Scalable Synthesis of Ertapenem Intermediates: A Technical Breakthrough for API Manufacturing
Scalable Synthesis of Ertapenem Intermediates: A Technical Breakthrough for API Manufacturing
The global demand for broad-spectrum carbapenem antibiotics continues to rise, driven by the increasing prevalence of resistant bacterial infections. At the heart of this therapeutic class lies Ertapenem, a critical agent known for its stability and efficacy. However, the commercial viability of Ertapenem has historically been constrained by complex synthetic routes that rely on harsh conditions and expensive reagents. The patent CN101376643B introduces a transformative methodology for preparing the key mercapto-pyrrolidine intermediate, designated as Formula VII. This technical insight report analyzes the profound implications of this invention for pharmaceutical manufacturers, highlighting how it shifts the paradigm from laboratory-scale curiosity to industrial reality. By leveraging mild reaction conditions and eliminating cumbersome purification steps, this route offers a compelling value proposition for supply chain optimization and cost reduction in antibiotic manufacturing.
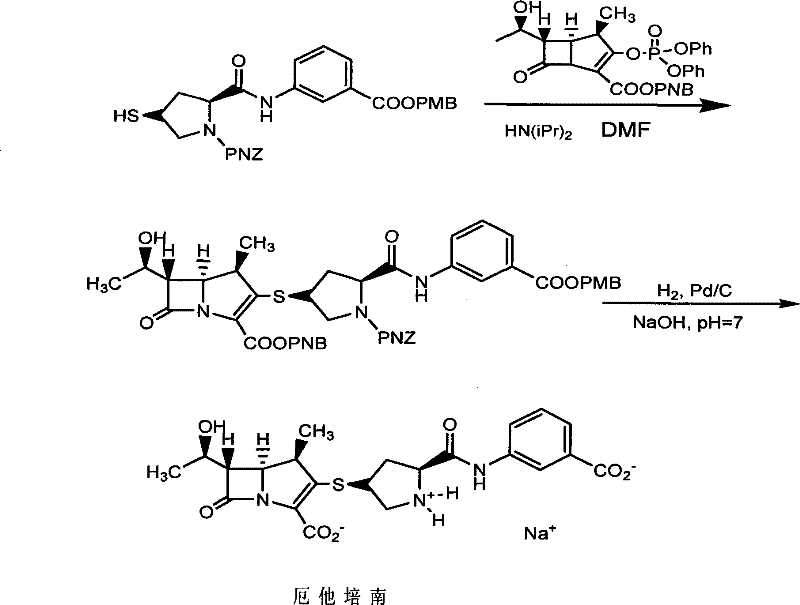
This patent specifically addresses the synthesis of the side chain precursor required for the final assembly of Ertapenem. The innovation is not merely incremental; it represents a fundamental re-engineering of the chemical pathway to prioritize scalability and safety. For R&D directors and process chemists, the shift away from cryogenic temperatures and phosphorus-based activating agents marks a significant reduction in process hazard and operational expenditure. The following analysis dissects the technical merits of this approach, contrasting it with legacy methods to demonstrate why it stands as a superior choice for reliable pharmaceutical intermediate supplier partnerships.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those described in US5478820 and US5872250, have long plagued the industry with inefficiencies that hinder mass production. These traditional routes typically necessitate a 'one-pot' synthesis for the side chain that must be maintained at cryogenic temperatures, often as low as -20°C. Maintaining such low temperatures on a multi-ton scale requires specialized refrigeration equipment and substantial energy input, directly inflating the cost of goods sold. Furthermore, these legacy processes frequently employ expensive reagents like diisopropylethylamine and diphenylphosphoryl chloride, which not only drive up raw material costs but also generate difficult-to-handle waste streams. Perhaps most critically, the purification of intermediates in these older methods often relies on column chromatography, a technique that is notoriously difficult to scale and impractical for GMP manufacturing of active pharmaceutical ingredients.
