Advanced Electroreduction Technology for High-Purity Gefitinib Intermediate Manufacturing and Commercial Scale-Up
Advanced Electroreduction Technology for High-Purity Gefitinib Intermediate Manufacturing and Commercial Scale-Up
The pharmaceutical industry is constantly seeking more sustainable and efficient pathways for the synthesis of critical oncology drug precursors. Patent CN110777391B introduces a groundbreaking electroreduction preparation method for the key gefitinib intermediate, methyl 2-amino-4-methoxy-5-(3-morpholinylpropoxy)benzoate. This technology represents a significant paradigm shift from traditional chemical reduction methods, utilizing electrons as clean reagents to drive the transformation of nitro groups to amino groups with exceptional selectivity. By leveraging precise control over electrode potential and current density, this process achieves high conversion rates and purity levels while eliminating the need for toxic reducing agents or expensive noble metal catalysts. For global supply chain leaders, this innovation offers a robust solution for cost reduction in pharmaceutical intermediate manufacturing, ensuring a stable supply of high-quality materials essential for the production of EGFR tyrosine kinase inhibitors.
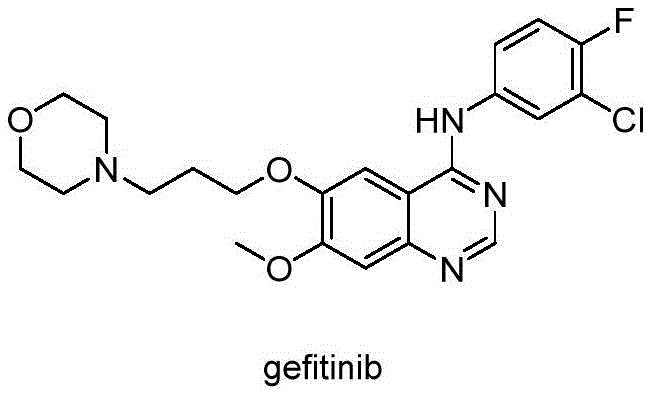
Gefitinib is a vital selective Epidermal Growth Factor Receptor (EGFR) tyrosine kinase inhibitor used extensively in the treatment of solid tumors such as lung and breast cancer. The synthesis of this active pharmaceutical ingredient relies heavily on the availability of high-purity intermediates, specifically the aniline derivative shown in the structural analysis. Traditional synthetic routes often face challenges related to impurity profiles and environmental impact, which can delay regulatory approvals and increase production costs. The electrochemical approach detailed in this patent addresses these pain points directly by providing a greener, safer, and more controllable alternative. As a reliable pharmaceutical intermediates supplier, understanding and adopting such advanced technologies is crucial for maintaining competitiveness in the global market and meeting the rigorous quality standards demanded by top-tier drug manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the reduction of the nitro precursor to the corresponding aniline intermediate has been achieved through catalytic hydrogenation or chemical reduction using inorganic salts. Catalytic hydrogenation typically employs palladium on carbon (Pd/C) under high pressure, which poses significant safety risks due to the handling of hydrogen gas and requires specialized, expensive equipment. Furthermore, the interaction between the palladium catalyst and the intermediate can form stable complexes that are difficult to separate, leading to potential heavy metal contamination in the final API, a critical failure point for oncology drugs. Alternatively, chemical reduction using sodium dithionite generates substantial amounts of inorganic waste and sulfur-containing byproducts, creating a heavy burden on wastewater treatment facilities and violating modern green chemistry principles. These conventional methods often struggle to balance yield with purity, necessitating complex downstream purification steps that erode profit margins and extend lead times.
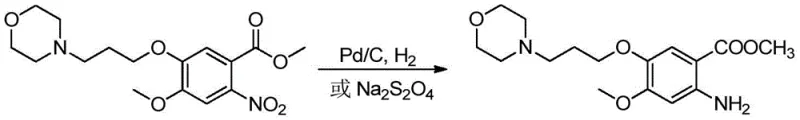
The Novel Approach
In stark contrast, the electroreduction method disclosed in patent CN110777391B utilizes a diaphragm electrolytic cell to perform the reduction in an acidic aqueous medium without any external chemical reducing agents. By applying a controlled cathode working voltage between 1.00V and 2.50V and maintaining a current density of 25.0mA/cm² to 250.0mA/cm², the process selectively reduces the nitro group to an amino group with minimal formation of side products. This technique operates under mild temperatures ranging from 25°C to 80°C and at atmospheric pressure, drastically improving operational safety and reducing energy consumption. The absence of organic solvents and toxic reagents simplifies the post-reaction workup, allowing for straightforward isolation of the product through alkalization and extraction. This novel approach not only enhances the purity of the gefitinib intermediate but also aligns perfectly with the industry's move towards sustainable and environmentally friendly manufacturing processes.
