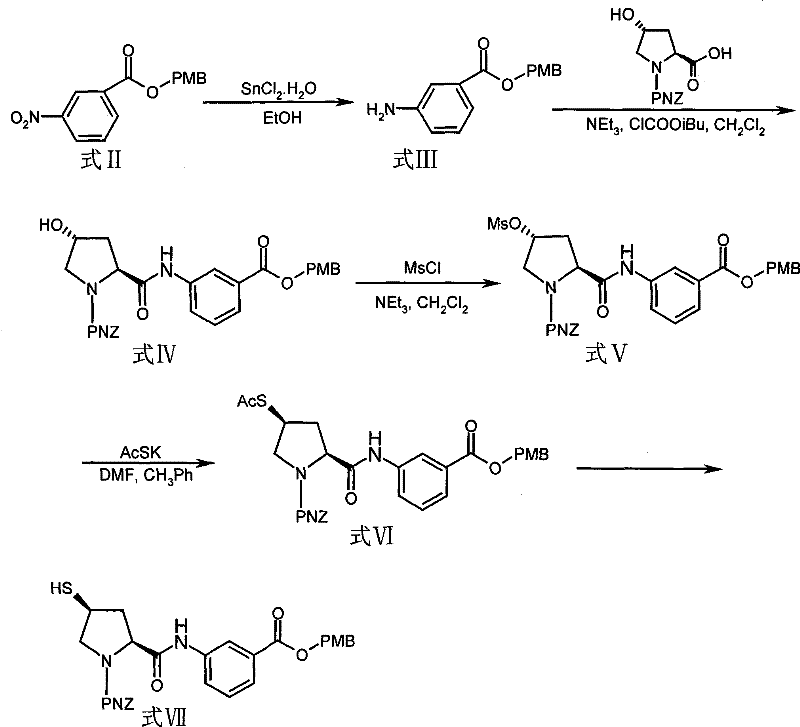
Advanced Synthesis of Ertapenem Intermediates for Commercial Scale-Up and Cost Efficiency
The pharmaceutical industry continuously seeks robust synthetic pathways for critical antibiotics, particularly carbapenems like Ertapenem, which are essential for treating severe bacterial infections. A pivotal advancement in this domain is documented in patent CN101376633B, which discloses a novel intermediate and its preparation method that addresses significant bottlenecks in existing manufacturing protocols. This technical breakthrough focuses on the synthesis of 3-nitrobenzoic acid 4-methoxybenzyl ester, a crucial building block for the Ertapenem side chain, offering a pathway that is both economically viable and technically superior. By shifting away from cryogenic dependencies and expensive reagents, this innovation provides a reliable foundation for high-purity pharmaceutical intermediate supplier networks aiming to optimize their production lines. The implications of this technology extend beyond mere chemical synthesis, influencing supply chain stability and cost structures for global healthcare manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Ertapenem intermediates has been plagued by stringent operational requirements that hinder efficiency and escalate costs. Prior art, such as methods described in US5872250 and US6180783, typically necessitates maintaining reaction temperatures as low as -20°C, demanding specialized cryogenic equipment and substantial energy consumption. Furthermore, these conventional routes often rely on costly reagents like diethylpropylethylamine and diphenylphosphoryl chloride, which inflate the raw material expenditure significantly. The need for multi-step column chromatography separation in older processes not only slows down throughput but also introduces potential points of failure regarding yield loss and solvent waste. These factors collectively create a fragile supply chain environment where lead times are extended, and the cost reduction in pharmaceutical manufacturing remains elusive for many producers struggling with legacy technologies.
