Industrial Scale-Up of Imipenem: A Novel Solvent Inclusion Strategy for High-Purity Carbapenem Production
Industrial Scale-Up of Imipenem: A Novel Solvent Inclusion Strategy for High-Purity Carbapenem Production
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical antibiotics, and the synthesis of Imipenem represents a pinnacle of complex beta-lactam chemistry. Patent CN101921274A discloses a groundbreaking industrialized process that fundamentally restructures the production workflow for this super-broad-spectrum carbapenem antibiotic. By introducing a unique solvent inclusion technique for key intermediates, this method addresses long-standing challenges regarding stability, purification, and operational efficiency that have plagued previous synthetic routes. For R&D directors and supply chain leaders, this technology offers a compelling value proposition: a streamlined pathway that minimizes unit operations while maximizing the integrity of the sensitive beta-lactam ring structure throughout the synthesis. The strategic elimination of intermediate isolation steps not only reduces the physical footprint of the manufacturing process but also mitigates the risk of degradation associated with handling unstable intermediates in solid form.
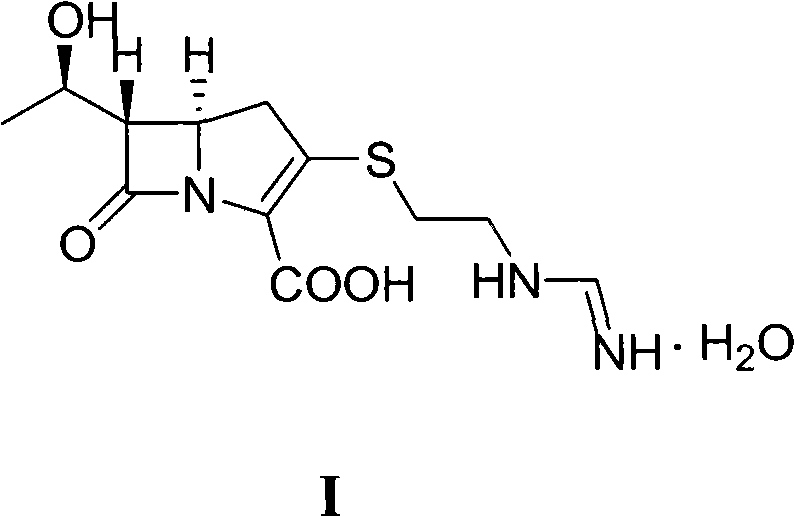
Imipenem, known chemically as a carbapenem beta-lactam antibiotic developed by Merck, possesses potent antimicrobial activity against a wide range of Gram-positive, Gram-negative, aerobic, and anaerobic bacteria. Its clinical significance is underscored by its formulation with cilastatin sodium, an organic ion transport inhibitor, to prevent renal metabolism. However, the chemical instability of the beta-lactam ring and the sensitivity of the formimidoyl side chain have historically made large-scale production technically demanding. Traditional methods often struggle with strict pH controls, complex protection-deprotection sequences, and low overall yields due to the accumulation of impurities at each isolation stage. The methodology presented in CN101921274A circumvents these bottlenecks by leveraging a continuous reaction sequence where the intermediate VI is stabilized as a solvent inclusion compound, thereby preserving its reactivity for the subsequent condensation and hydrogenation steps without the need for rigorous drying or purification that typically leads to product loss.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthetic routes for Imipenem have been fraught with operational complexities that hinder cost-effective commercial scale-up. For instance, early methods disclosed in US4374772 involved the direct reaction of thienamycin derivatives with imine benzyl ethers, a process that demanded extremely harsh reaction conditions and exceptionally strict pH control, making it unsuitable for reliable industrial application. Other approaches, such as those described in Tetrahedron Letters and CN200610111831, utilized double-protection strategies involving nitrobenzyl groups. While conceptually sound, these routes suffered from the inherent instability of the nitrobenzyl-protected side chains, which were prone to losing the formimino group during the hydrogenation process, leading to the formation of unwanted thienamycin byproducts and complicating the final purification. Furthermore, methods employing two-step side chain connections, as seen in US4894450, often resulted in excessive impurity profiles after hydrogenation, necessitating resource-intensive purification protocols that eroded overall process economics.
