Revolutionizing Mirabegron Intermediate Synthesis: A Safe, Scalable, and Cost-Effective Manufacturing Route
Introduction to Advanced Mirabegron Intermediate Synthesis
The pharmaceutical landscape for treating overactive bladder has been significantly shaped by the introduction of Mirabegron, a beta-3 adrenergic agonist. As global demand for this active pharmaceutical ingredient (API) continues to rise, the efficiency and safety of its supply chain have become paramount concerns for procurement and R&D leadership. Patent CN113880720A introduces a groundbreaking preparation method for the key intermediate, (R)-2-{[2-(4-aminophenyl)ethyl]amino}-1-phenylethanol (Formula III), addressing critical bottlenecks in traditional manufacturing. This innovation shifts the paradigm from hazardous high-pressure hydrogenation and waste-intensive iron reductions to a mild, catalytic system utilizing ferric chloride and hydrazine hydrate.
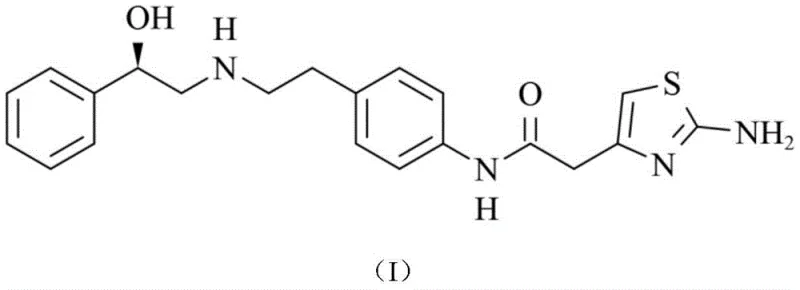
The strategic value of this patent lies in its ability to decouple production from volatile noble metal markets while simultaneously enhancing operational safety. By eliminating the need for pressurized hydrogen gas and expensive palladium catalysts, this methodology offers a robust pathway for reliable pharmaceutical intermediates supplier networks to stabilize costs and ensure continuity. The following analysis dissects the technical superiority of this route, providing actionable insights for stakeholders focused on cost reduction in API manufacturing and the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the critical aniline intermediate (Formula III) has relied heavily on the reduction of the corresponding nitro precursor (Formula IV). The most prevalent industrial approach involves catalytic hydrogenation using 10% palladium on carbon under hydrogen pressure. While effective in terms of yield, this method imposes severe constraints on facility infrastructure, requiring specialized high-pressure reactors that introduce significant safety risks due to the flammability and explosiveness of hydrogen gas. Furthermore, the reliance on palladium, a precious metal with fluctuating market prices, creates substantial cost volatility and supply chain vulnerability.
Alternative methods utilizing iron powder and hydrochloric acid attempt to bypass the need for high-pressure equipment but introduce their own set of logistical nightmares. These processes generate massive quantities of iron sludge waste, creating environmental compliance burdens and complicating post-reaction workups. Additionally, the high density of iron powder necessitates rigorous stirring and mixing protocols to prevent settling, making the process difficult to control and scale uniformly. Other approaches using ammonium formate as a hydrogen donor with palladium catalysts suffer from selectivity issues, often leading to the formation of difficult-to-remove formylated by-products that compromise the purity of the final API.
The Novel Approach
The methodology disclosed in CN113880720A represents a decisive break from these legacy constraints by employing a homogeneous-heterogeneous hybrid catalytic system. This novel approach utilizes ferric chloride hexahydrate as a catalyst in conjunction with hydrazine hydrate as the reducing agent, supported by activated carbon. This combination operates under atmospheric pressure and mild thermal conditions (55-75°C), completely eliminating the need for high-pressure hydrogenation equipment. The use of activated carbon as a suspension carrier effectively mitigates the mixing and settling issues associated with traditional iron powder reductions, ensuring consistent reaction kinetics across large batches.