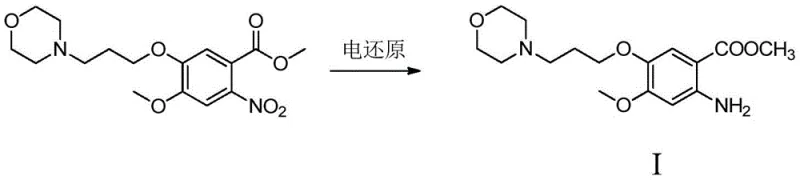
Mechanistic Insights into Electrochemical Nitro Reduction
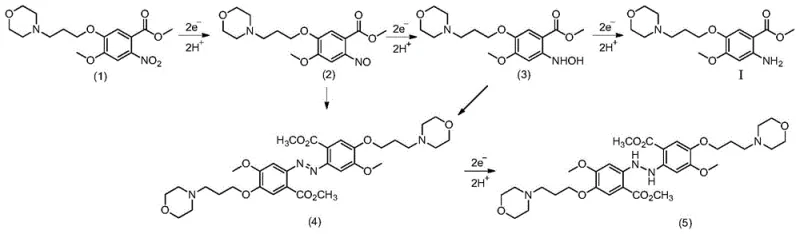
The core of this technology lies in the precise manipulation of electron transfer at the cathode surface. In the acidic catholyte, the nitro compound accepts electrons and protons in a stepwise manner to form the desired amine. The overall cathode reaction involves the transfer of 12 electrons and 12 protons to convert two molecules of the nitro substrate into two molecules of the amino product and water. The use of a diaphragm cell is critical here, as it separates the anodic and cathodic compartments, preventing the oxidation of the newly formed amine at the anode and ensuring high current efficiency. The choice of electrode materials, such as copper, titanium mesh, or graphite for the cathode and Dimensionally Stable Anodes (DSA) for the anode, plays a pivotal role in minimizing overpotential and maximizing reaction rates. This level of control allows manufacturers to fine-tune the process parameters to optimize yield and selectivity, a flexibility that is often lacking in batch chemical reductions.
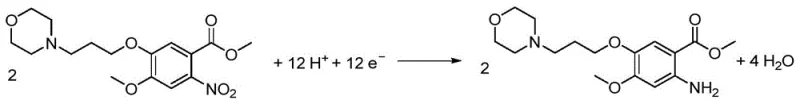
A major advantage of this electrochemical method is its ability to suppress the formation of common reduction byproducts such as nitroso compounds, hydroxylamines, and azo dimers. In traditional chemical reductions, controlling the reaction to stop at the amine stage without over-reduction or coupling is challenging. However, by strictly regulating the electrode potential within the specified range, the electroreduction process favors the direct pathway to the amine while kinetically hindering the condensation reactions that lead to azo impurities. The patent data indicates that under optimal conditions, the selectivity for the main product is maximized, and the yield of byproducts represented by structural formulas (2) through (5) is minimized. This inherent selectivity results in a cleaner crude product, reducing the load on crystallization and chromatography steps, which is a significant benefit for R&D teams focused on impurity profiling and process validation.
How to Synthesize Methyl 2-amino-4-methoxy-5-(3-morpholinylpropoxy)benzoate Efficiently
The synthesis of this critical gefitinib intermediate via electroreduction is a streamlined process that integrates seamlessly into existing pharmaceutical manufacturing workflows. The procedure begins with the preparation of the electrolyte solutions, where the nitro precursor is dissolved in an acidic aqueous medium such as phosphoric or sulfuric acid to serve as the catholyte. The electrolytic cell is then assembled with appropriate electrodes, and the reaction is initiated by applying a constant voltage or current. Monitoring the reaction progress is straightforward using Thin Layer Chromatography (TLC), allowing operators to stop the electrolysis precisely when the starting material is consumed. This operational simplicity, combined with the high yields reported in the patent examples (exceeding 97%), makes it an attractive option for both laboratory-scale optimization and large-scale production. For detailed standard operating procedures and specific parameter settings, please refer to the technical guide below.
- Prepare the catholyte by dissolving 4-methoxy-5-(3-morpholinylpropoxy)-2-nitrobenzoic acid methyl ester in an acidic aqueous solution (e.g., phosphoric or sulfuric acid) within a diaphragm electrolytic cell.