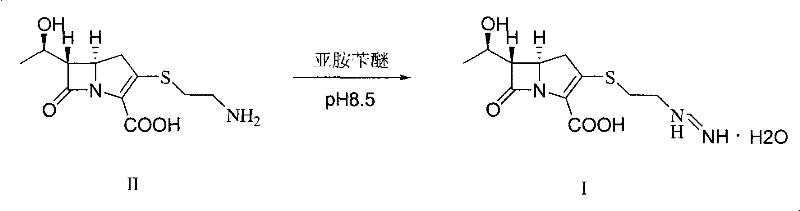
The Novel Approach
The innovative process detailed in CN101921274A introduces a paradigm shift by utilizing a solvent inclusion intermediate (VI) that bridges the gap between the bicyclic nucleus and the final active pharmaceutical ingredient. Unlike Route 5 (WO2002095034), which required a cumbersome solvent wash step to remove solvents from the inclusion compound before further reaction, this novel method allows the solvent inclusion VI to proceed directly to condensation with imine benzyl ether. This telescoping of steps eliminates the mechanical losses and exposure to degrading conditions associated with isolating and washing the intermediate. The reaction scheme demonstrates a seamless transition from the phosphorylated intermediate IV to the solvent inclusion VI, and subsequently to intermediate VII, which is then subjected to catalytic hydrogenation without any separation. This continuity ensures that the reactive species are maintained in an optimal chemical environment, significantly enhancing the liquid phase purity of the final Imipenem solution to levels exceeding 93%, a benchmark that simplifies downstream crystallization and ensures consistent product quality.
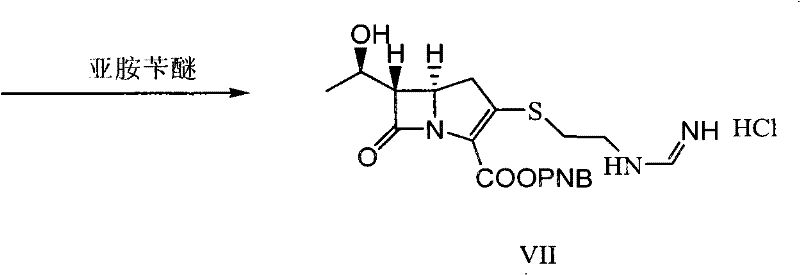
Mechanistic Insights into Solvent Inclusion Stabilization and Catalytic Hydrogenation
The core mechanistic advantage of this process lies in the formation and utilization of the solvent inclusion compound VI. In traditional synthesis, the intermediate containing the cysteamine side chain is highly susceptible to degradation and polymerization if not handled with extreme care. By precipitating this intermediate as a solvent inclusion complex using specific solvent systems—such as mixtures of chloroalkanes, nitriles, alcohols, or esters—the process effectively locks the molecule into a stable crystal lattice. This stabilization prevents the decomposition of the sensitive beta-lactam ring and preserves the nucleophilicity of the amino group required for the subsequent condensation reaction. The patent specifies that solvents like acetonitrile or ethyl acetate can be used to induce this crystallization, creating a solid form that is easier to handle than an oil or unstable solution, yet retains enough solvated character to redissolve readily for the next reaction step without requiring aggressive drying that could trigger thermal degradation.
Furthermore, the final catalytic hydrogenation step is optimized to minimize side reactions that typically plague carbapenem synthesis. The process employs a buffered hydrogenation system where organic bases, such as N-methylmorpholine or lutidines, are added to maintain a precise pH range of 7.2 to 7.5. This pH control is critical because the formimidoyl group is acid-sensitive, and deviations can lead to hydrolysis or rearrangement. The use of palladium carbon or platinum carbon catalysts with controlled metal loading (0.5% to 40%) ensures efficient removal of the benzyl protecting groups while leaving the delicate beta-lactam core intact. The ability to perform this hydrogenation directly on the crude condensation mixture (Intermediate VII) without prior purification is a testament to the cleanliness of the preceding steps, as the solvent inclusion strategy effectively filters out many potential impurities during the crystallization of VI, resulting in a hydrogenation feed stream that is remarkably free of catalyst poisons or competing reactive species.
How to Synthesize Imipenem Efficiently
The synthesis of Imipenem via this patented route involves a carefully orchestrated sequence of phosphorylation, nucleophilic substitution, and catalytic reduction, all designed to maximize throughput and minimize waste. The process begins with the activation of the bicyclic nucleus III using phosphoryl chloride to generate the enol phosphate intermediate IV, which is then immediately reacted with cysteamine hydrochloride. The addition of a specific anti-solvent induces the precipitation of the solvent inclusion compound VI, which serves as the pivotal stable intermediate. Detailed standardized operating procedures for temperature control, reagent addition rates, and crystallization times are essential to replicate the high purity and yield reported in the patent embodiments. For a comprehensive breakdown of the specific molar ratios, solvent volumes, and reaction temperatures required to execute this synthesis successfully, please refer to the technical guide below.
- React bicyclic nucleus III with phosphoryl chloride to form intermediate IV, then react directly with cysteamine hydrochloride and add a specific solvent system to precipitate solvent inclusion compound VI.
