Revolutionizing Remdesivir Intermediate Production: A High-Yield Chiral Synthesis Strategy for Global Supply Chains
The global pharmaceutical landscape has been profoundly impacted by the demand for effective antiviral therapeutics, with Remdesivir standing out as a critical nucleotide analogue for treating RNA virus infections. At the heart of manufacturing this complex molecule lies the efficient production of its key phosphoramidate precursor, often referred to as Compound I. Patent CN114685558A, published in July 2022, introduces a groundbreaking preparation method that addresses longstanding bottlenecks in the synthesis of this vital intermediate. By leveraging a novel catalytic crystallization technique, this technology enables the conversion of Compound II into Compound I with exceptional chiral purity exceeding 99.8 percent and yields surpassing 90 percent. This represents a significant leap forward compared to traditional routes, offering a robust solution for reliable pharmaceutical intermediate suppliers aiming to secure the global supply chain against disruptions.
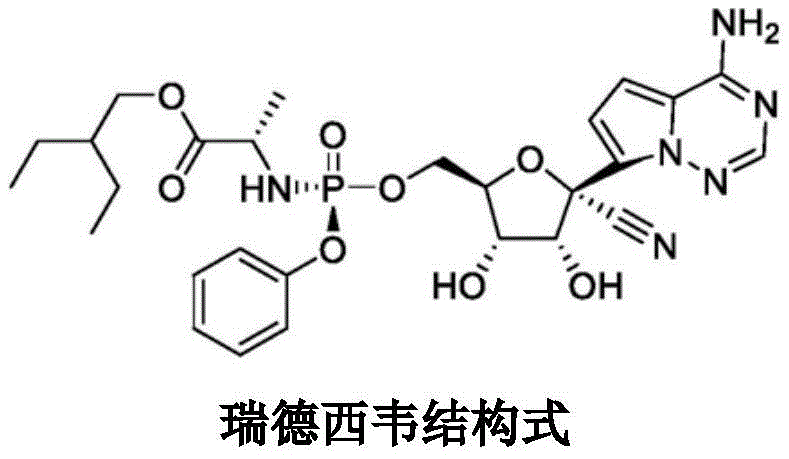
The structural complexity of Remdesivir, as illustrated in the chemical diagram, necessitates precise stereochemical control during the assembly of its phosphoramidate moiety. The intermediate Compound I serves as the pivotal building block that links the amino acid ester side chain to the nucleoside core. Ensuring the high fidelity of this linkage is not merely a chemical challenge but a commercial imperative, as impurities at this stage can propagate through the synthesis, complicating downstream purification and jeopardizing the safety profile of the final Active Pharmaceutical Ingredient (API). The innovation detailed in this patent provides a scalable pathway to meet these rigorous quality standards while simultaneously optimizing production economics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
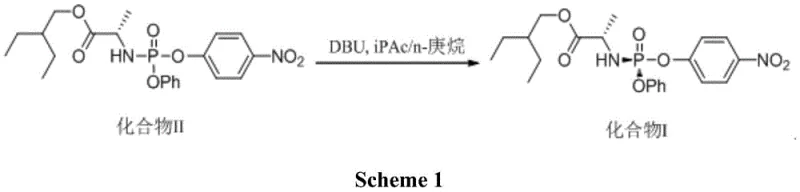
Prior to this innovation, the industrial synthesis of Compound I was plagued by inefficiencies that strained both cost structures and production timelines. One prominent method, disclosed in patent WO2016069826A1, relied on a cumbersome protocol involving the sequential addition of isopropyl acetate and n-heptane, followed by a protracted stirring period of 21 hours at 0°C in the presence of DBU. This extended reaction time not only tied up reactor capacity but also increased energy consumption and operational complexity. Furthermore, the requirement for precise temperature control over such a long duration introduced significant risks of batch variability, making it a less than ideal candidate for commercial scale-up of complex pharmaceutical intermediates.
