Advanced Synthesis of Saxagliptin Intermediates: A Scalable Green Chemistry Approach for Commercial Production
Advanced Synthesis of Saxagliptin Intermediates: A Scalable Green Chemistry Approach for Commercial Production
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for the production of critical antidiabetic agents, particularly DPP-IV inhibitors like saxagliptin. Patent CN114805177A introduces a groundbreaking preparation method for a key saxagliptin intermediate, specifically amino-protected (1S,3S,5S)-2-[(2S)-2-amino-2-(3-hydroxyadamantan-1-yl)acetyl]-2-azabicyclo[3.1.0]hexane-3-carbonitrile, referred to herein as Compound I. This innovation addresses long-standing challenges in amide bond formation involving sterically hindered secondary amines, offering a robust solution for manufacturers aiming to optimize their API supply chains. The structural complexity of Compound I, featuring a rigid adamantane cage fused with a cyclopropyl-pyrrolidine system, demands precise synthetic control to ensure high purity and stereochemical integrity.
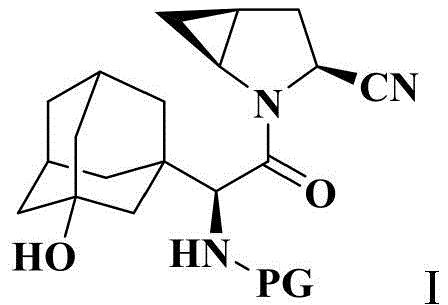
As a reliable saxagliptin intermediate supplier, understanding the nuances of this synthesis is vital for maintaining competitive advantage. The patent outlines a novel approach that bypasses the limitations of traditional coupling methods, utilizing di-tert-butyl dicarbonate and 4-dimethylaminopyridine (DMAP) to activate the carboxylic acid moiety of the precursor. This strategy not only streamlines the reaction sequence but also significantly reduces the environmental footprint by generating benign byproducts such as carbon dioxide and tert-butyl alcohol. For R&D directors and procurement managers alike, this represents a pivotal shift towards greener, more cost-effective manufacturing protocols that do not compromise on the stringent quality standards required for pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of saxagliptin intermediates has been plagued by inefficiencies inherent in forming amide bonds between bulky carboxylic acids and secondary amines. Conventional methods often rely on harsh coupling reagents or multi-step protection-deprotection sequences that result in poor atom economy and substantial waste generation. The steric hindrance presented by the secondary amino group in the azabicyclo[3.1.0]hexane ring system frequently leads to incomplete reactions or the formation of difficult-to-remove impurities, necessitating complex purification procedures. Furthermore, many existing processes require extreme temperatures or the use of toxic solvents, which escalates operational costs and poses significant safety risks in a commercial setting. These factors collectively contribute to longer lead times and higher production costs, creating bottlenecks in the supply of high-purity saxagliptin intermediates.
The Novel Approach
In stark contrast, the method disclosed in CN114805177A leverages the unique reactivity of di-tert-butyl dicarbonate in the presence of a nucleophilic catalyst to activate the carboxyl group of the adamantane glycine derivative. This activation step generates a highly reactive mixed anhydride intermediate in situ, which is sufficiently electrophilic to react with the sterically hindered secondary amine of Compound III without the need for excessive heat or aggressive reagents. The process is conducted in mild aprotic solvents such as ethyl acetate or acetonitrile at ambient temperatures ranging from 20°C to 30°C, drastically reducing energy consumption. By simplifying the reaction pathway and utilizing commercially available reagents, this novel approach offers a direct route to cost reduction in API manufacturing while adhering to the principles of green chemistry. The ability to achieve high yields with minimal side reactions makes this method particularly attractive for large-scale commercial production.
Mechanistic Insights into DMAP-Catalyzed Carboxylic Acid Activation
The core of this synthetic breakthrough lies in the mechanistic interplay between di-tert-butyl dicarbonate and 4-dimethylaminopyridine (DMAP). Initially, DMAP acts as a potent nucleophilic catalyst, attacking the carbonyl carbon of the di-tert-butyl dicarbonate to form a reactive acylpyridinium species. This species then transfers the tert-butoxycarbonyl group to the carboxylic acid of Compound II, generating a mixed anhydride that is far more susceptible to nucleophilic attack than the parent acid. This activation is crucial because the subsequent nucleophile, Compound III, contains a secondary amine within a rigid bicyclic framework, which typically exhibits low nucleophilicity due to steric constraints. The enhanced electrophilicity of the activated carboxyl group overcomes this barrier, facilitating the formation of the amide bond under mild conditions.
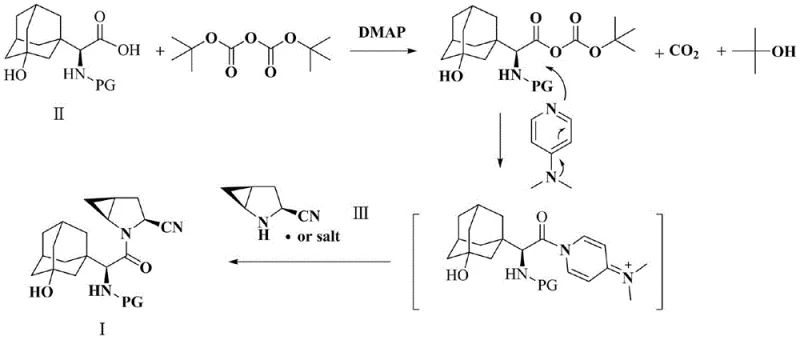
Furthermore, the choice of reagents plays a pivotal role in impurity control and downstream processing. Unlike phosphorus-based coupling reagents that generate stoichiometric amounts of phosphate waste, the byproducts of this reaction—tert-butyl alcohol and carbon dioxide—are volatile and easily removed during the workup phase. The patent specifies a rigorous purification protocol involving sequential washing with dilute acid, water, dilute alkali, and saturated brine, which effectively removes unreacted starting materials, DMAP, and any acidic or basic impurities. This meticulous attention to purification ensures that the final product meets the stringent purity specifications required for pharmaceutical applications. The mechanism thus not only drives the reaction forward efficiently but also inherently simplifies the isolation of the target molecule, reducing the burden on downstream purification units.
