Revolutionizing L-p-Hydroxyphenylglycine Production: A Streamlined 6-Step Process for High-Purity Antibiotic Intermediates
The pharmaceutical industry constantly seeks more efficient pathways for synthesizing critical antibiotic intermediates, and Patent CN110467537B represents a significant leap forward in the production of L-p-hydroxyphenylglycine (DHPG). This vital chiral building block is indispensable for the synthesis of broad-spectrum beta-lactam antibiotics such as amoxicillin, cefadroxil, and cefoperazone. Traditionally, the manufacturing landscape for DHPG has been plagued by lengthy, multi-step processes that involve the isolation of solid racemic intermediates, leading to excessive solvent usage, high energy consumption, and suboptimal yields. The disclosed invention fundamentally restructures this workflow by introducing a novel composite catalyst system and a direct resolution strategy that bypasses the need for solid isolation of the racemate. By transitioning from a conventional nine-step protocol to a streamlined six-step sequence, this technology not only enhances the stereochemical purity of the final product but also delivers substantial operational efficiencies. For global procurement and supply chain leaders, this patent signals a potential paradigm shift towards more sustainable and cost-effective manufacturing of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of L-p-hydroxyphenylglycine has relied on cumbersome methodologies that prioritize isolation over integration. Conventional routes typically involve the initial synthesis of racemic p-hydroxyphenylglycine sulfate, followed immediately by a hydrolysis step using ammonia water to precipitate the solid racemic amino acid. This solid must then undergo rigorous washing with large volumes of water or organic solvents to remove impurities, generating significant wastewater streams that require expensive treatment. Subsequently, this dry solid must be redissolved in acid before the critical asymmetric resolution can even begin. This disjointed approach creates multiple points of material loss, extends the production cycle unnecessarily, and inflates the cost basis due to the energy-intensive drying and redissolution phases. Furthermore, the repeated phase transitions from liquid to solid and back to liquid increase the risk of mechanical losses and contamination, ultimately capping the overall yield and compromising the economic viability of large-scale production.
The Novel Approach
In stark contrast, the methodology outlined in Patent CN110467537B introduces a continuous liquid-phase strategy that maintains the reaction mixture in solution until the final crystallization of the target enantiomer. The core innovation lies in the decision to forego the isolation of the solid racemic p-hydroxyphenylglycine entirely. Instead, the synthesized sulfate solution is purified and directly subjected to asymmetric resolution using a chiral resolving agent. This bold simplification eliminates three distinct unit operations: the initial hydrolysis to solid, the solid-liquid separation, and the subsequent acid dissolution. By maintaining the intermediate in the sulfate form, the process leverages the solubility characteristics of the complex salt to drive the resolution efficiently. This integrated approach not only compresses the timeline drastically but also minimizes the physical handling of materials, thereby reducing labor costs and exposure to potential contaminants while maximizing the throughput of the manufacturing facility.
Mechanistic Insights into Composite Catalysis and Direct Resolution
The chemical foundation of this improved process rests on the deployment of a sophisticated composite catalyst during the initial condensation of phenol and glyoxylic acid. The patent specifies a synergistic blend of octadecyl benzyl ammonium chloride and sodium succinate, utilized in a precise ratio ranging from 1:1 to 1:3. This dual-component system acts to steer the electrophilic substitution reaction with high regioselectivity, favoring the formation of the para-isomer over the thermodynamically competitive ortho- and meta-isomers. The quaternary ammonium salt likely functions as a phase-transfer catalyst, enhancing the interaction between the organic phenol and the aqueous acidic medium, while the sodium succinate may act as a buffer or co-catalyst to stabilize the transition state. This precise control over reaction orientation results in a crude sulfate solution with a significantly higher content of the desired DL-p-hydroxyphenylglycine, reducing the burden on downstream purification steps.