The Novel Approach
In stark contrast, the methodology disclosed in CN101376643B utilizes a stepwise approach that operates under remarkably mild conditions. The reduction of the nitro group, a pivotal transformation, is achieved using stannous chloride dihydrate in alcoholic solvents at temperatures between 40°C and 75°C. This eliminates the need for energy-intensive cooling systems. Additionally, the process avoids the use of hazardous phosphorus chlorides, substituting them with more benign activating agents like chloroformates for the proline coupling step. The purification strategy is equally revolutionary; instead of chromatography, the protocol relies on standard unit operations such as extraction, washing, and recrystallization. This shift ensures that the process is robust, reproducible, and fully compatible with existing industrial infrastructure, thereby facilitating the commercial scale-up of complex pharmaceutical intermediates.
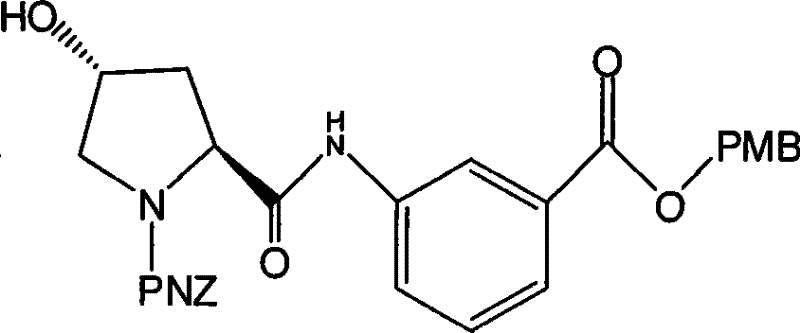
Mechanistic Insights into Stannous Chloride Reduction and Amide Coupling
The core of this synthetic innovation lies in the efficient conversion of the nitrobenzoic acid derivative to the corresponding aniline, followed by precise stereochemical control during the proline coupling. The reduction step employs stannous chloride dihydrate (SnCl2·2H2O) as the reducing agent. Mechanistically, the tin(II) species transfers electrons to the nitro group in an alcoholic medium, sequentially reducing it through nitroso and hydroxylamine intermediates to the final amine. The patent specifies that this reaction proceeds efficiently at 50-55°C, a temperature range that ensures rapid kinetics without compromising the integrity of the ester protecting group. Following the reduction, the pH is carefully adjusted to alkaline conditions (pH 10-11) using inorganic bases like sodium hydroxide. This step is crucial for liberating the free amine from its tin salt complex, allowing for efficient extraction into organic solvents and ensuring high purity of the resulting 3-aminobenzoic acid ester.
Subsequent steps focus on constructing the chiral pyrrolidine scaffold with high fidelity. The coupling of the aniline intermediate with PNZ L-hydroxyproline is mediated by an activated ester, typically generated in situ using chloroformates. This activation strategy minimizes racemization, preserving the critical (2S, 4R) stereochemistry required for biological activity. The introduction of the sulfur moiety is achieved via a classic nucleophilic substitution mechanism. First, the hydroxyl group of the proline ring is converted into a mesylate using methanesulfonyl chloride. This creates an excellent leaving group, which is then displaced by potassium thioacetate in an SN2 reaction. The use of thioacetate rather than direct thiolation protects the sensitive sulfur atom from oxidation during the synthesis. Finally, mild acidic or basic hydrolysis cleaves the acetyl group to reveal the free thiol, yielding the target intermediate Formula VII ready for final coupling.
How to Synthesize Ertapenem Intermediate Efficiently
The synthesis of this high-value carbapenem side chain requires strict adherence to the optimized parameters outlined in the patent to ensure maximum yield and purity. The process begins with the esterification of 3-nitrobenzoic acid, followed by the critical tin-mediated reduction and subsequent amide bond formation. Each step has been fine-tuned to balance reaction rate with impurity control, utilizing common solvents like dichloromethane, ethanol, and ethyl acetate. The detailed standardized synthesis steps see the guide below for specific molar ratios and temperature profiles that guarantee success.
