Advanced Industrial Synthesis of Imipenem: Enhancing Purity and Scalability for Global API Markets
Advanced Industrial Synthesis of Imipenem: Enhancing Purity and Scalability for Global API Markets
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical antibiotics, and the production of Imipenem, a broad-spectrum carbapenem antibiotic, remains a focal point for process innovation. Patent CN101921275B discloses a groundbreaking industrial method for preparing Imipenem that addresses longstanding challenges in yield, purity, and operational complexity. This technical disclosure outlines a streamlined synthetic route starting from the key intermediate 4AA, leveraging advanced catalytic systems and a unique solvent clathrate isolation strategy. By integrating a Reformatsky reaction, rhodium-catalyzed carbene insertion, and a direct hydrogenation protocol, this methodology offers a compelling value proposition for reliable pharmaceutical intermediate suppliers aiming to optimize their production lines. The process achieves a liquid phase purity of Imipenem exceeding 93%, facilitating straightforward crystallization of the monohydrate form, which is essential for meeting stringent global pharmacopoeia standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Imipenem has been plagued by complex multi-step sequences that introduce significant inefficiencies and quality risks. Early routes, such as those disclosed in US4374772, relied on the direct reaction of thienamycin derivatives with formimidates under extremely harsh pH conditions, making large-scale control difficult and hazardous. Subsequent improvements attempted to utilize double-protection strategies involving nitrobenzyl groups; however, these intermediates proved thermally and chemically unstable, often undergoing premature deprotection or degradation during the catalytic hydrogenation step. Furthermore, alternative pathways involving Schiff base protections introduced difficulties in hydrolysis and pH control, leading to inconsistent yields and complicated purification workflows. These conventional approaches typically require multiple isolation and washing steps to remove impurities, which not only increases solvent consumption and waste generation but also results in substantial material loss, thereby inflating the cost of goods sold for the final active pharmaceutical ingredient.
The Novel Approach
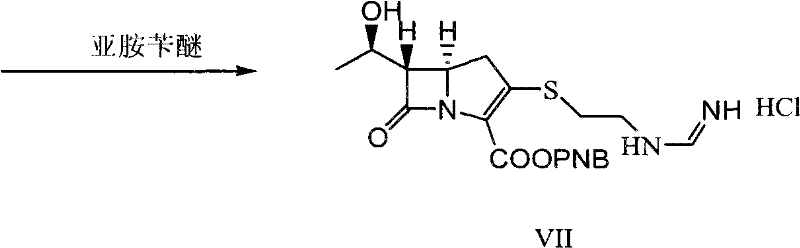
In stark contrast to these legacy methods, the technology described in CN101921275B introduces a highly efficient "one-pot" philosophy for critical transformation steps, drastically reducing the number of unit operations. The core innovation lies in the generation of a solid solvent clathrate compound (Intermediate VI) which serves as a purified precursor for the final coupling and hydrogenation stages. Instead of isolating the unstable phosphorylated intermediate, the process reacts it directly with cysteamine hydrochloride in a tailored solvent system to precipitate the clathrate. This solid intermediate can then be directly subjected to condensation with benzyl formimidate without the need for solvent removal or extensive washing, preserving the integrity of the molecule. This approach eliminates the instability issues associated with double-protected species and streamlines the workflow from the bicyclic nucleus to the final API.
Mechanistic Insights into Rhodium-Catalyzed Cyclization and Clathrate Formation
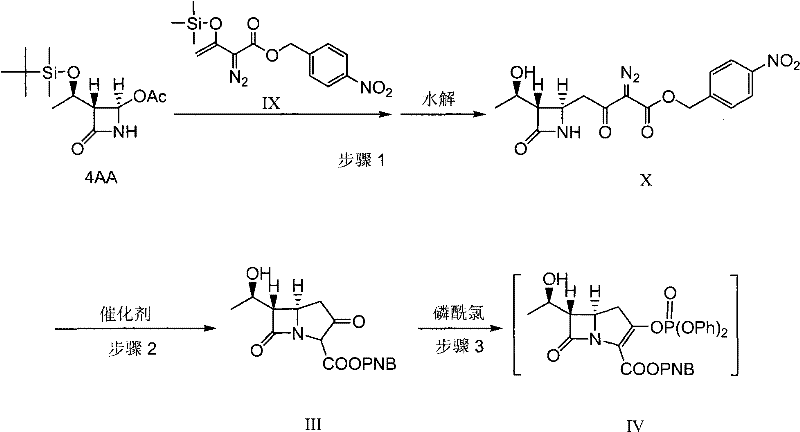
The success of this synthetic route hinges on the precise control of two critical mechanistic events: the formation of the carbapenem bicyclic nucleus and the stabilization of the side-chain intermediate. The construction of the bicyclic parent nucleus (Intermediate III) is achieved via an N-H carbene insertion reaction, a transformation that demands high selectivity to avoid ring-opening or polymerization by-products. The patent specifies the use of rhodium catalysts, such as rhodium acetate or rhodium caproate, potentially promoted by Lewis acids like zinc bromide or cuprous chloride. This catalytic system facilitates the intramolecular insertion of the carbene moiety into the nitrogen-hydrogen bond with exceptional efficiency, operating effectively at temperatures between 30°C and 100°C. The choice of solvent, typically esters like ethyl acetate or chlorinated hydrocarbons, further modulates the reaction kinetics to favor the desired bicyclic structure over linear oligomers.
