Scalable Synthesis of Ertapenem Intermediates via Mild Mesylation Technology
The pharmaceutical landscape for carbapenem antibiotics continues to evolve, driven by the critical need for efficient, scalable, and cost-effective manufacturing processes for life-saving drugs like Ertapenem. Patent CN101376641A introduces a groundbreaking advancement in the synthesis of key intermediates required for Ertapenem production, specifically addressing the bottlenecks associated with traditional side-chain assembly. This intellectual property details a novel mesylation strategy that transforms the preparation of the pyrrolidine-based side chain, shifting away from the energy-intensive and operationally complex cryogenic protocols of the past. By enabling reactions to proceed under mild conditions, typically between -5°C and 0°C, this technology offers a robust pathway for producing high-purity intermediates essential for the final antibiotic assembly. For global supply chain leaders and R&D directors, this represents a significant opportunity to optimize the manufacturing footprint of broad-spectrum antibiotics, ensuring both economic viability and consistent quality in the face of increasing regulatory scrutiny.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Ertapenem side chains has been plagued by significant operational challenges that hinder efficient scale-up and cost management. Prior art, such as the methodologies described in US5872250 and US6180783, relies heavily on "one-pot" reactions that must be maintained at严苛 low temperatures, often reaching -20°C or lower. Such cryogenic requirements impose a substantial burden on manufacturing infrastructure, necessitating specialized cooling equipment and driving up energy consumption drastically. Furthermore, these conventional routes frequently employ expensive and hazardous reagents, including diisopropylethylamine and diphenylphosphoryl chloride, which not only inflate raw material costs but also complicate waste disposal and environmental compliance. The reliance on column chromatography for purification in many of these older methods further exacerbates the issue, creating a bottleneck that limits throughput and introduces variability in product quality, making it difficult to achieve the consistent high purity demanded by modern pharmacopeial standards for active pharmaceutical ingredients.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN101376641A presents a streamlined and economically superior alternative that fundamentally reimagines the synthetic route. The core innovation lies in the utilization of a mild mesylation reaction to convert the hydroxy-proline precursor into the activated mesylate intermediate (Formula V) under significantly more forgiving thermal conditions. By operating at temperatures ranging from -5°C to 0°C, or even room temperature in certain optimization scenarios, this process eliminates the need for deep cryogenic cooling, thereby reducing capital expenditure on refrigeration systems and lowering ongoing utility costs. The protocol utilizes readily available and cost-effective reagents such as methanesulfonyl chloride and triethylamine, replacing the expensive phosphorylating agents of the past. Moreover, the workup procedure is remarkably simple, involving standard aqueous washes, drying, and crystallization to yield the product with high purity, effectively bypassing the need for time-consuming and solvent-intensive column chromatography. This shift not only accelerates production cycles but also enhances the overall safety profile of the manufacturing process by minimizing the handling of hazardous materials under extreme conditions.
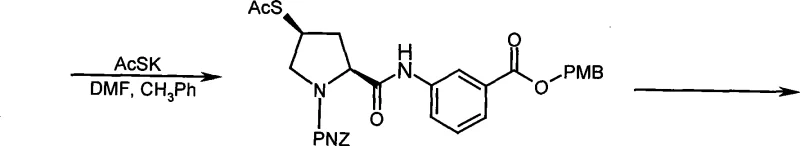
Mechanistic Insights into Mild Mesylation and Side Chain Assembly
The chemical elegance of this patented process is rooted in the precise control of stereochemistry and reactivity during the mesylation of the pyrrolidine ring. The transformation involves the reaction of the hydroxyl group on the proline scaffold with methanesulfonyl chloride in the presence of an organic base, typically triethylamine, within a non-polar solvent system like dichloromethane. This specific set of conditions is crucial for maintaining the integrity of the chiral centers at the 2S and 4R positions, which are vital for the biological activity of the final Ertapenem molecule. The mild thermal environment prevents potential side reactions such as elimination or racemization that could occur under more vigorous conditions, ensuring that the optical purity of the intermediate remains exceptionally high throughout the synthesis. The use of a slight excess of methanesulfonyl chloride, optimized to a molar ratio of 1.5 to 3 times relative to the substrate, drives the reaction to completion efficiently without generating excessive byproducts that would complicate downstream purification. This mechanistic precision allows for the direct isolation of the mesylate intermediate as a crystalline solid, facilitating easy handling and storage prior to the subsequent coupling steps required to build the final antibiotic structure.
Furthermore, the impurity profile of the resulting intermediate is tightly controlled through the strategic selection of protecting groups and reaction solvents. The use of the p-nitrobenzyloxycarbonyl (PNZ) group on the nitrogen atom and the p-methoxybenzyl (PMB) group on the aniline moiety provides orthogonal stability, ensuring that these functionalities remain intact during the mesylation step while being amenable to removal in later stages of the synthesis. The choice of dichloromethane as the reaction medium offers excellent solubility for both the organic substrates and the inorganic salts formed during the neutralization of the generated HCl, promoting a homogeneous reaction mixture that maximizes contact between reagents. Post-reaction, the sequential washing with dilute acid, saturated salt solutions, and bicarbonate effectively removes residual amines, unreacted acid chlorides, and acidic byproducts, resulting in a crude product that is already of high purity. This rigorous control over the chemical environment minimizes the formation of difficult-to-remove impurities, thereby reducing the burden on final purification steps and ensuring that the intermediate meets the stringent quality specifications required for GMP manufacturing of pharmaceutical actives.
