Advanced Synthetic Route for Levonorgestrel: Enhancing Safety and Scalability for Global Pharma Supply Chains
Advanced Synthetic Route for Levonorgestrel: Enhancing Safety and Scalability for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust manufacturing pathways that balance high purity with operational safety, particularly for critical hormonal active pharmaceutical ingredients (APIs). Patent CN111606962A introduces a transformative synthetic methodology for Levonorgestrel, a widely used emergency contraceptive and progestin, addressing significant limitations found in legacy production techniques. This innovation shifts the paradigm from hazardous lithium-based chemistries to a safer, three-step sequence involving protection, ethynylation, and hydrolysis, starting from the readily available DL-Ethyl Diketone. By leveraging mild reaction conditions and avoiding cryogenic lithium ammonia reagents, this process offers a compelling value proposition for reliable API intermediate supplier networks aiming to optimize their production portfolios. The technical depth of this patent provides a roadmap for achieving total mass yields exceeding 85% while maintaining product purity above 99.0%, ensuring that downstream formulation requirements are met with exceptional consistency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Levonorgestrel has relied heavily on routes involving Worth compounds or similar precursors that necessitate the use of lithium acetylide complexes. As illustrated in the prior art, these conventional pathways often require the generation of alkynyllithium species through the reaction of acetylene with lithium in liquid ammonia or similar hazardous solvents. This approach presents severe safety challenges, including the handling of pyrophoric reagents and the need for extreme cryogenic conditions to maintain reagent stability. Furthermore, the presence of ammonia and lithium residues introduces complex purification burdens, increasing the risk of heavy metal contamination and extending the overall production timeline. The reliance on such dangerous reagents not only elevates the cost of safety compliance and waste treatment but also limits the scalability of the process in facilities lacking specialized cryogenic infrastructure. Consequently, manufacturers face significant supply chain vulnerabilities when dependent on these archaic, high-risk synthetic strategies.
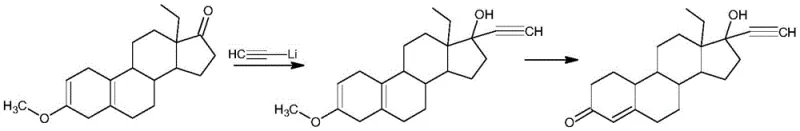
The Novel Approach
In stark contrast to the perilous legacy methods, the novel approach detailed in CN111606962A utilizes a streamlined three-step sequence that prioritizes safety and efficiency without compromising chemical integrity. The process initiates with a protection step where DL-Ethyl Diketone is converted into a stable enol ether intermediate using orthoformic acid triesters under mild acidic catalysis. This is followed by a highly efficient ethynylation step that employs alkali metal alcoholates, such as potassium tert-butoxide, instead of lithium amides, allowing the reaction to proceed safely at temperatures ranging from -20°C to 40°C. The final hydrolysis step gently removes the protecting group to reveal the target ketone functionality. This strategic redesign eliminates the need for liquid ammonia and metallic lithium, thereby removing the most significant safety bottlenecks associated with traditional manufacturing. The result is a process that is not only inherently safer but also operationally simpler, making it ideally suited for large-scale commercial production within standard pharmaceutical manufacturing facilities.