- Perform esterification of 3-nitrobenzoic acid with 4-methoxybenzyl chloride in an aprotic solvent with organic base catalysis.
- Reduce the nitro group to an amino group using stannous chloride dihydrate in an alcoholic solvent at moderate temperatures (40-75°C).
- Condense the resulting amine with activated PNZ L-hydroxyproline ester, followed by mesylation and thioacetate substitution to yield the final protected intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route translates into tangible strategic advantages beyond mere technical elegance. The primary benefit is the drastic simplification of the manufacturing process, which directly correlates to reduced operational risks and lower capital expenditure. By removing the dependency on cryogenic reactors and column chromatography columns, facilities can utilize standard glass-lined or stainless steel reactors, significantly lowering the barrier to entry for production. This flexibility enhances supply chain reliability, as the process is less susceptible to equipment failure or utility fluctuations. Moreover, the use of commercially available, low-cost reagents like stannous chloride and methanesulfonyl chloride replaces exotic and expensive phosphorus compounds, leading to substantial cost savings in raw material procurement.
- Cost Reduction in Manufacturing: The elimination of cryogenic cooling systems represents a major reduction in energy consumption. Traditional methods requiring -20°C operation incur high electricity costs for refrigeration, whereas this new route operates at near-ambient or moderately elevated temperatures. Furthermore, the replacement of column chromatography with crystallization and extraction reduces solvent consumption and waste disposal costs. The avoidance of expensive activating agents like diphenylphosphoryl chloride further drives down the variable cost per kilogram, making the final API more price-competitive in the global market.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as tin salts, alcohols, and common organic bases ensures a stable supply of raw materials. Unlike specialized reagents that may have long lead times or single-source suppliers, the inputs for this process are widely available from multiple vendors. This diversification mitigates the risk of supply disruptions. Additionally, the robustness of the reaction conditions means that batch-to-batch variability is minimized, ensuring consistent quality and on-time delivery to downstream API manufacturers who depend on a steady flow of high-purity intermediates.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing unit operations that are standard in the fine chemical industry. The absence of chromatographic purification simplifies the workflow and reduces the volume of hazardous waste generated. The mild reaction conditions also enhance workplace safety by reducing the risk of thermal runaways associated with highly exothermic low-temperature reactions. This alignment with green chemistry principles facilitates easier regulatory approval and environmental compliance, streamlining the path from pilot plant to full commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for technical teams evaluating this technology for adoption.
Q: What are the temperature advantages of this new Ertapenem intermediate synthesis route?
A: Unlike conventional methods requiring cryogenic conditions at -20°C, this patented process operates at mild temperatures ranging from room temperature to 75°C, significantly reducing energy consumption and equipment complexity.
Q: How does this method improve purification compared to prior art?
A: The process eliminates the need for column chromatography, which is common in older synthetic routes. Instead, it utilizes crystallization and extraction techniques, making it highly suitable for large-scale industrial production.
Q: Is the stannous chloride reduction step safe for scale-up?
A: Yes, the reduction using stannous chloride dihydrate is conducted in standard alcoholic solvents under controlled pH adjustments, offering a robust and safe profile for commercial manufacturing without hazardous phosphorus reagents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ertapenem Intermediate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of implementing advanced synthetic methodologies like CN101376643B to deliver superior pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Ertapenem intermediate meets the exacting standards required for antibiotic formulation. Our commitment to process excellence allows us to offer a product that is not only chemically superior but also economically advantageous for our partners.
We invite global pharmaceutical companies to engage with us for a Customized Cost-Saving Analysis tailored to your specific production needs. By leveraging this optimized synthetic route, we can help you reduce your overall cost of goods while securing a stable supply of critical materials. Please contact our technical procurement team to request specific COA data and route feasibility assessments. Let us collaborate to bring life-saving antibiotics to the market more efficiently and affordably.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
