Advanced Levonorgestrel Synthesis: A Safe, High-Yield Route for Industrial Pharmaceutical Manufacturing
Advanced Levonorgestrel Synthesis: A Safe, High-Yield Route for Industrial Pharmaceutical Manufacturing
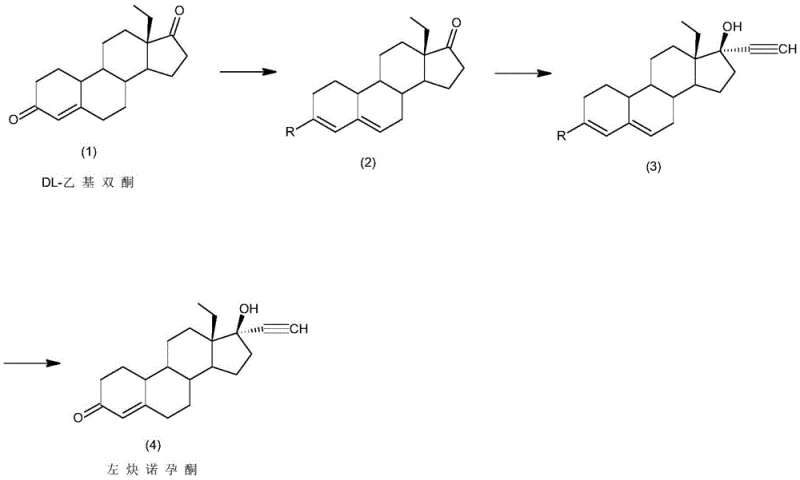
The pharmaceutical industry continuously seeks robust manufacturing pathways that balance safety, efficiency, and regulatory compliance, particularly for high-volume hormonal intermediates. Patent CN111606962B introduces a transformative synthesis method for levonorgestrel, a critical active pharmaceutical ingredient widely used in contraceptive formulations. This technology addresses the longstanding safety hazards associated with traditional lithiation processes by implementing a three-step sequence involving protection, ethynylation, and hydrolysis. By shifting away from dangerous lithium-ammonia reagents to milder alkali metal alcoholate systems, this innovation offers a compelling value proposition for manufacturers aiming to optimize their supply chains. The method demonstrates exceptional performance metrics, achieving total mass yields exceeding 85% and product purity levels surpassing 99.0%, thereby establishing a new benchmark for reliable hormonal intermediate supplier capabilities in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of levonorgestrel has relied heavily on routes such as the Wolff synthesis, which typically initiates from 18-methylestra-2,5(10)-diene-3-methoxy-17-ketone. A critical bottleneck in these legacy processes is the ethynylation step, which necessitates the use of lithium acetylide-ethylene diamine complexes or similar lithium-ammonia reagents. These reagents are inherently unstable and pose severe safety risks, including potential explosions and the release of toxic ammonia gas during handling and quenching. Furthermore, the reaction conditions often require rigorous cryogenic control and specialized pressure vessels to manage the volatility of ammonia, leading to inflated capital expenditure and operational complexity. The generation of significant chemical waste and the difficulty in managing exothermic profiles during scale-up further exacerbate the environmental footprint, making these conventional routes increasingly untenable for modern, sustainability-focused manufacturing facilities seeking cost reduction in pharmaceutical manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in CN111606962B utilizes DL-ethyl diketone as a robust starting material, initiating a streamlined three-step cascade that fundamentally alters the risk profile of the synthesis. The process begins with a protection step using orthoformic acid triester, followed by a strategic ethynylation that employs acetylene gas in the presence of alkali metal alcoholates rather than hazardous lithium reagents. This substitution is pivotal; it eliminates the generation of water which typically obstructs reaction efficiency in traditional bases, while simultaneously operating under significantly milder temperature ranges of -20 to 40°C. The final hydrolysis step efficiently unveils the target ketone functionality without requiring harsh conditions. This approach not only simplifies the operational workflow but also enhances the overall safety posture of the plant, making it an ideal candidate for the commercial scale-up of complex steroid intermediates.
Mechanistic Insights into Protection and Alkali-Mediated Ethynylation
The core chemical innovation lies in the precise manipulation of the steroid backbone to facilitate nucleophilic attack without compromising sensitive functional groups. In the initial protection phase, the ketone moiety at the C-3 position is masked using orthoformic acid triester under acidic catalysis, typically employing p-toluenesulfonic acid or pyridine hydrobromide at temperatures between 20°C and 60°C. This creates a stable enol ether intermediate that prevents unwanted side reactions during the subsequent aggressive ethynylation. The mechanism relies on the formation of a kinetic enolate or stabilized intermediate that directs the incoming acetylene nucleophile specifically to the C-17 position. By utilizing organic solvents such as tetrahydrofuran or acetone, the system maintains optimal solubility for the bulky steroid framework, ensuring homogeneous reaction kinetics that are critical for high conversion rates.
