Advanced Purification Technology for High-Purity Imipenem Monohydrate: A Commercial Scale-Up Perspective
Introduction to Next-Generation Imipenem Purification
The pharmaceutical landscape for carbapenem antibiotics demands increasingly stringent purity profiles to ensure patient safety and therapeutic efficacy. Patent CN112142740B introduces a transformative approach to the preparation of high-purity imipenem monohydrate, addressing long-standing challenges in the refinement of this critical beta-lactam antibiotic. The core innovation lies in the utilization of a specific ammonium chloride-ammonia water inorganic buffer solution, which creates a thermodynamically stable environment for the fragile imipenem molecule during the dissolution and crystallization phases. This technology represents a significant leap forward for any reliable pharmaceutical intermediates supplier aiming to deliver material that meets the rigorous standards of global regulatory bodies. By shifting away from unstable aqueous environments and complex chromatographic separations, this process ensures that the final active pharmaceutical ingredient retains its structural integrity while achieving exceptional purity levels.
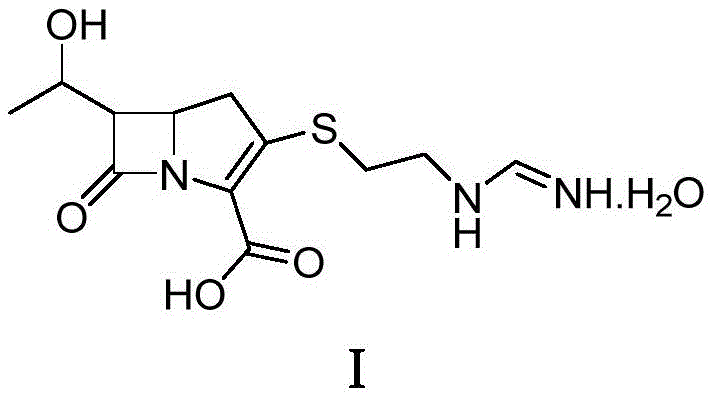
The structural complexity of imipenem, characterized by its fused beta-lactam and pyrroline rings along with a sensitive side chain, makes it highly susceptible to degradation under non-optimal conditions. The patented method specifically targets the stabilization of this intricate architecture, as seen in the molecular diagram, ensuring that the potent antibacterial activity is preserved throughout the manufacturing workflow. For R&D directors and process chemists, this offers a robust pathway to minimize batch-to-batch variability and maximize yield without compromising on quality attributes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of imipenem has been plagued by significant technical hurdles that impact both cost and quality. Traditional synthetic routes, particularly Route 2 which utilizes a carbapenem bicyclic mother nucleus, often result in the formation of persistent impurities that are notoriously difficult to remove. A primary concern is the generation of Impurity II, a dimeric byproduct formed during catalytic hydrogenation steps, which co-elutes with the product in many standard purification systems. Furthermore, conventional refining methods such as column chromatography and freeze-drying, while effective on a small scale, are economically prohibitive and operationally cumbersome for industrial applications. These methods suffer from low throughput, high solvent consumption, and the risk of product degradation due to prolonged exposure to varying pH conditions or thermal stress.
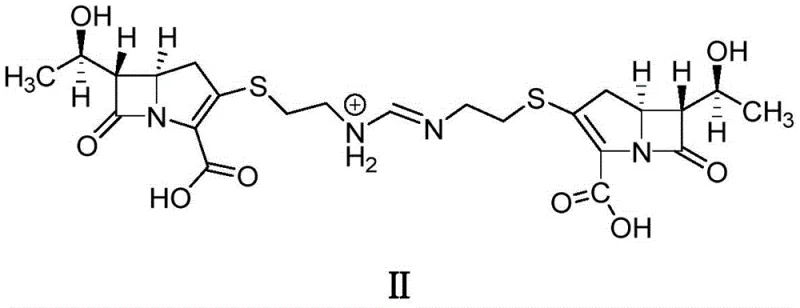
The presence of Impurity II, depicted in the structural analysis above, serves as a critical marker for process inefficiency in legacy methods. Its removal often requires multiple recrystallization cycles or expensive preparative HPLC, driving up the cost of goods sold (COGS) and extending lead times. Additionally, alternative purification strategies involving simple water dissolution followed by pH adjustment with hydrochloric acid have proven inadequate. Research indicates that such acidic conditions promote the degradation of imipenem into Thienamycin (Impurity III) and other unknown degradants, while strongly alkaline environments can lead to ring-opening reactions forming Impurity V. This delicate balance makes traditional water-based crystallization a high-risk operation for supply chain continuity.