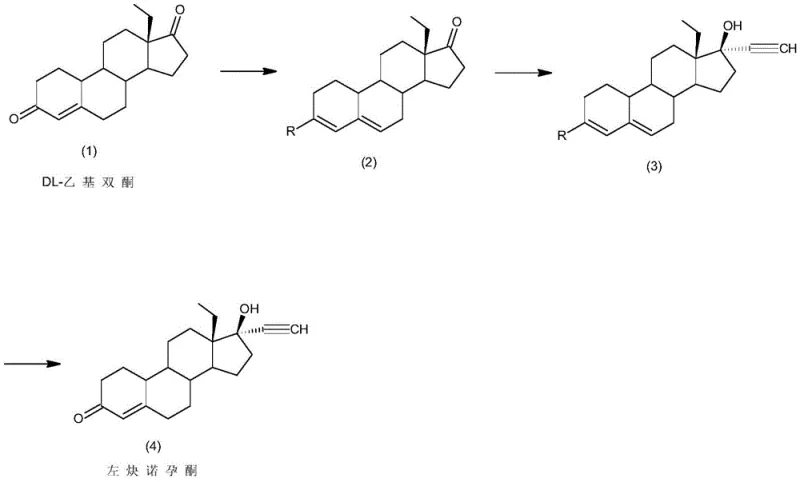
Mechanistic Insights into Alkali Metal Alcoholate Catalyzed Ethynylation
The core chemical innovation of this patent lies in the mechanistic execution of the ethynylation step, where the choice of base plays a pivotal role in determining reaction kinetics and impurity profiles. Unlike traditional methods that generate water as a byproduct when using hydroxides, the utilization of alkali metal alcoholates ensures an anhydrous environment crucial for the successful addition of the acetylene moiety to the steroid backbone. The alkoxide base deprotonates the terminal alkyne to form a reactive acetylide species in situ, which then nucleophilically attacks the C17 ketone of the protected intermediate. This mechanism avoids the equilibrium issues and side reactions often caused by moisture, leading to a cleaner reaction profile and higher conversion rates. The careful control of temperature between -20°C and 40°C further modulates the reactivity of the acetylide, preventing over-reaction or degradation of the sensitive steroid skeleton, which is essential for maintaining the stereochemical integrity required for biological activity.
Furthermore, the protection strategy employed in the first step serves as a critical control point for impurity management throughout the synthesis. By converting the C3 ketone into an enol ether using orthoformates, the molecule is shielded from unwanted nucleophilic attacks during the subsequent harsh ethynylation conditions. This temporary masking ensures that the acetylene addition occurs exclusively at the C17 position, thereby minimizing the formation of regio-isomers and other structural impurities that are difficult to separate in later stages. The subsequent acid-catalyzed hydrolysis is designed to be highly selective, cleaving the enol ether bond rapidly while leaving the newly formed tertiary alcohol and ethynyl groups intact. This precise orchestration of protective group chemistry and base-mediated addition results in a final crude product with exceptionally high purity, reducing the burden on downstream crystallization and purification units.
How to Synthesize Levonorgestrel Efficiently
The synthesis of Levonorgestrel via this patented route offers a practical and scalable solution for manufacturers seeking to modernize their production capabilities. The process is characterized by its use of common, commercially available reagents and solvents, which simplifies procurement and reduces dependency on specialized chemical supply chains. Operators can execute the protection, ethynylation, and hydrolysis steps using standard stainless steel reactors equipped with basic temperature control and gas sparging systems, eliminating the need for exotic cryogenic vessels. The following guide outlines the standardized operational parameters derived from the patent examples, providing a clear framework for process engineers to implement this technology. For detailed standard operating procedures and specific batch records, please refer to the technical guidelines below.
- Protection Reaction: React DL-Ethyl Diketone with orthoformic acid triester and an acidic catalyst in an organic solvent at 20-60°C, followed by neutralization with an organic base to isolate the protected intermediate.
- Ethynylation Reaction: Dissolve the protected intermediate in a polar aprotic solvent, add an alkali metal alcoholate base, and introduce acetylene gas at temperatures between -20°C and 40°C to form the ethynyl adduct.
- Hydrolysis and Purification: Treat the ethynyl intermediate with an aqueous acid solution in an organic solvent at -10 to 50°C to remove the protecting group, followed by neutralization, crystallization, and drying to obtain Levonorgestrel.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates into tangible strategic advantages that extend beyond mere chemical yield. By fundamentally altering the reagent profile to exclude hazardous lithium and ammonia, the process significantly lowers the barrier to entry for contract manufacturing organizations (CMOs) that may lack specialized hazard-handling certifications. This broadens the potential supplier base, enhancing supply chain resilience and reducing the risk of production stoppages due to regulatory audits or safety incidents. The simplified workflow, consisting of only three main steps with straightforward work-up procedures involving filtration and crystallization, allows for faster batch turnover times. This efficiency gain means that manufacturers can respond more agilely to market demand fluctuations, ensuring a steady flow of high-quality hormonal intermediates to downstream API producers without the delays typical of complex multi-step syntheses.
