Revolutionizing Levonorgestrel Production: A Safer, High-Yield One-Pot Synthesis Strategy for Global Pharma Supply Chains
Revolutionizing Levonorgestrel Production: A Safer, High-Yield One-Pot Synthesis Strategy for Global Pharma Supply Chains
The pharmaceutical industry is constantly seeking robust, scalable, and safe methodologies for the production of critical hormonal active pharmaceutical ingredients (APIs). A recent technological breakthrough detailed in patent CN114181272B introduces a transformative synthesis method for levonorgestrel, a potent progestin widely used in contraceptive formulations. This innovation addresses long-standing safety and efficiency bottlenecks associated with traditional acetylene-based alkynylation. By replacing hazardous acetylene gas with stable trialkylsilylacetylene reagents and employing a streamlined one-pot protocol, this process offers a compelling value proposition for reliable hormonal API intermediate suppliers aiming to optimize their manufacturing portfolios. The following analysis dissects the technical merits, mechanistic advantages, and commercial implications of this novel route for R&D directors, procurement managers, and supply chain strategists.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of levonorgestrel has relied heavily on the direct ethynylation of ketone precursors using acetylene gas. This conventional approach presents severe operational challenges that hinder efficient cost reduction in contraceptive manufacturing. The primary issue lies in the physical properties of acetylene; its limited solubility in organic solvents like tetrahydrofuran necessitates the use of large solvent volumes and excessive amounts of potassium hydroxide to drive the reaction to completion. Furthermore, the introduction of acetylene gas often results in the formation of viscous pastes within the reactor, creating significant mass transfer limitations and making effective stirring nearly impossible. Beyond these process inefficiencies, the safety profile is concerning, as acetylene is a highly explosive and combustible gas, requiring rigorous safety protocols and specialized infrastructure that increase capital expenditure and operational risk.
The Novel Approach
In stark contrast, the methodology disclosed in CN114181272B utilizes a liquid trialkylsilylacetylene reagent, such as trimethylsilylacetylene, to perform the critical carbon-carbon bond formation. This substitution fundamentally alters the safety and efficiency landscape of the production line. The process operates as a seamless one-pot sequence where the alkynylation, desilylation, and hydrolysis steps occur without the need to isolate unstable intermediates. This integration not only accelerates the production cycle but also drastically minimizes solvent usage and waste generation. By eliminating the need for gaseous acetylene, the process removes the risk of explosion entirely, allowing for safer operation in standard chemical reactors. The result is a streamlined workflow that delivers high-purity levonorgestrel with superior yields compared to legacy methods, making it an ideal candidate for commercial scale-up of complex steroid intermediates.
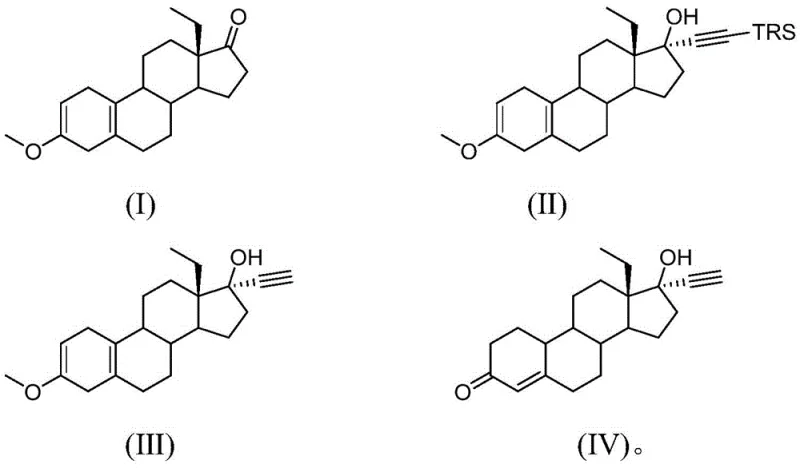
Mechanistic Insights into Organolithium-Mediated Alkynylation and Hydrolysis
The core of this synthetic innovation lies in the precise control of organometallic chemistry. The reaction initiates with the deprotonation of trialkylsilylacetylene by a strong base, typically n-butyllithium or lithium diisopropylamide, in a first organic solvent such as tetrahydrofuran (THF) at cryogenic temperatures ranging from -30°C to 10°C. This generates a reactive lithium acetylide species in situ. This nucleophile then attacks the carbonyl group of the starting material, 18-methylestra-2,5(10)-diene-3-methoxy-17-ketone (Compound I), to form the tertiary alcohol intermediate (Compound II). The use of the silyl protecting group is crucial here; it stabilizes the acetylene moiety during the addition step and prevents side reactions that often plague direct acetylene additions. The stereochemistry is carefully managed to ensure the formation of the desired 17-alpha-ethynyl configuration, which is essential for the biological activity of the final API.
Following the alkynylation, the process demonstrates remarkable elegance in its workup procedure. Instead of a traditional aqueous quench which could be violent with excess organolithium, an alcohol solvent (the second organic solvent) is added directly to the reaction mixture. The residual organolithium reagent reacts with the alcohol to form a metal alkoxide, which subsequently converts to a metal hydroxide upon the addition of water. This in-situ generated base facilitates the removal of the trialkylsilyl group under mild conditions (10-50°C), yielding the free ethynyl intermediate (Compound III). Finally, acid-catalyzed hydrolysis using mineral acids like hydrochloric acid cleaves the methoxy enol ether to restore the conjugated enone system, delivering the final levonorgestrel (Compound IV). This mechanism ensures high selectivity and minimizes the formation of regio-isomeric impurities, a critical factor for meeting stringent pharmacopeial standards.
