to collaborate with us to explore how this innovative synthesis technology can optimize your supply chain and reduce your overall manufacturing costs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating the tangible economic benefits of switching to this greener, more efficient route. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us partner with you to accelerate the development and delivery of life-saving medicines to patients worldwide.
