Scalable Production of High-Purity Amorphous Ertapenem Intermediates for Global Antibiotic Supply Chains
The pharmaceutical industry continuously seeks robust manufacturing routes for critical antibiotic intermediates, particularly for broad-spectrum carbapenems like ertapenem. Patent CN101875665B introduces a transformative approach to synthesizing ertapenem intermediates, specifically addressing the longstanding challenges of product isolation and purity control. This technology enables the production of the intermediate in a stable, amorphous solid form through a streamlined condensation and precipitation process. By shifting away from complex extraction protocols that often yield oily residues or impure solids, this method ensures a reliable supply of high-quality starting materials for final API synthesis. The strategic implementation of acid-induced precipitation allows manufacturers to bypass traditional bottlenecks, offering a significant advantage for reliable ertapenem intermediate supplier networks aiming to secure global health supply chains against disruption.
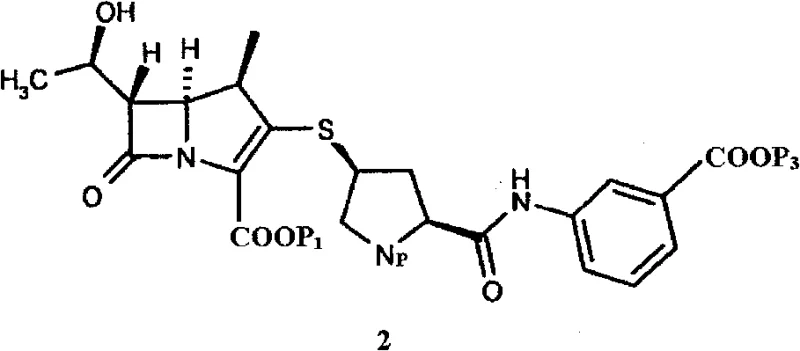
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the isolation of ertapenem intermediates has been plagued by significant downstream processing difficulties that compromise both yield and operational efficiency. Prior art methods, such as those described in WO 2008/062279, often rely on pouring reaction mixtures into buffer solutions or mixed solvent systems containing ethyl acetate. These conventional techniques frequently result in the formation of viscous oils rather than discrete solids, making filtration and washing exceptionally difficult. Furthermore, when solids are obtained through these older routes, they often exhibit poor purity levels, typically falling below 90%, necessitating additional and costly purification steps like column chromatography. The reliance on large volumes of organic solvents for extraction not only increases environmental burden but also introduces safety hazards and escalates production costs, creating a fragile supply chain vulnerable to solvent price volatility and regulatory scrutiny regarding waste disposal.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes a direct acid precipitation strategy that fundamentally simplifies the isolation workflow. By quenching the condensation reaction mixture directly into an aqueous acid solution with a controlled pH range of 2 to 6, the intermediate precipitates immediately as a free-flowing amorphous powder. This elegant solution eliminates the need for organic solvent extraction during the workup phase, drastically reducing solvent consumption and waste generation. The resulting solid possesses excellent physical properties, including high bulk density and ease of handling, which facilitates automated processing in large-scale reactors. As illustrated in the reaction scheme below, the direct conversion from soluble precursors to an isolable solid streamlines the entire manufacturing timeline, supporting the commercial scale-up of complex antibiotic intermediates with unprecedented reliability and consistency.
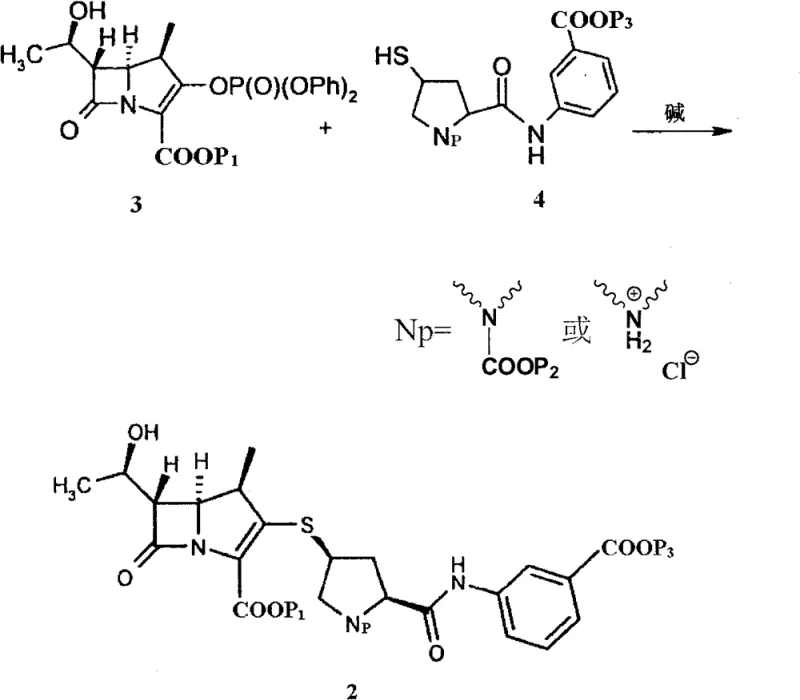
Mechanistic Insights into Base-Catalyzed Condensation and Acid Precipitation
The core of this synthesis lies in the nucleophilic attack of the thiol group on the ertapenem side chain onto the activated leaving group of the carbapenem nucleus, typically a phosphate or similar activating group at the C3 position. The reaction is conducted in a polar aprotic solvent like DMF at low temperatures, often around -35°C, to preserve the stereochemical integrity of the sensitive beta-lactam ring system. The use of a non-nucleophilic base, such as diisopropylethylamine, ensures efficient deprotonation of the thiol without inducing beta-lactam ring opening or epimerization at the C5/C6 positions. This precise control over reaction conditions is critical for maintaining the biological activity of the final antibiotic, as even minor stereochemical deviations can render the molecule ineffective against resistant bacterial strains.