Furthermore, the choice of base in the ethynylation step is mechanistically superior to hydroxide-based systems. The patent specifies the use of alkali metal alcoholates, such as potassium tert-butoxide or sodium ethoxide, which act as strong, non-nucleophilic bases. Unlike hydroxides, these alcoholates do not introduce water into the reaction medium, a common impurity that can protonate the acetylide anion and terminate the chain growth prematurely. The absence of water allows the acetylene gas to dissolve and react efficiently with the C-17 ketone of the protected intermediate, forming the crucial 17-alpha-ethynyl-17-beta-hydroxy structure. Following this, the hydrolysis step utilizes dilute mineral acids to cleave the protecting group, restoring the conjugated enone system essential for biological activity. This meticulous control over reaction parameters ensures that impurity profiles remain minimal, directly supporting the production of high-purity levonorgestrel required for regulatory approval.
How to Synthesize Levonorgestrel Efficiently
Implementing this synthesis route requires strict adherence to the sequential addition of reagents and temperature controls outlined in the patent examples to maximize yield and safety. The process is designed to be operationally simple, utilizing standard glass-lined or stainless steel reactors commonly found in fine chemical facilities, without the need for specialized ammonia-handling infrastructure. Operators must ensure that the protection step reaches completion before cooling for filtration, and that the ethynylation is conducted under a controlled acetylene atmosphere to prevent oxidation. For detailed procedural specifics regarding stoichiometry, workup procedures, and purification techniques, please refer to the standardized synthesis guide below.
- Perform a protection reaction on DL-ethyl diketone using orthoformic acid triester and an acidic catalyst at 20-60°C to obtain the protected intermediate.
- Conduct an alkynylation reaction by dissolving the intermediate in organic solvent, adding organic base, and introducing acetylene at -20 to 40°C.
- Execute a hydrolysis reaction on the alkynylated intermediate using an acid solution at -10 to 50°C, followed by neutralization and refining to obtain pure levonorgestrel.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthesis pathway offers substantial strategic benefits beyond mere technical feasibility. The elimination of high-risk reagents translates directly into reduced insurance premiums and lower costs associated with safety compliance and hazardous waste disposal. By removing the dependency on lithium-ammonia complexes, facilities can operate with greater flexibility and reduced downtime related to safety audits or incident investigations. Moreover, the use of commodity chemicals like DL-ethyl diketone and common organic solvents ensures a resilient supply chain that is less susceptible to the volatility of specialized reagent markets. This stability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of downstream pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the simplification of the reaction infrastructure and the enhancement of overall yield efficiency. By avoiding the need for cryogenic ammonia systems and high-pressure reactors, capital investment requirements are significantly lowered, allowing for faster ROI on production assets. Additionally, the higher mass yield exceeding 85% means that less raw material is wasted per kilogram of final product, directly improving the cost of goods sold (COGS). The mild reaction conditions also result in lower energy consumption for heating and cooling, contributing to a leaner operational budget and improved margin potential for the final API.
- Enhanced Supply Chain Reliability: Sourcing reliability is markedly improved as the key starting material, DL-ethyl diketone, is a widely available bulk chemical compared to the specialized precursors required for older routes. The robustness of the reaction conditions, which tolerate a broader range of temperatures and solvent qualities, reduces the risk of batch failures due to minor fluctuations in raw material specifications. This resilience ensures that production volumes can be maintained consistently, reducing lead time for high-purity hormonal intermediates and allowing suppliers to honor long-term contracts with greater confidence even during market disruptions.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the process generates significantly less hazardous waste, aligning with increasingly stringent global environmental regulations. The absence of ammonia emissions and lithium waste streams simplifies effluent treatment protocols, reducing the burden on wastewater treatment facilities. The straightforward three-step sequence is inherently easier to scale from pilot plant to multi-ton production without the exponential increase in complexity often seen with cryogenic chemistries. This scalability ensures that suppliers can rapidly ramp up capacity to meet surging market demand for contraceptives without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their own manufacturing portfolios or for procurement teams assessing supplier qualifications.
Q: Why is the new levonorgestrel synthesis method safer than traditional routes?
A: The novel method described in patent CN111606962B eliminates the use of hazardous lithium-ammonia reagents required in traditional Wolff synthesis. By utilizing alkali metal alcoholates for ethynylation under mild temperatures (-20 to 40°C), it significantly reduces explosion risks and operational hazards associated with liquid ammonia handling.
Q: What is the expected purity and yield of this synthesis route?
A: According to the experimental data provided in the patent, this optimized route achieves a total mass yield exceeding 85% (specifically up to 86.6% in examples) and delivers a final product purity greater than 99.0% as determined by HPLC analysis, meeting stringent pharmaceutical standards.
Q: How does this process improve scalability for industrial production?
A: The process utilizes readily available starting materials like DL-ethyl diketone and avoids complex cryogenic conditions or high-pressure ammonia systems. The mild reaction conditions (20-60°C for protection, ambient to mild cooling for ethynylation) simplify equipment requirements, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Levonorgestrel Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting safer, more efficient synthetic routes to maintain competitiveness in the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless and compliant. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of levonorgestrel or related intermediate adheres to the highest international pharmacopoeia standards. Our facility is equipped to handle the specific solvent and temperature requirements of this novel protection-ethynylation-hydrolysis sequence with precision.
We invite forward-thinking pharmaceutical partners to collaborate with us to leverage this advanced technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your production of life-saving contraceptives is built on a foundation of safety, efficiency, and reliability.