Following the formation of the nucleus, the process employs a sophisticated phosphorylation and thiol-addition sequence to install the critical side chain. Rather than isolating the reactive phosphate ester (Intermediate IV), the protocol dictates a direct reaction with cysteamine hydrochloride in the presence of an organic base. The brilliance of this step lies in the solvent engineering; by utilizing a mixture of pyrrolidinone compounds (like N-methylpyrrolidone) and nitriles (like acetonitrile), the resulting amino-thiol intermediate spontaneously forms a stable solid solvent clathrate (Intermediate VI). This clathrate formation acts as an in-situ purification mechanism, trapping the desired product in a crystalline lattice while excluding soluble impurities. This mechanistic feature ensures that the subsequent condensation with benzyl formimidate proceeds with high fidelity, as the starting material for this step is already of elevated purity, minimizing the burden on the final catalytic hydrogenation.
How to Synthesize Imipenem Efficiently
Implementing this synthesis requires careful attention to reaction conditions, particularly regarding temperature control and solvent ratios during the clathrate formation stage. The process begins with the preparation of the diazo intermediate X, followed by the cyclization to nucleus III. Once the nucleus is secured, the phosphorylation must be conducted at low temperatures (-70°C to 0°C) to prevent decomposition, followed by the controlled addition of the thiol component. The precipitation of the clathrate is driven by the addition of anti-solvents like acetonitrile or ethyl acetate at specific temperatures. For a comprehensive understanding of the operational parameters, reagent grades, and work-up procedures required to replicate this high-yield pathway, please refer to the standardized synthesis guide below.
- Perform a Reformatsky reaction between compound 4AA and intermediate IX using a zinc halide catalyst, followed by hydrolysis to obtain intermediate X.
- Conduct an N-H carbene insertion reaction on intermediate X using a rhodium or Lewis acid catalyst to form the bicyclic parent nucleus III.
- Phosphorylate nucleus III and react directly with cysteamine hydrochloride in a specific solvent system to precipitate the solid solvent clathrate compound VI without isolation of the phosphate intermediate.
- Condense the solvent clathrate VI with benzyl formimidate to form intermediate VII, followed by direct catalytic hydrogenation to yield Imipenem solution.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible operational efficiencies and risk mitigation. The elimination of intermediate isolation steps, particularly the avoidance of washing and drying the solvent clathrate before the next reaction, significantly reduces the cycle time per batch. This reduction in processing time allows for higher throughput within existing reactor capacity, effectively increasing plant utilization rates without the need for capital expenditure on new equipment. Furthermore, the simplified workflow reduces the consumption of auxiliary materials such as filtration media and washing solvents, contributing to a leaner manufacturing cost structure. The robustness of the rhodium-catalyzed cyclization also ensures consistent batch-to-batch quality, reducing the incidence of out-of-specification products that can disrupt supply schedules.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by removing the need to isolate and purify the unstable phosphorylated intermediate IV. By proceeding directly to the clathrate formation, the method saves on solvent volumes and energy costs associated with evaporation and drying. Additionally, the high molar yields reported in the initial steps (exceeding 80% for intermediate X) ensure that expensive starting materials like compound 4AA are utilized with maximum efficiency, minimizing raw material waste and lowering the overall cost of goods for the final API.
- Enhanced Supply Chain Reliability: The reliance on commercially available catalysts such as zinc halides and rhodium salts, alongside common industrial solvents like toluene, ethyl acetate, and acetonitrile, ensures that the supply chain is not vulnerable to shortages of exotic reagents. The ability to produce a high-purity Imipenem solution directly from the clathrate without complex purification trains means that production timelines are more predictable. This predictability is crucial for maintaining continuous supply to downstream formulation partners, especially in the event of surging demand for broad-spectrum antibiotics.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the reduction in solvent usage and the minimization of waste streams align with green chemistry principles. The process avoids the generation of large volumes of acidic or basic wash waters typically associated with traditional protection-deprotection sequences. The direct hydrogenation step, performed in a buffered system, further simplifies waste treatment protocols. These factors make the technology highly scalable from pilot plants to multi-ton commercial production facilities while maintaining compliance with increasingly stringent environmental regulations regarding solvent emissions and effluent discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this Imipenem synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms historical precedents in terms of purity and operational simplicity.
Q: What is the primary advantage of using the solvent clathrate intermediate VI in Imipenem synthesis?
A: The formation of the solid solvent clathrate compound VI allows for the effective removal of impurities generated during the phosphorylation and thiol addition steps. Unlike traditional methods that require complex solvent washing and drying which can degrade yield, this method enables the clathrate to be used directly in the subsequent condensation reaction, significantly simplifying the workflow and improving overall product purity.
Q: Which catalysts are critical for the formation of the bicyclic parent nucleus in this process?
A: The process utilizes a highly efficient catalytic system for the N-H carbene insertion step. Preferred catalysts include rhodium-based compounds such as rhodium acetate or rhodium caproate, often used in conjunction with Lewis acid promoters like zinc bromide or cuprous chloride. This combination ensures high conversion rates and minimizes the formation of by-products compared to older copper-only systems.
Q: How does this patent address the stability issues associated with double-protected Imipenem intermediates?
A: Traditional routes often suffer from the instability of nitrobenzyl-protected side chains, leading to the loss of the formimino group during hydrogenation. This novel method avoids the formation of unstable double-protected intermediates by generating the formimino group in the final stages via condensation with benzyl formimidate, followed immediately by hydrogenation. This sequence preserves the structural integrity of the sensitive beta-lactam ring and side chain.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imipenem Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate details of the solvent clathrate technology and rhodium-catalyzed steps are executed with precision. We are committed to delivering high-purity Imipenem intermediates and APIs that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our facility is equipped to handle the specific solvent systems and low-temperature reactions required by this patent, guaranteeing consistent quality and supply continuity for our partners.
We invite potential collaborators to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential efficiencies of this method for your specific volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and reliability in your antibiotic manufacturing operations.