How to Synthesize Saxagliptin Intermediate Efficiently
The synthesis of this complex intermediate is designed for operational simplicity and scalability, making it ideal for transfer from laboratory to pilot and commercial scales. The process begins with the dissolution of the amino-protected hydroxyadamantane glycine in a suitable aprotic solvent, followed by the controlled addition of the activating agents. The reaction progress is monitored to ensure complete conversion before proceeding to the workup phase, which involves standard liquid-liquid extraction techniques and vacuum distillation. The detailed parameters regarding stoichiometry, temperature control, and washing sequences are critical for reproducibility and yield optimization. For technical teams looking to implement this route, the following guide outlines the standardized operational steps derived from the patent data.
- Dissolve amino-protected (S)-3-hydroxyadamantane glycine (Compound II) in an aprotic solvent and activate the carboxyl group using di-tert-butyl dicarbonate and 4-dimethylaminopyridine (DMAP).
- Add (1S,3S,5S)-2-azabicyclo[3.1.0]hexane-3-carbonitrile (Compound III) to the activated mixture to form the amide bond under mild stirring conditions.
- Purify the resulting mixture through a sequential washing protocol involving acid, water, alkali, and brine, followed by drying and vacuum distillation to isolate the target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method translates into tangible strategic benefits beyond mere technical feasibility. The reliance on commodity chemicals such as di-tert-butyl dicarbonate and DMAP, rather than specialized or proprietary coupling agents, mitigates the risk of supply disruptions and price volatility. Additionally, the mild reaction conditions eliminate the need for specialized high-pressure or cryogenic equipment, allowing for production in standard stainless steel reactors commonly found in multipurpose chemical facilities. This flexibility enhances supply chain resilience and enables rapid scale-up to meet fluctuating market demands for antidiabetic medications.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of inexpensive, recyclable solvents contribute to a substantial decrease in raw material costs. Moreover, the generation of gaseous carbon dioxide and volatile tert-butyl alcohol as byproducts simplifies waste treatment protocols, leading to significant savings in environmental compliance and disposal fees. The high atom economy of the reaction ensures that a greater proportion of the input mass is converted into the desired product, further driving down the cost per kilogram of the final intermediate.
- Enhanced Supply Chain Reliability: By utilizing raw materials that are widely available in the global chemical market, this process reduces dependency on single-source suppliers for exotic reagents. The robustness of the reaction conditions means that production schedules are less likely to be impacted by minor variations in utility availability or environmental conditions. This reliability is crucial for maintaining continuous supply lines to downstream API manufacturers, ensuring that drug production timelines are met without interruption.
- Scalability and Environmental Compliance: The process is inherently scalable due to its exothermic nature being manageable at ambient temperatures, removing the thermal hazards often associated with large-scale amide couplings. The alignment with green chemistry principles, specifically the reduction of hazardous waste and the use of safer solvents, positions manufacturers favorably regarding increasingly strict environmental regulations. This proactive approach to sustainability not only future-proofs the production facility but also enhances the corporate reputation among stakeholders who prioritize eco-friendly manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this saxagliptin intermediate synthesis. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, offering clarity on process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: Why is di-tert-butyl dicarbonate preferred for activating the carboxylic acid in this synthesis?
A: Di-tert-butyl dicarbonate serves as a highly effective activating agent that generates environment-friendly byproducts like tert-butyl alcohol and carbon dioxide. Unlike traditional coupling reagents that may leave toxic residues, this method aligns with green chemistry principles while effectively overcoming the steric hindrance associated with forming amide bonds with secondary amines.
Q: What are the critical reaction conditions for ensuring high yield in this process?
A: The process operates under remarkably mild conditions, specifically at temperatures between 20°C and 30°C, eliminating the need for energy-intensive heating or cooling systems. The use of 4-dimethylaminopyridine (DMAP) as a catalyst is crucial for facilitating the reaction between the activated carboxylic acid and the sterically hindered secondary amine of the azabicyclo hexane derivative.
Q: How does this method improve supply chain reliability compared to conventional routes?
A: This method utilizes readily available raw materials such as amino-protected hydroxyadamantane glycine and common solvents like ethyl acetate or acetonitrile. By simplifying the synthesis into fewer steps and avoiding exotic or hazardous reagents, the process reduces dependency on complex supply chains and minimizes the risk of production delays caused by raw material shortages.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Saxagliptin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthesis routes in the modern pharmaceutical landscape. Our team of expert chemists has extensively analyzed the methodology described in CN114805177A and possesses the technical capability to execute this process with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our facilities are equipped with state-of-the-art rigorous QC labs that enforce stringent purity specifications, guaranteeing that every batch of saxagliptin intermediate delivered meets the highest international standards for pharmaceutical use.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your antidiabetic drug manufacturing operations.
