Advanced Synthesis of Ertapenem Intermediate: Enhancing Purity and Commercial Scalability for Global Supply Chains
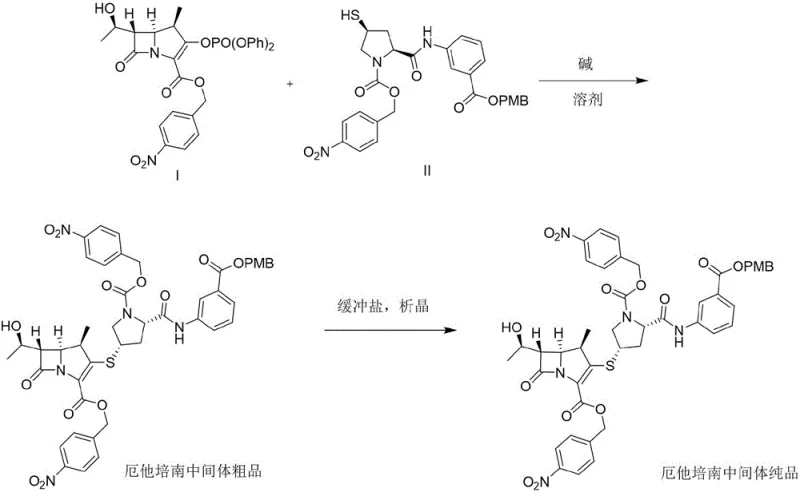
The pharmaceutical landscape for broad-spectrum carbapenem antibiotics is continuously evolving, driven by the urgent need for stable, high-purity intermediates that can withstand rigorous industrial scaling. Patent CN110698480A introduces a transformative synthesis and purification method for an ertapenem intermediate, addressing critical bottlenecks that have long plagued the manufacturing of this vital antibiotic. This technical breakthrough focuses on the condensation of a protected penem mother nucleus MAP with a protected ertapenem side chain, followed by a novel crystallization protocol that ensures the final white solid achieves a purity exceeding 99%. For R&D Directors and Supply Chain Heads, this represents a significant leap forward, as it directly mitigates the risks associated with intermediate instability and low yields that characterize older synthetic routes. By optimizing the reaction conditions and purification steps, this method not only enhances the chemical integrity of the molecule but also streamlines the downstream processing required for the final active pharmaceutical ingredient.
The commercial implications of adopting this synthesis route are profound for any organization seeking a reliable ertapenem intermediate supplier. Traditional methods often involve multiple, cumbersome purification stages that erode profit margins and extend lead times. In contrast, the approach detailed in CN110698480A simplifies the purification operation of the final product by front-loading the quality control into the intermediate stage. This strategic shift allows for cost reduction in pharmaceutical intermediates manufacturing by minimizing solvent usage, reducing waste generation, and shortening the overall production cycle. For procurement managers, this translates into a more predictable supply chain with reduced exposure to the volatility often seen in complex antibiotic synthesis. The ability to consistently produce high-purity intermediates means fewer batch failures and a more robust inventory management system, which is essential for maintaining the continuity of supply for life-saving medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ertapenem intermediates has been fraught with challenges, primarily stemming from the inherent instability of the beta-lactam ring and the complexity of the side-chain coupling. Many existing routes, including those referenced in earlier patents such as US2004/0235817A1, suffer from significant drawbacks that hinder efficient commercial scale-up of complex pharmaceutical intermediates. These conventional methods often result in intermediates that are chemically unstable, leading to decomposition during storage or subsequent reaction steps. Furthermore, the yields associated with these older processes are frequently suboptimal, necessitating larger starting material inputs to achieve the same output, which drives up raw material costs. The purification processes in these traditional routes are also notoriously difficult, often requiring multiple chromatographic separations or recrystallizations that are time-consuming and expensive. This inefficiency creates a bottleneck in the supply chain, making it difficult for manufacturers to respond quickly to market demand fluctuations.
The Novel Approach
The methodology presented in CN110698480A offers a decisive break from these inefficiencies by introducing a controlled condensation and crystallization strategy that stabilizes the intermediate early in the process. By reacting the penem mother nucleus MAP with the ertapenem side chain under specific basic conditions and in selected organic solvents, the process generates an intermediate product that is far more robust than those produced by conventional means. The key innovation lies in the subsequent crystallization step, which utilizes a buffer salt system to precipitate the intermediate as a high-purity white solid. This approach effectively locks in the chemical quality before the molecule undergoes further transformation, thereby reducing the burden on final product purification. For technical teams, this means a more predictable reaction profile and a significant reduction in the formation of difficult-to-remove impurities. The result is a synthesis route that is not only chemically superior but also economically advantageous, offering a clear path toward reducing lead time for high-purity pharmaceutical intermediates.
Mechanistic Insights into Base-Catalyzed Condensation and Crystallization
The core of this synthetic advancement lies in the precise control of the condensation reaction between the protected penem mother nucleus and the side chain. The reaction is facilitated by a base, such as tetramethylguanidine or triethylamine, which deprotonates the thiol group on the side chain, enabling a nucleophilic attack on the activated leaving group of the penem nucleus. This step is critical, as the stereochemistry at the C2 and C3 positions of the penem ring must be preserved to maintain biological activity. The choice of solvent, ranging from tetrahydrofuran to N-dimethylacetamide, plays a vital role in solubilizing the reactants while maintaining a reaction environment that minimizes side reactions. The temperature control, maintained between -30°C and -10°C during the condensation phase, is essential to prevent the thermal degradation of the sensitive beta-lactam structure. This low-temperature regime ensures that the kinetic energy of the molecules is sufficient for the desired coupling but low enough to suppress competing decomposition pathways.