How to Synthesize Ertapenem Intermediate Formula V Efficiently
The practical implementation of this synthesis route is designed for seamless integration into existing pharmaceutical manufacturing facilities, requiring only standard reactor setups and common chemical feedstocks. The process begins with the protection and functionalization of the starting materials, followed by the critical mesylation step that defines the novelty of this approach. Operators are guided to maintain strict temperature control during the addition of methanesulfonyl chloride to manage the exotherm, although the range is far more accessible than the -20°C required by older patents. The detailed standardized synthesis steps below outline the precise stoichiometry, timing, and workup procedures necessary to achieve the reported high yields and purity levels consistently. By adhering to these optimized parameters, production teams can minimize batch-to-batch variability and maximize the efficiency of their raw material utilization, ensuring a reliable supply of this critical intermediate for the global market.
- Protect the starting amine with PMB-Cl in dichloromethane using triethylamine at 45°C.
- Reduce the nitro group to an amine using stannous chloride dihydrate in ethanol at 50°C.
- Couple the resulting aniline with activated PNZ-L-hydroxyproline ester in dichloromethane at low temperature.
- Perform the key mesylation reaction on the hydroxy-proline moiety using methanesulfonyl chloride and triethylamine at -5°C to 0°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible strategic advantages that extend far beyond simple chemical efficiency. The primary value proposition lies in the substantial reduction of operational complexity and associated costs, which directly impacts the bottom line of API manufacturing. By eliminating the dependency on deep cryogenic cooling systems, facilities can repurpose existing infrastructure for other campaigns or avoid the capital investment entirely, leading to significant long-term savings in both equipment maintenance and energy consumption. The substitution of expensive, specialized reagents with commodity chemicals like methanesulfonyl chloride and triethylamine further de-risks the supply chain, as these materials are widely available from multiple global vendors, reducing the likelihood of shortages or price volatility. Additionally, the simplified workup procedure, which relies on crystallization rather than chromatography, drastically reduces solvent consumption and waste generation, aligning with increasingly strict environmental regulations and sustainability goals that modern pharmaceutical companies must meet. These factors combine to create a manufacturing process that is not only cheaper but also more resilient and environmentally responsible, providing a competitive edge in the crowded marketplace of generic and branded antibiotics.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the elimination of energy-intensive cryogenic operations and the replacement of high-cost reagents with affordable alternatives. Traditional methods requiring -20°C conditions demand specialized chillers and insulated reactors that consume vast amounts of electricity, whereas this new route operates near ambient or mild cooling temperatures, slashing utility bills significantly. Furthermore, the avoidance of column chromatography reduces the volume of silica gel and elution solvents required, lowering both material costs and the expenses associated with solvent recovery and disposal. The high yield and purity achieved directly translate to better mass balance and less raw material waste, ensuring that every kilogram of input generates maximum output value for the organization.
- Enhanced Supply Chain Reliability: From a sourcing perspective, this methodology leverages a portfolio of reagents that are commercially available in bulk quantities from established chemical suppliers worldwide. Unlike proprietary or niche catalysts that may have limited supply chains, reagents such as methanesulfonyl chloride and triethylamine are commodity products with robust logistics networks, ensuring continuity of supply even during market fluctuations. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality or environmental factors, reducing the risk of batch failures that can disrupt production schedules. This reliability allows supply chain planners to forecast demand more accurately and maintain leaner inventory levels without compromising the ability to meet customer orders, ultimately strengthening the overall resilience of the pharmaceutical supply network.
- Scalability and Environmental Compliance: The simplicity of the unit operations involved in this synthesis makes it inherently scalable from pilot plant to commercial tonnage production without the need for complex engineering modifications. The absence of difficult separations like chromatography means that larger batches can be processed in standard stirred-tank reactors using straightforward filtration and drying equipment, accelerating the timeline from development to market launch. Environmentally, the process generates less hazardous waste due to the use of safer reagents and reduced solvent volumes, facilitating easier compliance with local and international environmental protection laws. The ability to recycle solvents like dichloromethane and ethyl acetate further enhances the green chemistry profile of the process, appealing to stakeholders who prioritize sustainability and corporate social responsibility in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Ertapenem intermediate synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational feasibility. Understanding these details is crucial for technical teams evaluating the transition from legacy processes to this improved methodology, as it highlights the specific areas where value is created through chemical innovation. The responses cover aspects ranging from reaction conditions and purity profiles to the broader implications for industrial scale-up and regulatory compliance.
Q: How does this synthesis route improve upon previous methods like US5872250?
A: Unlike prior art requiring harsh cryogenic conditions (-20°C) and expensive reagents like diphenylphosphoryl chloride, this novel route operates at mild temperatures (-5°C to 0°C) using cost-effective methanesulfonyl chloride, significantly lowering equipment and reagent costs.
Q: What purity levels can be achieved with this mesylation process?
A: The process consistently yields the intermediate (Formula V) with purity greater than 95%, often exceeding 99% after simple crystallization, eliminating the need for complex column chromatography purification steps.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method is specifically designed for industrial scalability, utilizing simple workup procedures like washing, drying, and filtration, and avoiding sensitive low-temperature constraints that hinder large-batch processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ertapenem Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex antibiotics like Ertapenem depends on a partnership grounded in technical excellence and supply chain reliability. As a premier CDMO and manufacturer, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped to handle the specific requirements of this mild mesylation process, including precise temperature control and advanced purification capabilities to meet stringent purity specifications. With rigorous QC labs dedicated to monitoring every batch for impurities and stereochemical integrity, we guarantee that our Ertapenem intermediates serve as a flawless foundation for your final API synthesis, minimizing risk and maximizing efficiency in your manufacturing operations.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific production needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this technology within your own supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and competitiveness in the global pharmaceutical market. Let us be your trusted partner in delivering high-quality chemical solutions that power the next generation of life-saving medicines.