The Novel Approach
In stark contrast to these legacy techniques, the novel approach detailed in CN112142740B leverages a precisely controlled ammonium chloride-ammonia buffer system to overcome these stability and purity barriers. By dissolving the crude imipenem in a buffer maintained at a pH of 7.1 to 9.0, preferably around 8.5, the process creates a 'sweet spot' where the drug substance is highly soluble yet chemically stable. This specific ionic environment prevents the acid-catalyzed hydrolysis that leads to Impurity III and inhibits the base-catalyzed ring opening responsible for Impurity V. Moreover, the buffer system facilitates the effective exclusion of Impurity II during the subsequent crystallization step, allowing for the isolation of high-purity crystals in a single, streamlined operation. This method eliminates the need for harsh pH swings and complex chromatographic columns, offering a direct route to clinical-grade material.
Mechanistic Insights into Buffer-Stabilized Crystallization
The success of this purification strategy is rooted in the fundamental chemical interactions between the imipenem zwitterion and the ammonium chloride-ammonia buffer components. Imipenem possesses both acidic and basic functional groups, making its solubility and stability highly dependent on the protonation state of the molecule. In the patented buffer system, the ammonia acts as a weak base to neutralize excess protons, while the ammonium ion provides a common ion effect that can be tuned to control solubility. This buffering capacity ensures that local pH gradients, which often occur during the addition of acids or bases in unbuffered systems, are instantly neutralized. Consequently, the imipenem molecules remain in their most stable ionization state, minimizing the kinetic pathways available for degradation reactions such as beta-lactam ring hydrolysis.
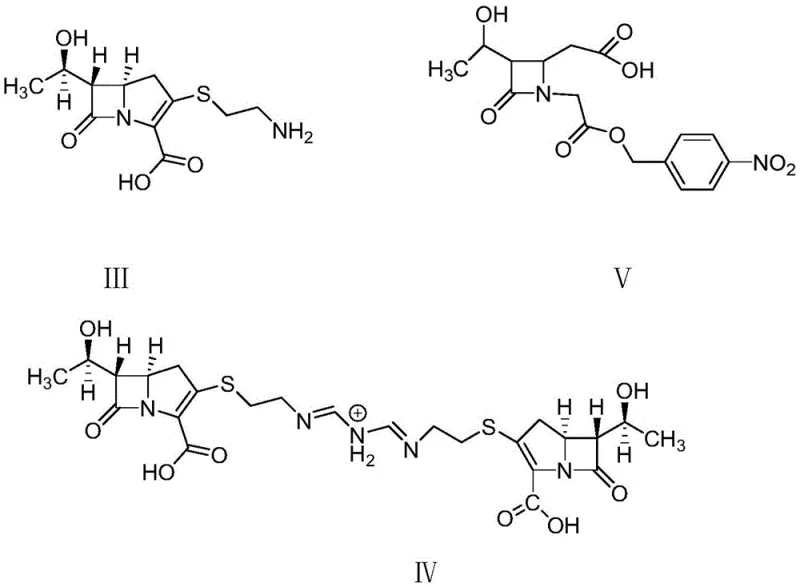
Furthermore, the mechanism extends to the selective crystallization of the target monohydrate over potential impurities. The presence of ammonium ions in the solution likely modifies the solvation shell around the imipenem molecules, altering the nucleation and growth kinetics in a way that favors the exclusion of the dimeric Impurity II and the open-ring Impurity V. As illustrated by the degradation pathways in the figure above, avoiding the extreme pH conditions that generate Impurities III, IV, and V is paramount. The buffer acts as a chemical shield, maintaining the solution environment within a narrow window where these degradation pathways are kinetically inhibited. This mechanistic understanding allows process engineers to confidently scale the reaction, knowing that the buffer capacity will protect the product integrity even in large-volume reactors where mixing efficiency might vary.
How to Synthesize High-Purity Imipenem Efficiently
Implementing this advanced purification protocol requires precise control over temperature, pH, and solvent ratios, but the operational steps are fundamentally simpler than traditional chromatographic methods. The process begins with the dissolution of the crude material in the pre-heated buffer solution, followed by a decolorization step using activated carbon to remove trace organic contaminants. The critical phase involves the controlled addition of an organic anti-solvent, such as acetone, to the cooled filtrate. This induces supersaturation in a controlled manner, prompting the formation of well-defined imipenem monohydrate crystals while leaving soluble impurities in the mother liquor. For a detailed breakdown of the specific parameters and operational sequence required to replicate this high-yield process, please refer to the standardized synthesis guide below.