The Novel Approach
In stark contrast, the methodology outlined in the referenced patent introduces a streamlined approach that operates under remarkably mild conditions, typically between 40°C and 60°C, eliminating the need for energy-intensive cooling systems. This novel route utilizes readily available organic solvents such as dichloromethane, toluene, or ethyl acetate, paired with common organic bases like triethylamine, to drive the esterification reaction efficiently. The process achieves yields exceeding 90% with purity levels greater than 99%, often without the need for complex purification steps like column chromatography, relying instead on simple extraction and recrystallization. This shift represents a paradigm change in commercial scale-up of complex pharmaceutical intermediates, allowing facilities to maximize reactor utilization and minimize downtime. By simplifying the operational parameters, this approach ensures a more resilient and continuous supply of high-quality intermediates for downstream API synthesis.
Mechanistic Insights into Organic Base Catalyzed Esterification
The core of this technological advancement lies in the efficient esterification mechanism facilitated by organic base catalysis in aprotic solvents. The reaction involves the nucleophilic attack of the carboxylate anion, generated in situ from 3-nitrobenzoic acid and the organic base, upon the benzylic carbon of 4-methoxybenzyl chloride. This SN2-type substitution is carefully controlled by maintaining a molar ratio of the acid to the chloride between 1:1.5 and 1:3, ensuring complete conversion while minimizing side reactions. The choice of solvent plays a critical role in stabilizing the transition state and solubilizing the reactants, with dichloromethane and toluene proving particularly effective in enhancing reaction kinetics. Understanding this mechanistic nuance is vital for R&D directors focused on purity and impurity profiles, as precise control over stoichiometry and temperature directly correlates with the suppression of by-products.
Impurity control is further enhanced by the mild thermal profile of the reaction, which prevents the decomposition of sensitive functional groups often observed in harsher synthetic environments. The resulting intermediate, 3-nitrobenzoic acid 4-methoxybenzyl ester, exhibits a clean structural profile that facilitates subsequent transformation into the Ertapenem side chain without carrying over detrimental contaminants. High purity specifications, often reaching 100% in optimized examples, are achieved through straightforward workup procedures involving aqueous extraction and drying, followed by recrystallization from ethanol. This level of chemical integrity is paramount for ensuring the safety and efficacy of the final antibiotic product, aligning with the rigorous quality standards expected by regulatory bodies worldwide. The structural clarity of the intermediate is depicted below, highlighting the key functional groups involved in the downstream coupling reactions.
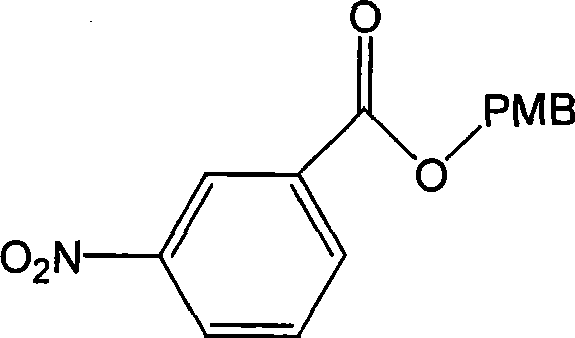
How to Synthesize 3-Nitrobenzoic Acid 4-Methoxybenzyl Ester Efficiently
Implementing this synthesis requires careful attention to reagent addition rates and temperature monitoring to replicate the high yields reported in the patent literature. The process begins with the dissolution of the nitrobenzoic acid starting material in a selected aprotic solvent, followed by the neutralization with an organic base to form the reactive salt species. Subsequent addition of the benzyl chloride derivative must be controlled to manage exothermicity, although the overall reaction remains manageable at moderate temperatures between 40°C and 60°C. Detailed standardized synthesis steps see the guide below, which outlines the specific molar ratios and workup procedures necessary to achieve the reported purity levels consistently. Adhering to these parameters ensures that the production environment remains safe and efficient, maximizing the output of this valuable pharmaceutical building block.
- Dissolve 3-nitrobenzoic acid in an aprotic organic solvent such as dichloromethane or toluene under stirring.
- Add an organic base reagent like triethylamine to the solution at room temperature to activate the reaction environment.
- Dropwise add 4-methoxybenzyl chloride while controlling the temperature between 40°C and 60°C for optimal conversion.