Another documented approach, published in Angewandte Chemie, attempted to simplify the process through recrystallization using isopropyl ether. However, this method suffered from a catastrophic loss of material efficiency, with reported yields hovering around a mere 39 percent. In the context of high-value API manufacturing, losing more than 60 percent of the starting material is economically unsustainable and environmentally irresponsible. Such low yields necessitate the processing of vastly larger quantities of raw materials to achieve the same output, thereby inflating the cost of goods sold (COGS) and generating excessive chemical waste that requires costly disposal. These legacy methods highlight an urgent industry need for a more atom-economical and operationally streamlined solution.
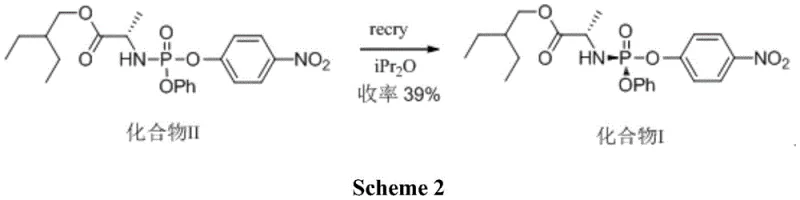
The Novel Approach
The methodology presented in CN114685558A fundamentally reimagines the transformation of Compound II into Compound I by introducing a catalytic system driven by mono-substituted or poly-substituted phenols. Instead of relying on stoichiometric reagents or prolonged thermal treatments, this process dissolves Compound II in a crystallization solvent and introduces a catalytic amount of an aromatic phenol (ArOH) alongside a base. This elegant modification triggers a highly selective crystallization event that enriches the desired chiral species directly from the solution. The result is a dramatic improvement in process metrics, achieving yields greater than 90 percent while maintaining mild reaction conditions that are far easier to control in a large-scale manufacturing environment.
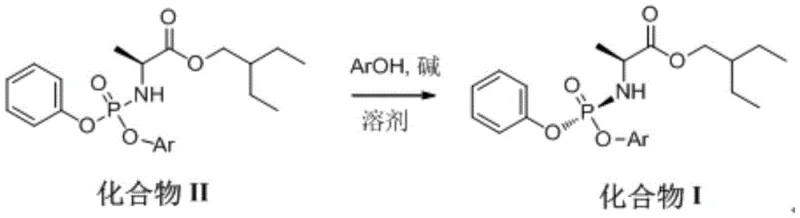
This new route eliminates the need for the harsh conditions and extended timelines associated with previous techniques. By utilizing catalysts such as 4-nitrophenol or pentafluorophenol in conjunction with bases like triethylamine, the reaction proceeds rapidly and cleanly. The simplicity of the operation—essentially a dissolve, treat, cool, and filter sequence—drastically reduces the operational burden on manufacturing teams. This shift from complex, multi-step workups to a direct crystallization-driven purification exemplifies the kind of process intensification that modern fine chemical manufacturing demands to remain competitive and agile in a volatile market.
Mechanistic Insights into Catalytic Chiral Enrichment
The core of this technological breakthrough lies in the subtle interplay between the chiral phosphoramidate structure and the added phenolic catalyst. While the exact molecular dynamics are complex, the process likely operates through a mechanism of diastereomeric salt formation or selective solvation modulation. The introduction of the chiral or achiral phenol additive, in the presence of a base, alters the solubility profile of the target enantiomer relative to its impurities or the opposite enantiomer. As the solution is cooled to the 0-10°C range, the thermodynamic stability of the desired crystal lattice is favored, prompting the selective precipitation of Compound I. This phenomenon effectively acts as a dynamic resolution or a powerful enrichment step, scrubbing impurities from the solid phase and locking them into the mother liquor.
From an impurity control perspective, this mechanism is exceptionally robust. The high chiral purity of >99.8% indicates that the crystallization kinetics are highly discriminatory. Traditional methods often struggle to remove trace stereoisomers without resorting to preparative chromatography, which is prohibitively expensive at scale. In contrast, this catalytic crystallization leverages the inherent physical properties of the molecule to achieve purification. The choice of solvent, ranging from isopropyl ether to esters like ethyl acetate, further tunes the supersaturation levels, ensuring that nucleation occurs only for the desired species. This level of control is essential for meeting the stringent regulatory requirements for high-purity pharmaceutical intermediates, where even minor deviations in stereochemistry can lead to batch rejection.