- Cost Reduction in Manufacturing: The elimination of expensive and dangerous lithium ammonia reagents directly impacts the bill of materials, replacing them with cost-effective alkali metal alcoholates and orthoformates. Additionally, the avoidance of cryogenic cooling requirements drastically reduces energy consumption and utility costs associated with maintaining sub-zero temperatures over extended periods. The high total mass yield reported in the patent, consistently exceeding 85%, ensures that raw material utilization is optimized, minimizing waste disposal costs and maximizing the output per unit of input. These factors combine to create a substantially lower cost of goods sold (COGS), providing a competitive pricing advantage in the global marketplace for hormonal contraceptives.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as acetone, methanol, and potassium tert-butoxide ensures that the supply chain is robust and less susceptible to the volatility often seen with specialized organometallic reagents. Since these solvents and bases are produced at massive scales for various industries, their availability is generally stable, reducing the risk of raw material shortages disrupting production schedules. Furthermore, the mild reaction conditions reduce wear and tear on reactor equipment, leading to lower maintenance downtime and longer asset life. This operational stability guarantees consistent delivery timelines, a critical metric for pharmaceutical buyers who must adhere to strict Just-In-Time inventory models to support their own formulation lines.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process offers a greener alternative by removing toxic ammonia emissions and lithium waste streams from the facility footprint. The simplified aqueous work-ups and the ability to recycle common organic solvents align well with modern sustainability goals and increasingly stringent environmental regulations. The process is inherently scalable, as demonstrated by the patent's emphasis on industrial suitability, allowing for seamless transition from pilot plant batches to multi-ton commercial production without significant re-engineering. This scalability ensures that suppliers can easily ramp up capacity to meet surging global demand for emergency contraception and hormone replacement therapies without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Levonorgestrel synthesis method. These answers are derived directly from the experimental data and beneficial effects described in patent CN111606962A, providing clarity on the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer and for procurement specialists assessing the long-term viability of this supply source. We encourage stakeholders to review these insights to fully appreciate the operational improvements offered by this innovative pathway.
Q: How does this synthetic method improve safety compared to traditional lithium ammonia routes?
A: This method eliminates the use of hazardous lithium ammonia reagents and cryogenic conditions typically required for acetylide generation. Instead, it utilizes alkali metal alcoholates under mild temperatures (-20°C to 50°C), significantly reducing operational risks and equipment requirements for industrial production.
Q: What are the expected yield and purity specifications for Levonorgestrel using this process?
A: According to the patent data, the total mass yield exceeds 85%, with specific examples demonstrating yields of 86% to 86.6%. The final product achieves an HPLC purity greater than 99.0%, meeting stringent pharmaceutical standards without complex purification steps.
Q: Why is the use of alkali metal alcoholates preferred over hydroxides in the ethynylation step?
A: Using alkali metal alcoholates (such as potassium tert-butoxide) prevents the generation of water during the reaction, which can hinder the ethynylation process. This anhydrous condition drastically improves reaction efficiency and conversion rates compared to methods using hydroxides.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Levonorgestrel Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain competitiveness in the global pharmaceutical landscape. Our team of expert process chemists has thoroughly analyzed the methodology presented in CN111606962A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering high-purity Levonorgestrel and its key intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical instrumentation. Our facility is equipped to handle the specific solvent systems and reaction conditions required for this route, ensuring that every batch delivered reflects the highest standards of quality and consistency demanded by international regulatory bodies.
We invite pharmaceutical partners and procurement leaders to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By leveraging our expertise in this optimized synthetic route, we can help you secure a stable supply of high-quality hormonal intermediates while achieving significant operational efficiencies. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and comprehensive quotations that demonstrate how our capabilities align with your strategic sourcing goals.
