Advanced Metal-Free Synthesis of Alkenyl Sulfones for Scalable Pharmaceutical Intermediate Production
Introduction to Next-Generation Alkenyl Sulfone Synthesis
The landscape of organic synthesis is constantly evolving towards greener, more efficient methodologies that align with the rigorous demands of modern pharmaceutical manufacturing. A significant breakthrough in this domain is documented in patent CN111269155B, which discloses a novel, metal-free synthetic method for constructing alkenyl sulfone compounds. These structural motifs are pivotal in medicinal chemistry, serving as key intermediates for cysteine protease inhibitors and various bioactive molecules. The disclosed technology leverages a radical cascade strategy initiated by the in situ generation of arylsulfonyl radicals from aryl diazonium salts and sodium metabisulfite. This approach circumvents the traditional reliance on toxic sulfur dioxide gas or unstable sulfonyl chlorides, offering a safer and more operationally simple pathway. For R&D teams and procurement specialists alike, this innovation represents a substantial opportunity to streamline the supply chain for complex sulfur-containing intermediates while adhering to strict environmental compliance standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of alkenyl sulfone scaffolds has been fraught with synthetic challenges that hinder large-scale adoption in industrial settings. Traditional routes often involve Knoevenagel condensation, oxidation of thioethers, or beta-elimination reactions, which frequently suffer from limited substrate applicability and multi-step sequences. A major bottleneck in conventional sulfonylation is the requirement for gaseous sulfur dioxide or pre-formed sulfonyl chlorides, both of which pose significant safety hazards and handling difficulties in a production environment. Furthermore, many existing protocols necessitate the use of transition metal catalysts, which not only inflate raw material costs but also introduce the risk of heavy metal residues in the final product—a critical failure point for pharmaceutical intermediates destined for API synthesis. The need for harsh reaction conditions, such as strong acids or high temperatures, further exacerbates energy consumption and waste generation, making these legacy processes economically and environmentally unsustainable for modern green chemistry initiatives.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the technology outlined in patent CN111269155B introduces a streamlined, one-pot radical addition strategy that operates under exceptionally mild conditions. By utilizing sodium metabisulfite as a solid, easy-to-handle surrogate for sulfur dioxide, the process eliminates the need for specialized gas handling equipment and mitigates safety risks associated with toxic gases. The reaction proceeds at room temperature in 1,2-dichloroethane, driven by the in situ formation of arylsulfonyl radicals which subsequently add to propargyl alcohols. This metal-free paradigm shift ensures that the resulting alkenyl sulfones are free from transition metal contaminants, drastically reducing downstream purification burdens. The robustness of this method is evidenced by its tolerance to a wide array of functional groups, including electron-withdrawing halogens and electron-donating alkyl groups, thereby enabling the rapid diversification of chemical libraries without the need for protecting group strategies or cryogenic conditions.
Mechanistic Insights into Metal-Free Radical Sulfonylation
The elegance of this synthesis lies in its intricate yet efficient radical mechanism, which orchestrates the formation of the carbon-sulfur bond and the subsequent rearrangement in a single operational sequence. The process initiates with the interaction between the aryl diazonium salt and sodium metabisulfite, facilitating the release of sulfur dioxide and the generation of a highly reactive arylsulfonyl radical species. This radical adds regioselectively to the triple bond of the propargyl alcohol substrate, forming a vinyl radical intermediate. Crucially, the system is designed to undergo a spontaneous intramolecular 1,5-hydrogen atom transfer (1,5-HAT), followed by a functional group transfer that stabilizes the molecule into the final alkenyl sulfone architecture. This cascade avoids the need for external oxidants or reductants, relying instead on the inherent thermodynamic driving force of the radical rearrangement. The use of Na2HPO4 as an additive likely plays a role in buffering the reaction medium and stabilizing the radical intermediates, ensuring high conversion rates without the degradation of sensitive functional groups.
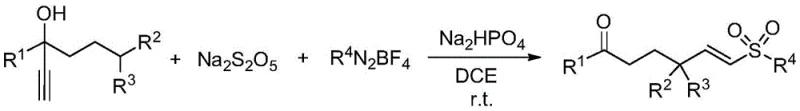
From an impurity control perspective, this mechanism offers distinct advantages over ionic pathways that often generate complex byproduct profiles. Since the reaction does not involve metal catalysts, there is no risk of metal-ligand complexation side reactions or catalyst decomposition products contaminating the crude mixture. The primary byproducts are typically nitrogen gas and inorganic salts, which are easily removed during the aqueous workup or column chromatography stages. The specificity of the 1,5-hydrogen transfer ensures that the double bond geometry is controlled, minimizing the formation of regioisomers that are common in non-catalyzed addition reactions. For quality assurance teams, this translates to a cleaner crude profile and higher overall purity after standard purification, reducing the need for resource-intensive recrystallization steps. The ability to predict and control the radical trajectory allows for precise tuning of the reaction parameters to maximize the yield of the desired E-isomer, which is often the biologically active configuration in drug candidates.