- Dissolve crude imipenem in an ammonium chloride-ammonia water inorganic buffer solution (pH 7.1-9.0) at 35-70°C to ensure complete solubility.
- Add activated carbon to the solution, cool to 0-20°C, stir for 0.5-1.0 hour, and filter to obtain a clear filtrate free of particulate matter.
- Induce crystallization by adding an organic anti-solvent (such as acetone) to the filtrate at 0-5°C, followed by filtration and vacuum drying to isolate pure imipenem monohydrate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this buffer-based purification technology translates into tangible strategic advantages beyond mere technical specifications. The elimination of column chromatography and freeze-drying steps represents a drastic simplification of the manufacturing workflow, directly impacting the cost structure and throughput capabilities of the production facility. By removing these bottlenecks, manufacturers can achieve significantly faster cycle times and reduce the dependency on specialized, high-maintenance equipment. This streamlining of the process enhances the overall agility of the supply chain, allowing for more responsive production scheduling and better alignment with market demand fluctuations for critical antibiotics.
- Cost Reduction in Manufacturing: The transition from chromatographic purification to a direct crystallization process driven by a simple buffer system results in substantial cost savings. The removal of expensive stationary phases, high-pressure pumping systems, and energy-intensive freeze-dryers lowers the capital expenditure (CAPEX) and operational expenditure (OPEX) associated with API production. Furthermore, the high recovery yield inherent in this method minimizes raw material waste, ensuring that a greater proportion of the input crude product is converted into saleable high-purity inventory. This efficiency gain is critical for maintaining competitive pricing in the generic pharmaceutical market without sacrificing margin.
- Enhanced Supply Chain Reliability: The robustness of the ammonium chloride-ammonia buffer system contributes to superior batch consistency, a key factor in supply chain reliability. Unlike sensitive pH-adjustment methods that are prone to operator error and batch failure, this buffered approach provides a wider operating window that tolerates minor variations in processing conditions. This resilience reduces the risk of out-of-specification (OOS) batches and the associated delays in release testing. Consequently, partners can rely on a more predictable supply of high-quality imipenem, mitigating the risks of stockouts and ensuring continuous availability for downstream formulation into life-saving composite preparations.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method offers a greener alternative to traditional refining. The reduction in solvent usage, particularly the avoidance of large volumes of organic solvents required for chromatography elution, aligns with modern green chemistry principles and reduces the burden on waste treatment facilities. The process is inherently scalable, moving seamlessly from pilot plant to commercial production scales of hundreds of kilograms or more without the need for complex re-engineering. This scalability ensures that the supply can grow in tandem with global health needs, while the simplified waste profile facilitates easier regulatory compliance and environmental permitting.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this patented technology, we have compiled answers to the most pertinent questions raised by industry stakeholders. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, highlighting the specific advantages over prior art methods. Understanding these nuances is essential for making informed decisions regarding technology transfer and long-term sourcing strategies.
Q: Why is the ammonium chloride-ammonia buffer system critical for imipenem purification?
A: The buffer system maintains a stable pH between 7.1 and 9.0, preventing the acidic degradation that forms Thienamycin (Impurity III) and the alkaline degradation that forms open-loop Impurity V, while effectively solubilizing the crude product.
Q: How does this process address the issue of Impurity II (Dimer)?
A: Unlike traditional water-based or acid-adjusted methods where Impurity II persists, the specific ionic strength and pH control of the ammonium chloride-ammonia buffer facilitate the effective separation and removal of this difficult-to-remove dimer during the crystallization phase.
Q: Is this purification method suitable for large-scale industrial production?
A: Yes, the method eliminates the need for complex column chromatography and freeze-drying, utilizing standard crystallization and filtration equipment which significantly simplifies operations and reduces production costs for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imipenem Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex antibiotics like imipenem requires more than just a patent; it demands a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical instrumentation. Our dedication to quality assurance guarantees that every batch of imipenem supplied adheres to the highest international standards, providing peace of mind to our global clientele.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this advanced purification technology for their supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our optimized manufacturing processes can drive value and reliability for your critical antibiotic portfolios.