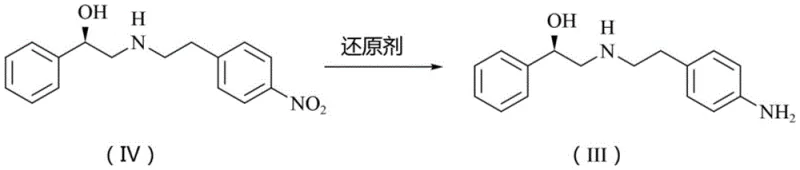
From a commercial perspective, this shift translates to a drastic simplification of the manufacturing workflow. The process avoids the generation of heavy metal waste streams associated with palladium recovery and the solid waste issues of iron reductions. By operating under mild conditions with readily available reagents, the method significantly lowers the barrier to entry for scaling production, offering a streamlined path for reducing lead time for high-purity pharmaceutical intermediates while maintaining rigorous safety standards essential for modern GMP facilities.
Mechanistic Insights into FeCl3-Catalyzed Nitro Reduction
The core innovation of this patent resides in the specific interaction between the ferric chloride catalyst and hydrazine hydrate within the reaction matrix. Unlike traditional catalytic hydrogenation which relies on surface adsorption of hydrogen on a metal lattice, this system likely proceeds through a transfer hydrogenation mechanism where hydrazine serves as the hydrogen source, activated by the Lewis acidic nature of the iron species. The presence of activated carbon plays a dual role: it acts as a physical support to keep the catalytic species dispersed, preventing agglomeration, and potentially aids in the adsorption of organic substrates to facilitate electron transfer. This mechanistic pathway allows for the selective reduction of the nitro group to the amine without affecting other sensitive functional groups present in the molecule, such as the chiral hydroxyl center.
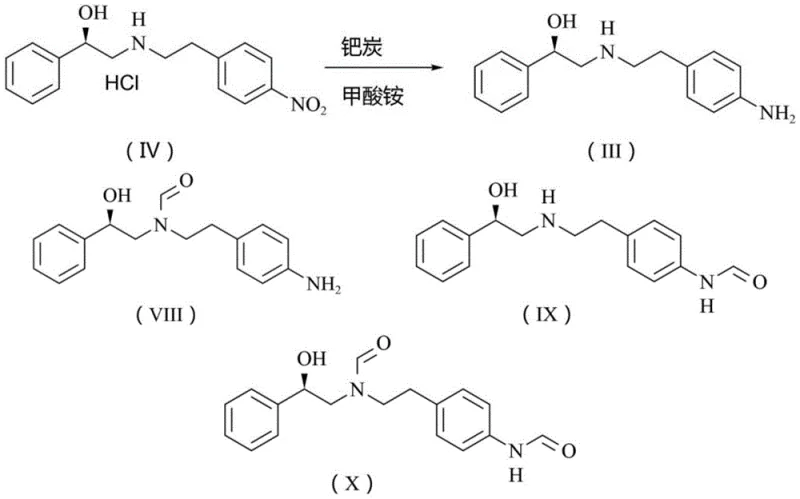
Crucially, this mechanism provides superior impurity control compared to formate-based reduction systems. In methods utilizing ammonium formate, the formyl group can inadvertently transfer to the amine nitrogen, generating N-formyl impurities (Compounds VIII, IX, and X) that are structurally similar to the product and notoriously difficult to separate. The hydrazine/ferric chloride system avoids this side reaction entirely, as evidenced by chromatographic data showing non-detection of these specific impurities. This high selectivity ensures that the resulting intermediate possesses a cleaner impurity profile, reducing the burden on downstream purification steps and safeguarding the quality of the final Mirabegron API against genotoxic or hard-to-remove contaminants.
How to Synthesize Mirabegron Intermediate Efficiently
The synthesis of the target intermediate involves a multi-step sequence starting from chiral mandelic acid derivatives. The process begins with an amide coupling to establish the carbon-nitrogen backbone, followed by a reduction of the amide to the amine, and culminates in the critical nitro reduction step described above. The patent details specific molar ratios and temperature controls that optimize yield and purity, emphasizing the importance of solvent selection (preferably methanol) and catalyst loading. For R&D teams looking to implement this technology, strict adherence to the specified addition rates of hydrazine hydrate is crucial to manage exothermicity and ensure complete conversion.