How to Synthesize Alkenyl Sulfone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized protocol to ensure reproducibility and safety. The procedure begins with the careful charging of reagents into a dry reaction vessel under an inert atmosphere to prevent radical quenching by oxygen. The stoichiometry is critical, with a slight excess of sodium metabisulfite and aryl diazonium salt recommended to drive the reaction to completion relative to the more valuable propargyl alcohol substrate. Following the reaction, the workup is remarkably straightforward, involving simple concentration and silica gel chromatography, which aligns perfectly with the needs of process chemists looking to minimize unit operations. For detailed operational parameters and specific troubleshooting tips regarding solvent selection and temperature control, please refer to the standardized synthesis guide below.
- Charge a dry reaction tube with aryl diazonium salt (1.5 equiv), sodium metabisulfite (2.0 equiv), propargyl alcohol (1.0 equiv), and Na2HPO4 (1.5 equiv) at room temperature.
- Purge the system with high-purity nitrogen or argon three times to ensure an oxygen-free environment, then add 1,2-dichloroethane (DCE) as the solvent.
- Stir the mixture at room temperature until TLC indicates complete reaction, then concentrate under reduced pressure and purify via column chromatography using petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free methodology presents a compelling value proposition centered on cost efficiency and supply continuity. The elimination of precious metal catalysts removes a volatile cost component from the bill of materials, shielding the project from fluctuations in the prices of palladium, copper, or rhodium. Moreover, the reliance on commodity chemicals like sodium metabisulfite and aryl diazonium salts ensures a robust and diversified supply base, reducing the risk of single-source bottlenecks that can plague specialized reagent markets. The mild reaction conditions also imply lower energy consumption for heating or cooling, contributing to a reduced carbon footprint and lower utility costs per kilogram of product. These factors collectively enhance the economic viability of producing high-purity alkenyl sulfones at scale, making it an attractive option for long-term commercial contracts.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the complete removal of transition metal catalysts from the process flow. In traditional cross-coupling or sulfonylation reactions, the cost of the catalyst and the subsequent scavenging resins required to remove metal traces can account for a substantial portion of the total manufacturing cost. By switching to a metal-free radical protocol, manufacturers can eliminate these line items entirely, resulting in direct savings on raw material expenditures. Additionally, the simplified workup procedure reduces the consumption of solvents and stationary phase materials during purification, further lowering the variable costs associated with production. This lean manufacturing approach allows for more competitive pricing strategies when bidding for large-volume API intermediate projects.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available, bulk-grade reagents rather than bespoke, custom-synthesized catalysts. Sodium metabisulfite is a global commodity with stable pricing and abundant availability, ensuring that production schedules are not disrupted by reagent shortages. The robustness of the reaction to various substituents means that a single platform technology can be applied to synthesize a diverse library of analogs without needing to requalify new catalyst systems for each derivative. This flexibility accelerates the timeline from process development to commercial launch, allowing supply chain teams to respond more agilely to market demands. Furthermore, the stability of the reagents at room temperature simplifies logistics and storage requirements, reducing the need for cold chain shipping or specialized containment facilities.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden complexities, but this metal-free route is inherently designed for scalability due to its exothermic management and lack of sensitive catalytic species. The absence of heavy metals simplifies the regulatory approval process for new drug applications, as residual metal limits are a stringent checkpoint for regulatory agencies. From an environmental standpoint, the process generates minimal hazardous waste, primarily consisting of inorganic salts that are easier to treat than organometallic sludge. This alignment with green chemistry principles not only reduces waste disposal costs but also enhances the corporate sustainability profile of the manufacturing site. The ability to run the reaction at ambient temperature also lowers the facility's energy load, supporting broader corporate goals for carbon neutrality and energy efficiency in chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this alkenyl sulfone synthesis technology. These insights are derived directly from the experimental data and scope analysis provided in the patent literature, aiming to clarify the practical implications for industrial adoption. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the balance between synthetic efficiency and practical operability that defines this innovative approach.
Q: What are the primary advantages of this metal-free synthesis method over traditional sulfonylation routes?
A: This method eliminates the need for expensive transition metal catalysts and harsh acidic conditions typically required for sulfonyl chloride preparation. It utilizes inexpensive sodium metabisulfite as a solid sulfur dioxide surrogate, operating at room temperature with a broad substrate scope, which significantly simplifies purification and reduces environmental impact.
Q: Can this process be scaled for commercial production of pharmaceutical intermediates?
A: Yes, the process is highly scalable due to its mild reaction conditions (room temperature) and the use of stable, commercially available reagents like aryl diazonium salts and sodium metabisulfite. The absence of sensitive metal catalysts reduces the risk of metal contamination, a critical factor for API intermediate manufacturing.
Q: What is the expected yield range for alkenyl sulfone compounds using this protocol?
A: According to the patent data, the reaction yields range from 32% to 86% depending on the electronic nature of the substituents on the aryl diazonium salt and the propargyl alcohol. Electron-withdrawing and electron-donating groups are both tolerated, providing flexibility for diverse molecular scaffolds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkenyl Sulfone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free synthetic methodologies in advancing the production of high-value pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative lab-scale discoveries like this radical sulfonylation process can be seamlessly translated into robust industrial operations. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of alkenyl sulfone meets the exacting standards required for downstream API synthesis. Our commitment to quality and consistency makes us a trusted partner for global pharmaceutical companies seeking reliable sources of complex sulfur-containing building blocks.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this metal-free approach can optimize your overall budget. Please contact us today to request specific COA data for our available alkenyl sulfone inventory or to discuss route feasibility assessments for your custom synthesis projects. Together, we can drive efficiency and innovation in your supply chain.