- Subject the solvent inclusion VI to condensation with imine benzyl ether to generate intermediate VII, maintaining the reaction mixture without isolation.
- Perform direct catalytic hydrogenation on the crude intermediate VII mixture using palladium or platinum catalysts to yield high-purity Imipenem I.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this solvent inclusion technology translates into tangible operational efficiencies and risk mitigation. The primary economic driver is the drastic simplification of the manufacturing workflow; by eliminating the isolation and solvent-washing steps for intermediate VI and the isolation of intermediate VII, the process reduces the number of batch cycles and equipment turnover times. This reduction in unit operations directly correlates to lower labor costs, decreased energy consumption for drying and solvent recovery, and a smaller physical footprint required for production. Moreover, the enhanced stability of the solvent inclusion intermediate reduces the risk of batch failures due to intermediate degradation, a common issue in carbapenem manufacturing that can lead to significant financial losses and supply disruptions. The ability to telescope multiple reaction steps into a continuous sequence also accelerates the overall production timeline, allowing for faster response to market demand fluctuations.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation and washing steps significantly lowers the consumption of processing solvents and utilities. By avoiding the need to dry and re-dissolve unstable intermediates, the process reduces the thermal stress on the product, thereby minimizing yield losses associated with degradation. The streamlined workflow requires fewer reactor vessels and less manpower per kilogram of finished API, leading to substantial cost savings in variable manufacturing expenses. Additionally, the high liquid phase purity achieved (>93%) reduces the burden on downstream purification and crystallization units, further optimizing the cost structure of the final product.
- Enhanced Supply Chain Reliability: The robustness of the solvent inclusion strategy ensures a more consistent supply of high-quality intermediates, reducing the variability that often plagues multi-step antibiotic syntheses. The use of commercially available reagents and standard catalysts like palladium carbon ensures that raw material sourcing remains stable and unaffected by niche supply constraints. The process tolerance for slight variations in reaction conditions, afforded by the stabilizing effect of the solvent lattice, enhances the reliability of batch-to-batch reproducibility, which is critical for maintaining regulatory compliance and uninterrupted supply to downstream formulation partners.
- Scalability and Environmental Compliance: This methodology is inherently scalable, as the telescoped reactions reduce the total volume of waste solvents generated per unit of product. The avoidance of complex protection-deprotection sequences involving nitrobenzyl groups eliminates the generation of specific hazardous waste streams associated with those moieties. The process aligns well with green chemistry principles by improving atom economy through higher yields and reducing the E-factor (mass of waste per mass of product). The simplified equipment requirements also facilitate easier technology transfer to larger scale reactors, ensuring that commercial production can be ramped up quickly to meet global antibiotic demand without compromising environmental safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this novel Imipenem synthesis route. These insights are derived directly from the experimental data and process descriptions found in patent CN101921274A, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding the nuances of solvent selection and catalyst performance is key to successfully adopting this method for commercial production.
Q: What is the primary advantage of using the solvent inclusion intermediate VI in Imipenem synthesis?
A: The use of solvent inclusion intermediate VI eliminates the need for complex isolation and solvent washing steps required in conventional methods. This telescoping of reactions significantly reduces processing time and material loss, while the specific solvent system enhances the stability and purity of the intermediate prior to the final hydrogenation step.
Q: Which catalysts are suitable for the final hydrogenation step in this novel process?
A: The patent specifies the use of palladium carbon (Pd/C) or platinum carbon (Pt/C) catalysts with metal content ranging from 0.5% to 40%. The process allows for flexibility in catalyst loading, typically between 0.1% to 10% mass ratio relative to the intermediate, ensuring efficient conversion under mild temperature conditions.
Q: What liquid phase purity can be achieved with this manufacturing method?
A: Analytical data from the patent embodiments indicates that the liquid phase purity of the resulting Imipenem solution can reach above 93%. This high initial purity facilitates downstream crystallization processes, allowing for the direct production of Imipenem monohydrate crystals with excellent quality specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imipenem Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and reliable synthesis routes for life-saving antibiotics like Imipenem. Our team of expert chemists has extensively analyzed the solvent inclusion technology described in CN101921274A and possesses the technical capability to implement this advanced process at an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless and compliant with international regulatory standards. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Imipenem intermediate or API we produce adheres to the highest quality benchmarks required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this streamlined process can optimize your total cost of ownership. Please contact us today to request specific COA data for our Imipenem intermediates and to discuss route feasibility assessments for your upcoming projects. Let us partner with you to secure a sustainable and cost-effective supply of high-purity carbapenem antibiotics.