Following the condensation, the purification mechanism shifts to a sophisticated crystallization process driven by a buffer salt system. The introduction of buffers such as sodium bicarbonate/acetic acid or sodium hydrogen phosphate/phosphoric acid creates a specific pH environment that optimizes the solubility profile of the intermediate. By dripping the reaction mixture into this buffer system at controlled temperatures between 0°C and 20°C, the intermediate precipitates out of the solution in a highly ordered crystalline form. This crystallization is not merely a physical separation but a chemical purification step that excludes impurities and by-products from the crystal lattice. The result is an intermediate with a purity of over 99%, as evidenced by the experimental data in the patent. This high level of purity at the intermediate stage is crucial because it simplifies the final deprotection and salt formation steps, ensuring that the final ertapenem sodium salt meets stringent pharmacopeial standards with minimal additional processing.
How to Synthesize Ertapenem Intermediate Efficiently
Implementing this synthesis route requires a disciplined approach to reaction conditions and purification parameters to fully realize the benefits outlined in the patent data. The process begins with the careful selection of solvents and bases that are compatible with the sensitive functional groups present in both the penem nucleus and the side chain. Operators must ensure that the condensation reaction is conducted under strict temperature control to avoid thermal stress on the beta-lactam ring. Once the coupling is complete, the transition to the crystallization phase must be seamless, with the buffer system prepared in advance to receive the reaction mixture. The detailed standardized synthesis steps see the guide below for specific operational parameters and safety considerations.
- Dissolve penem mother nucleus MAP (Formula I) and ertapenem side chain (Formula II) in an organic solvent such as tetrahydrofuran or acetonitrile.
- Perform a condensation reaction under the action of a base like tetramethylguanidine or triethylamine at temperatures between -30°C and -10°C.
- Crystallize the intermediate product in a buffer salt system (e.g., sodium bicarbonate/acetic acid) at 0-20°C to obtain purity exceeding 99%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis method offers tangible strategic advantages that go beyond simple chemical yield. The primary benefit is the significant simplification of the manufacturing workflow, which directly correlates to reduced operational costs and enhanced supply chain reliability. By achieving high purity at the intermediate stage, manufacturers can eliminate several downstream purification steps that are typically resource-intensive. This reduction in processing complexity means that production facilities can operate with higher throughput and lower energy consumption. Furthermore, the stability of the intermediate allows for more flexible inventory management, reducing the risk of stockouts due to material degradation. These factors combine to create a more resilient supply chain capable of meeting the rigorous demands of the global pharmaceutical market.
- Cost Reduction in Manufacturing: The elimination of complex and multiple purification stages significantly lowers the overall cost of goods sold. By avoiding the need for expensive chromatographic separations or repeated recrystallizations, the process reduces solvent consumption and waste disposal costs. The high yield reported in the patent examples, consistently above 90%, ensures that raw material utilization is maximized, further driving down the cost per kilogram of the final product. Additionally, the use of common organic solvents and bases reduces the reliance on exotic or prohibitively expensive reagents. This economic efficiency makes the final ertapenem product more competitive in the market, allowing for better margin management without compromising on quality standards.
- Enhanced Supply Chain Reliability: The stability of the intermediate produced by this method is a critical factor in ensuring a continuous supply of the final antibiotic. Unlike unstable intermediates that require immediate processing or specialized storage conditions, this high-purity solid can be stored and transported with greater ease. This flexibility reduces the logistical pressure on the supply chain and minimizes the risk of production delays caused by material spoilage. For global buyers, this means a more dependable source of supply that can withstand disruptions in the manufacturing schedule. The robustness of the process also allows for easier technology transfer between manufacturing sites, ensuring consistent quality regardless of the production location.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing reaction conditions and equipment that are standard in fine chemical manufacturing. The simplified workflow reduces the overall environmental footprint by minimizing waste generation and solvent usage. This alignment with green chemistry principles facilitates easier regulatory compliance and reduces the burden of environmental reporting. The ability to scale from laboratory to commercial production without significant process re-engineering ensures that the technology can be rapidly deployed to meet market demand. This scalability is essential for maintaining a steady supply of essential medicines, particularly in times of increased global health needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this ertapenem intermediate. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation. They are intended to provide clarity on the process capabilities and the quality standards achievable through this method. Understanding these details is crucial for making informed decisions regarding the sourcing and manufacturing of carbapenem antibiotics.
Q: How does this synthesis method improve intermediate stability compared to prior art?
A: Unlike conventional routes which suffer from unstable intermediates and low yields, this method utilizes a specific buffer salt crystallization system that stabilizes the molecular structure, ensuring consistent purity above 99%.
Q: What solvents are compatible with this condensation reaction?
A: The process is robust across several polar aprotic solvents, including tetrahydrofuran, acetonitrile, N-dimethylformamide, and N-ethylpyrrolidone, offering flexibility for industrial scale-up.
Q: Does this route eliminate the need for complex final purification?
A: Yes, by achieving high purity at the intermediate stage through controlled crystallization, the subsequent purification operations for the final ertapenem product are significantly simplified, reducing overall processing time.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ertapenem Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving antibiotics like ertapenem. Our team of experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN110698480A are fully realized in a commercial setting. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of ertapenem intermediate meets the highest international standards. Our capability to handle complex synthetic routes allows us to offer a reliable ertapenem intermediate supplier partnership that is built on technical excellence and operational reliability.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. We encourage you to contact us to request specific COA data and route feasibility assessments that demonstrate how our advanced synthesis capabilities can enhance your product portfolio. By partnering with us, you gain access to a wealth of technical knowledge and a supply chain infrastructure designed for efficiency and quality.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