- Preparation of Amide Precursor (Formula V): React (R)-mandelic acid (Formula VII) with 4-nitrophenethylamine hydrochloride (Formula VI) using EDCI/HOBt coupling agents in DMF at 20-30°C.
- Reduction to Nitro-Alcohol (Formula IV): Reduce the amide (Formula V) using borane dimethyl sulfide complex in THF at reflux temperatures (60-70°C) followed by acidification.
- Catalytic Nitro Reduction (Formula III): Reduce the nitro compound (Formula IV) using hydrazine hydrate, ferric chloride hexahydrate (catalyst), and activated carbon in methanol at 55-75°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this FeCl3-catalyzed route offers tangible strategic benefits that extend beyond simple unit cost calculations. The primary advantage is the decoupling from the volatile precious metals market. By replacing palladium catalysts with abundant iron salts, manufacturers can insulate their production costs from geopolitical fluctuations in platinum group metal prices. Furthermore, the elimination of high-pressure hydrogenation removes the requirement for specialized, capital-intensive reactor vessels, allowing production to be shifted to more versatile multipurpose facilities, thereby increasing overall asset utilization and flexibility in the supply network.
- Cost Reduction in Manufacturing: The patent explicitly states that the process cost is approximately 40% of that associated with palladium-carbon catalysts. This dramatic reduction is driven not only by the cheaper reagent cost but also by the simplified downstream processing. Without the need for extensive filtration to remove fine metal catalysts or the treatment of heavy metal waste, the operational expenditure (OPEX) per kilogram of product is significantly lowered. Additionally, the high yield (reported up to 94.9% in examples) minimizes raw material waste, further enhancing the economic efficiency of the process.
- Enhanced Supply Chain Reliability: The reagents used in this protocol—ferric chloride, hydrazine hydrate, and activated carbon—are commodity chemicals with robust, global supply chains. Unlike specialized catalysts that may have long lead times or single-source dependencies, these materials are readily available from multiple vendors. This redundancy ensures that production schedules are not disrupted by raw material shortages. Moreover, the mild reaction conditions reduce the risk of unplanned shutdowns due to equipment failure or safety incidents, guaranteeing a more consistent and reliable flow of intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns perfectly with green chemistry principles. The absence of high-pressure hydrogen eliminates explosion hazards, lowering insurance premiums and safety compliance costs. The reduction in waste generation, particularly the avoidance of iron sludge and heavy metal residues, simplifies wastewater treatment and solid waste disposal. This environmental friendliness facilitates easier regulatory approval for new manufacturing sites and supports corporate sustainability goals, making the supply chain more resilient to tightening environmental regulations globally.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational parameters.
Q: How does the FeCl3/Hydrazine method compare to Palladium-Carbon hydrogenation in terms of cost?
A: According to patent CN113880720A, the process cost of the new ferric chloride method is approximately 40% of that associated with traditional palladium-carbon metal catalysts, primarily due to the elimination of expensive noble metals and high-pressure equipment requirements.
Q: What specific impurities are avoided by using this new reduction method?
A: Unlike methods using ammonium formate which can lead to formylation side reactions, this method effectively prevents the formation of formylated impurities (Compounds VIII, IX, and X), ensuring higher purity of the final Mirabegron intermediate without difficult purification steps.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method utilizes activated carbon as a carrier for suspension, which resolves dependence on high-shear stirring and mixing often required by iron powder reductions. This makes the process highly scalable, environmentally friendly, and safe for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mirabegron Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced manufacturing processes requires a partner with deep technical expertise and proven execution capabilities. Our team has extensively analyzed the FeCl3-catalyzed reduction pathway and is fully equipped to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are realized in actual manufacturing output. Our state-of-the-art facilities are designed to handle sensitive catalytic reactions with stringent purity specifications, supported by rigorous QC labs that utilize advanced analytical techniques to verify the absence of critical impurities like the formylated by-products.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cost-effective and safe production technology. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your supply chain for Mirabegron intermediates is optimized for both performance and profitability in the coming years.