How to Synthesize Remdesivir Intermediate Efficiently
Implementing this synthesis route requires careful attention to solvent selection and temperature profiling, but the overall workflow is markedly simpler than legacy processes. The patent outlines a versatile protocol that accommodates various substituted phenols, with 4-nitrophenol being a preferred embodiment due to its balance of acidity and solubility characteristics. Operators begin by dissolving the crude Compound II in a heated solvent, ensuring a homogeneous starting phase. The subsequent addition of the catalytic system initiates the transformation, and the controlled cooling ramp is critical for maximizing crystal growth and yield. For a detailed breakdown of the specific reagent quantities and timing, please refer to the standardized synthesis guide below.
- Dissolve Compound II in a suitable crystallization solvent such as isopropyl ether or isopropyl acetate under heating.
- Add a catalytic amount of substituted phenol (e.g., 4-nitrophenol) and a base (e.g., triethylamine) to the solution.
- Cool the mixture to 0-10°C, add seed crystals of Compound I, stir to induce crystallization, then filter and dry to obtain the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented method translates into tangible strategic advantages that go beyond simple chemistry. The primary benefit is the substantial enhancement in material throughput. By boosting yields from the sub-40% range typical of older recrystallization methods to over 90%, manufacturers can nearly double their output from the same quantity of raw materials. This efficiency gain directly mitigates the risk of raw material shortages and reduces the frequency of purchasing cycles, thereby stabilizing the supply chain against market volatility. Furthermore, the simplified operational profile reduces the dependency on specialized equipment capable of long-duration low-temperature stirring, freeing up reactor assets for other campaigns.
- Cost Reduction in Manufacturing: The economic implications of this process are profound. By eliminating the need for stoichiometric amounts of expensive resolving agents and reducing solvent consumption through higher yields, the overall cost of production is significantly lowered. The removal of the 21-hour stirring step also results in drastic energy savings, as refrigeration units do not need to run for extended periods. Additionally, the higher purity of the intermediate reduces the burden on downstream purification steps, potentially lowering the cost of the final API. These cumulative savings create a more competitive pricing structure for the final product without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions. The tolerance for various solvents, including common ethers and esters, means that procurement teams are not locked into single-source suppliers for exotic reagents. The shorter cycle time—from days to hours—allows for faster turnaround on orders, enabling manufacturers to respond more swiftly to sudden spikes in demand. This agility is crucial in the antiviral market, where demand can fluctuate rapidly based on epidemiological trends. The ability to reliably produce high-quality intermediates ensures continuity of supply for downstream API formulators.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method offers a greener alternative. The reduction in waste generation, driven by higher yields and fewer purification steps, aligns with increasingly strict global environmental regulations. The use of catalytic amounts of phenols rather than large excesses of reagents minimizes the chemical load in wastewater streams. Moreover, the simplicity of the process facilitates easier scale-up from pilot plants to multi-ton commercial production, reducing the technical risks associated with technology transfer. This makes the process highly attractive for contract development and manufacturing organizations (CDMOs) looking to expand their antiviral portfolio.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear picture of what partners can expect when adopting this route. Understanding these nuances is key to evaluating the feasibility of integrating this method into existing manufacturing lines.
Q: What is the chiral purity achieved by this new synthesis method?
A: The method described in patent CN114685558A achieves a chiral purity of greater than 99.8% for Compound I, which is critical for the efficacy of the final Remdesivir API.
Q: How does this method improve upon previous industrial processes?
A: Unlike prior art methods that suffered from low yields (around 39%) or required complex, long-duration stirring (21 hours), this novel approach utilizes a catalytic phenol system to achieve yields over 90% with simplified operations.
Q: What solvents are compatible with this crystallization process?
A: The process is robust and compatible with various green solvents including isopropyl ether, isopropyl acetate, and n-heptane, allowing for flexible supply chain sourcing and environmental compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Remdesivir Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields and purity profiles demonstrated in patent CN114685558A can be faithfully reproduced at an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of Remdesivir intermediate meets the highest global pharmacopeial standards.
We invite pharmaceutical companies and generic manufacturers to collaborate with us to leverage this advanced synthesis technology. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing processes can enhance your supply chain security and drive down your overall cost of goods.