Following the condensation, the mechanism shifts to a protonation-driven precipitation event that serves as a powerful purification tool. When the reaction mixture is introduced into the acidic aqueous medium, the carboxylate groups on the intermediate are protonated to their free acid forms, significantly reducing their solubility in the aqueous environment. This sudden change in solubility parameters forces the product out of the solution as a solid, while many polar impurities and unreacted starting materials remain dissolved in the aqueous phase or are washed away. This selective precipitation acts as an in-situ purification step, effectively removing residual side chains and inorganic salts without the need for additional chromatographic media, thereby ensuring the high-purity ertapenem intermediates required for subsequent deprotection steps meet stringent regulatory specifications.
How to Synthesize Ertapenem Intermediate Efficiently
The synthesis protocol outlined in the patent provides a clear pathway for manufacturing teams to replicate these high-yield results in a GMP environment. The process is designed to be operationally simple, requiring standard reactor equipment capable of low-temperature control and efficient mixing. Operators must pay close attention to the pH of the precipitation bath, as maintaining the acidity within the optimal window of 2.5 to 4.5 ensures maximum recovery and purity. Detailed standard operating procedures regarding reagent addition rates and temperature profiles are essential to prevent localized exotherms that could degrade the sensitive carbapenem core. For a comprehensive breakdown of the specific operational parameters and safety considerations, please refer to the standardized guide below.
- Dissolve the protected carbapenem nucleus and ertapenem side chain in a polar aprotic solvent such as DMF under inert atmosphere.
- Add an organic base dropwise at low temperature (approximately -35°C) to initiate the condensation reaction while maintaining strict thermal control.
- Pour the reaction mixture into an aqueous acid solution with a pH between 2 and 6 to precipitate the product as a free-flowing amorphous solid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain directors, the adoption of this amorphous solid synthesis route offers tangible benefits that extend far beyond simple chemical yield improvements. The elimination of organic solvents from the isolation phase represents a major shift in cost structure, removing the need for expensive solvent recovery systems and reducing the volume of hazardous waste requiring disposal. This simplification of the post-reaction workup translates directly into shorter batch cycles and increased throughput capacity, allowing manufacturers to respond more agilely to market demand fluctuations. Furthermore, the robustness of the precipitation method across a wide range of acid types provides flexibility in raw material sourcing, mitigating risks associated with the supply of specific reagents.
- Cost Reduction in Manufacturing: The transition to an aqueous acid workup fundamentally alters the cost equation by removing the most expensive unit operations from the process. Traditional methods relying on ethyl acetate extractions and subsequent evaporations consume vast amounts of energy and solvent, whereas this new method utilizes inexpensive mineral or organic acids and water. By avoiding the use of chromatography columns and complex recrystallization solvents, the overall cost of goods sold is significantly lowered. This efficiency gain allows for cost reduction in carbapenem manufacturing that can be passed down the supply chain, making life-saving antibiotics more economically accessible without compromising on quality standards.
- Enhanced Supply Chain Reliability: The ability to produce a stable, free-flowing amorphous solid greatly enhances the logistics and storage capabilities of the intermediate. Unlike oily residues that require immediate processing or specialized cold storage to prevent degradation, this solid form can be stored for extended periods with minimal risk of quality loss. This stability acts as a buffer against supply chain disruptions, allowing manufacturers to build strategic inventory reserves. Additionally, the high purity achieved directly from the reactor reduces the dependency on external purification services, consolidating the supply chain and reducing lead time for high-purity ertapenem intermediates delivery to downstream API producers.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the reduction in organic solvent usage aligns perfectly with modern green chemistry initiatives and increasingly strict environmental regulations. The process generates significantly less hazardous waste, simplifying compliance reporting and lowering disposal fees. The simplicity of the filtration and washing steps means that the process scales linearly from pilot plant to multi-ton commercial production without encountering the mixing or heat transfer limitations often seen in complex extraction workflows. This scalability ensures that the technology can support global demand surges, providing a sustainable foundation for long-term production planning.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and validation of this synthesis method. Understanding the nuances of the amorphous form and the flexibility of the acid precipitation step is crucial for technology transfer teams evaluating this process for adoption. The answers provided are derived directly from the experimental data and embodiments detailed in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What is the physical form of the ertapenem intermediate produced by this method?
A: The process yields the intermediate in an amorphous solid form, which offers superior free-flowing properties and storage stability compared to prior art methods that often result in oils or low-purity solids.
Q: Which acids are suitable for the precipitation step?
A: The method is highly versatile and supports a wide range of inorganic and organic acids, including hydrochloric acid, sulfuric acid, phosphoric acid, and acetic acid, provided the final pH is maintained between 2 and 6.
Q: What purity levels can be achieved with this synthesis route?
A: By utilizing the optimized acid precipitation technique, HPLC purity levels exceeding 98% are consistently achieved without the need for complex chromatographic purification or recrystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ertapenem Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the consistent supply of high-quality intermediates is the backbone of the global antibiotic market. Our technical team has extensively analyzed advanced synthetic routes like the one described in CN101875665B to ensure our manufacturing capabilities remain at the forefront of innovation. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you need pilot quantities for clinical trials or metric tons for commercial launch, our facilities are ready to deliver. Our rigorous QC labs enforce stringent purity specifications, guaranteeing that every batch of ertapenem intermediate meets the exacting standards required for regulatory submission and patient safety.
We invite potential partners to engage with our technical procurement team to discuss how this advanced amorphous synthesis technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this more efficient process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your specific production requirements, ensuring a seamless transition to a more robust and cost-effective supply model.