- Upon completion, extract with dichloromethane and water, dry the organic layer, and concentrate to obtain the solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend well beyond the laboratory bench, impacting the overall economics of antibiotic production. The elimination of cryogenic cooling requirements drastically reduces utility costs and capital expenditure on specialized refrigeration equipment, leading to substantial cost savings in facility operations. Additionally, the use of commodity chemicals and solvents mitigates the risk of supply disruptions associated with specialty reagents, enhancing the reliability of the raw material supply chain. The simplified purification process reduces solvent consumption and waste generation, aligning with increasingly strict environmental compliance standards while lowering disposal costs. These factors combine to create a more predictable and cost-effective manufacturing model that supports long-term strategic planning for pharmaceutical companies.
- Cost Reduction in Manufacturing: The transition to ambient or moderately heated reaction conditions removes the significant energy burden associated with maintaining sub-zero temperatures, directly lowering operational expenditures. By replacing expensive phosphorylating agents with cost-effective alkyl halides and organic bases, the raw material cost profile is significantly optimized without compromising reaction efficiency. The high yield performance minimizes the loss of valuable starting materials, ensuring that every kilogram of input translates effectively into saleable product output. Furthermore, the avoidance of column chromatography reduces solvent usage and labor hours, contributing to a leaner and more economical production process overall.
- Enhanced Supply Chain Reliability: Utilizing widely available commercial reagents reduces dependency on single-source suppliers for niche chemicals, thereby diversifying the supply base and reducing procurement risk. The robustness of the reaction conditions means that production schedules are less susceptible to delays caused by equipment failures or environmental fluctuations, ensuring consistent delivery timelines. This stability is crucial for maintaining the continuity of API manufacturing, preventing stockouts that could impact patient access to critical medications. The simplified logistics of handling non-hazardous, stable reagents also streamline warehouse management and transportation requirements.
- Scalability and Environmental Compliance: The mild nature of the chemistry allows for seamless translation from pilot scale to multi-ton commercial production without the need for extensive process re-engineering. Reduced solvent waste and the absence of heavy metal catalysts simplify effluent treatment processes, making it easier to meet environmental discharge regulations. This environmental compatibility enhances the corporate sustainability profile of the manufacturer, appealing to partners who prioritize green chemistry initiatives. The ability to scale efficiently ensures that market demand can be met rapidly, supporting the global distribution of life-saving antibiotics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this intermediate synthesis technology in industrial settings. These insights are derived directly from the patent specifications and practical considerations for scaling organic synthesis processes. Understanding these details helps stakeholders evaluate the feasibility and advantages of adopting this newer methodology over established but less efficient routes. The answers provided reflect the consensus on best practices for maximizing yield and purity while maintaining operational safety.
Q: How does this new method improve upon traditional Ertapenem intermediate synthesis?
A: Unlike prior art requiring cryogenic conditions at -20°C and expensive reagents, this method operates at mild temperatures (40-60°C) using commercially available materials, significantly simplifying industrial operations.
Q: What purity levels can be achieved with this synthetic route?
A: The patented process consistently achieves purity levels exceeding 99% without the need for complex column chromatography, ensuring high-quality material suitable for API production.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the elimination of harsh low-temperature requirements and the use of standard solvents make this route highly scalable for multi-ton production facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Nitrobenzoic Acid 4-Methoxybenzyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of advanced antibiotics like Ertapenem. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 3-nitrobenzoic acid 4-methoxybenzyl ester meets the highest industry standards. We are committed to supporting your R&D and manufacturing goals by providing materials that facilitate efficient downstream processing and final API synthesis.
We invite you to engage with our technical procurement team to discuss how our capabilities can align with your specific project requirements. Request a Customized Cost-Saving Analysis to understand the economic benefits of switching to this optimized synthetic route for your production line. Our experts are ready to provide specific COA data and route feasibility assessments to demonstrate our commitment to quality and transparency. Partner with us to secure a stable, cost-effective supply chain for your pharmaceutical intermediates today.