- Set up the electrolytic cell with a copper or titanium mesh cathode and a DSA anode, maintaining the cathode working voltage between 1.00V and 2.50V relative to a reference electrode.
- Conduct electrolysis at a current density of 25.0mA/cm² to 250.0mA/cm² and a temperature of 25°C to 80°C until TLC indicates complete conversion, then isolate the product via alkalization and extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electroreduction technology translates into tangible strategic advantages beyond mere technical feasibility. The elimination of expensive palladium catalysts and high-pressure hydrogenation equipment significantly lowers the capital expenditure and operational costs associated with the manufacturing of this intermediate. Furthermore, the removal of hazardous chemical reducing agents like sodium dithionite reduces the costs related to hazardous waste disposal and environmental compliance, contributing to a leaner and more sustainable cost structure. The mild reaction conditions also enhance equipment longevity and reduce maintenance downtime, ensuring a more reliable and continuous supply of materials. These factors collectively contribute to substantial cost savings and improved margin protection for downstream API manufacturers.
- Cost Reduction in Manufacturing: The electroreduction process fundamentally alters the cost drivers of the synthesis by removing the dependency on precious metal catalysts and complex high-pressure reactors. Without the need for palladium recovery systems or the purchase of stoichiometric amounts of chemical reducing agents, the variable cost per kilogram of the intermediate is drastically reduced. Additionally, the simplified workup procedure, which avoids complex filtration steps to remove catalyst residues, reduces labor and solvent consumption. This economic efficiency allows suppliers to offer more competitive pricing while maintaining healthy margins, a critical factor for procurement teams managing tight budgets in the generic drug sector.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the inherent safety and simplicity of the electrochemical process. Unlike catalytic hydrogenation, which carries risks associated with high-pressure hydrogen gas and requires specialized safety protocols, electroreduction operates at atmospheric pressure with aqueous solutions. This reduces the risk of unplanned shutdowns due to safety incidents and simplifies regulatory inspections. Moreover, the raw materials required, such as electricity and common acids, are readily available and less subject to geopolitical supply disruptions compared to specialized catalysts. This stability ensures consistent delivery schedules and reduces the lead time for high-purity pharmaceutical intermediates, enabling manufacturers to respond more agilely to market demand fluctuations.
- Scalability and Environmental Compliance: Scaling electrochemical processes is generally more linear and predictable than scaling batch chemical reactions, as it often involves adding more electrode surface area or cells in parallel rather than dealing with heat and mass transfer limitations in large vessels. The process generates minimal waste, primarily consisting of benign aqueous streams that are easier to treat than the sulfur-laden waste from dithionite reduction. This aligns with increasingly stringent global environmental regulations, reducing the risk of fines or production halts due to non-compliance. For supply chain heads, this means a future-proof manufacturing route that can be expanded to meet growing demand for gefitinib without encountering significant environmental bottlenecks or requiring massive infrastructure upgrades.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electroreduction synthesis of gefitinib intermediates. These insights are derived directly from the experimental data and claims presented in patent CN110777391B, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing supply chains and quality assurance frameworks. The answers highlight the specific advantages in terms of purity, safety, and operational efficiency that distinguish this method from legacy technologies.
Q: How does electroreduction improve the purity of gefitinib intermediates compared to catalytic hydrogenation?
A: Electroreduction eliminates the use of palladium catalysts, thereby avoiding the formation of difficult-to-remove metal complexes and ensuring the final product meets stringent heavy metal specifications required for oncology APIs.
Q: What are the environmental benefits of this electrochemical method over sodium dithionite reduction?
A: This method replaces hazardous chemical reducing agents like sodium dithionite with electrons, significantly reducing toxic waste generation and simplifying wastewater treatment processes in compliance with green chemistry principles.
Q: Can this electroreduction process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process operates under mild conditions (25°C-80°C) and atmospheric pressure without explosive hydrogen gas, making it inherently safer and easier to scale using standard diaphragm electrolytic cells for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl 2-amino-4-methoxy-5-(3-morpholinylpropoxy)benzoate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in the production of complex pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative lab-scale methods like this electroreduction process can be successfully translated into robust industrial operations. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required for oncology APIs. We are committed to delivering high-quality intermediates that comply with international regulatory standards, providing our partners with the confidence they need to accelerate their drug development pipelines.
We invite global pharmaceutical companies and procurement leaders to collaborate with us to leverage this advanced technology for their gefitinib supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and quotations that reflect the economic and environmental benefits of this next-generation manufacturing process. Let us help you secure a sustainable and cost-effective supply of critical intermediates for the future.