How to Synthesize Levonorgestrel Efficiently
Implementing this synthesis requires strict adherence to temperature controls and reagent stoichiometry to maximize yield and purity. The patent outlines a robust protocol where the molar ratios of the silyl-acetylene to the ketone precursor are optimized between 1:1 and 2:1. The reaction environment must be kept anhydrous during the initial lithiation phase to prevent premature quenching of the organometallic reagent. Once the alkynylation is complete, the sequential addition of methanol or ethanol followed by water allows for a controlled exotherm during the desilylation phase. The final hydrolysis step is conducted at elevated temperatures (30-70°C) to ensure complete conversion of the enol ether. For detailed operational parameters, safety guidelines, and specific workup procedures, please refer to the standardized synthesis guide below.
- Perform alkynylation by reacting 18-methylestra-2,5(10)-diene-3-methoxy-17-ketone with trialkylsilylacetylene using n-butyllithium in THF at low temperatures (-30°C to 10°C).
- Add an alcohol solvent (e.g., methanol) and water directly to the reaction mixture to remove the trialkylsilyl group via in-situ generated metal hydroxide.
- Conduct acid-catalyzed hydrolysis using hydrochloric acid at 30-70°C to convert the intermediate into the final levonorgestrel product, followed by neutralization and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers tangible strategic benefits beyond mere technical novelty. The elimination of acetylene gas removes a major logistical and safety bottleneck, simplifying the sourcing of raw materials and reducing the regulatory burden associated with storing explosive gases on-site. This inherently safer process translates to lower insurance premiums and reduced downtime for safety inspections. Furthermore, the one-pot nature of the reaction significantly reduces the number of unit operations, thereby lowering energy consumption and labor costs associated with intermediate isolation and drying. These factors collectively contribute to a more resilient and cost-effective supply chain for hormonal intermediates.
- Cost Reduction in Manufacturing: The economic advantages of this method are driven by process intensification. By combining three distinct chemical transformations into a single reactor vessel, the requirement for multiple reaction vessels, filtration units, and drying ovens is eliminated. This consolidation reduces the overall footprint of the manufacturing suite and lowers utility costs. Additionally, the avoidance of expensive and hazardous acetylene gas infrastructure means capital expenditure can be redirected towards capacity expansion or quality control improvements. The higher yields reported in the patent examples further enhance the cost-efficiency by maximizing the output per kilogram of starting material, effectively lowering the cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: Supply continuity is paramount in the pharmaceutical sector. Traditional acetylene-based processes are susceptible to disruptions due to the specialized transport and storage requirements of the gas. In contrast, trialkylsilylacetylene reagents are stable liquids that can be stored in standard chemical warehouses and transported via常规 logistics channels. This stability ensures a consistent supply of key reagents, mitigating the risk of production stoppages. Moreover, the simplified process flow reduces the lead time for batch completion, allowing manufacturers to respond more agilely to fluctuations in market demand for contraceptive APIs.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental sustainability intensify, this synthesis method offers a greener alternative. The reduction in solvent usage and the elimination of heavy metal catalysts (often used in alternative coupling methods) align with green chemistry principles. The waste stream is simpler to treat, primarily consisting of aqueous salts and organic solvents that can be recovered and recycled. This ease of waste management facilitates smoother regulatory approvals and supports corporate sustainability goals. The process has been demonstrated to scale effectively, maintaining high purity and yield even as batch sizes increase, ensuring that commercial production targets can be met without compromising quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical disclosures within patent CN114181272B, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of adopting this route for large-scale production.
Q: Why is the trialkylsilylacetylene method safer than traditional acetylene gas methods?
A: Traditional methods require saturating solvents with explosive acetylene gas, posing significant safety risks and requiring specialized equipment. The novel method uses liquid trialkylsilylacetylene, eliminating explosion hazards and simplifying handling.
Q: How does the one-pot procedure impact production costs?
A: By avoiding the isolation and purification of intermediates, the one-pot method significantly reduces solvent consumption, labor hours, and waste generation, leading to substantial operational cost reductions.
Q: What purity levels can be achieved with this synthesis route?
A: Experimental data from the patent indicates that this method consistently achieves HPLC purity levels exceeding 98%, with optimized examples reaching up to 99.5% after simple recrystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Levonorgestrel Supplier
The synthesis method described in CN114181272B represents a significant leap forward in the manufacturing of hormonal intermediates, offering a blend of safety, efficiency, and scalability that modern pharmaceutical supply chains demand. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to leverage such advanced synthetic routes for our global partners. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to industrial manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of levonorgestrel meets the highest international standards.
We invite procurement leaders and R&D directors to collaborate with us to evaluate the potential of this technology for your specific product pipelines. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data, route feasibility assessments, and samples to verify the superior quality of our intermediates. Let us help you secure a sustainable and competitive supply of high-value pharmaceutical ingredients.