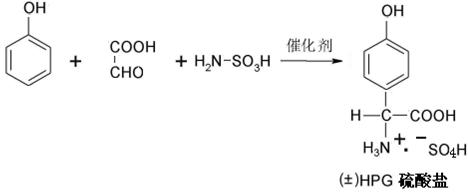
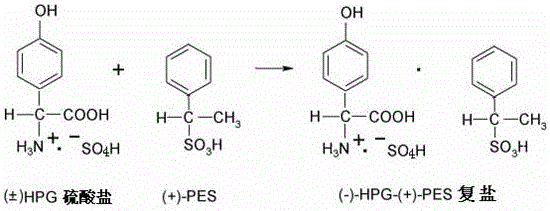
Following the synthesis, the process moves directly to the chiral resolution phase without isolating the racemate. The purified sulfate solution is treated with D-phenylethanesulfonic acid ((+)-PES), which acts as the resolving agent. The mechanism relies on the differential solubility of the diastereomeric salts formed in the reaction medium. The L-enantiomer of the amino acid preferentially complexes with the D-resolving agent to form the less soluble (-)-HPG-(+)-PES complex salt, which precipitates out of the solution. This direct precipitation from the sulfate matrix is a critical engineering feat, as it avoids the dilution effects and solubility hurdles associated with redissolving a solid cake. The reaction is conducted at elevated temperatures, typically between 40°C and 90°C, to ensure kinetic control and optimal crystal growth of the desired diastereomer.
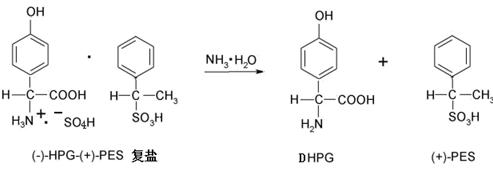
The final liberation of the free amino acid is achieved through a controlled hydrolysis of the isolated complex salt. Unlike the harsh conditions often required to break down stable salts, this process utilizes a mild alkali liquor, such as sodium hydroxide, potassium hydroxide, or ammonia water, added dropwise at temperatures between 20°C and 60°C. The pH is meticulously controlled to a range of 4.5 to 5.5, which corresponds to the isoelectric point of the amino acid, inducing crystallization of the pure L-p-hydroxyphenylglycine. This gentle hydrolysis ensures that the delicate chiral center remains intact, preventing racemization and preserving the high optical purity required for antibiotic synthesis. The mother liquor, containing the resolving agent, is subsequently recycled, closing the loop on one of the most expensive reagents in the process and further enhancing the atom economy of the entire operation.
How to Synthesize L-p-Hydroxyphenylglycine Efficiently
The implementation of this streamlined synthesis route requires precise adherence to the optimized parameters defined in the patent to achieve the reported yield and purity improvements. The process begins with the condensation reaction under the influence of the composite catalyst, followed by a crucial decolorization step using activated carbon in a water-alcohol mixture to remove trace organic impurities that could interfere with resolution. The subsequent addition of the resolving agent must be timed and dosed accurately to maximize the precipitation of the target diastereomer. For R&D teams looking to replicate or scale this technology, the detailed standard operating procedures regarding temperature ramps, stirring rates, and pH control are critical for success. The following guide outlines the essential workflow derived from the patent examples to ensure reproducible high-quality output.
- Synthesize racemic p-hydroxyphenylglycine sulfate using phenol, glyoxylic acid, and sulfamic acid with a composite catalyst.
- Purify the sulfate solution via decolorization with activated carbon and a water-alcohol mixture.
- Perform asymmetric resolution directly on the solution using phenylethanesulfonic acid to form the complex salt.
- Separate the solid complex salt via centrifugation and hydrolyze with alkali to obtain the final L-p-hydroxyphenylglycine crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates into tangible strategic advantages beyond mere technical elegance. The reduction of the production cycle by approximately 12 hours per batch implies a significant increase in asset turnover, allowing manufacturing facilities to produce more tons of product per annum without capital expansion. This acceleration in throughput directly addresses the volatility often seen in the supply of antibiotic intermediates, providing a more reliable flow of goods to downstream API manufacturers. Furthermore, the elimination of solid isolation steps drastically reduces the consumption of process water and the generation of wastewater, aligning production with increasingly stringent environmental regulations and lowering the overhead costs associated with waste treatment and disposal.
- Cost Reduction in Manufacturing: The economic impact of removing three unit operations is profound, as it eliminates the energy costs associated with drying solid cakes and the labor costs of multiple filtration and redissolution cycles. By processing the intermediate directly in the liquid phase, the facility saves on utility consumption and reduces the wear and tear on equipment such as centrifuges and dryers. Additionally, the recycling of the phenylethanesulfonic acid resolving agent from the mother liquor significantly lowers the raw material cost per kilogram of finished product, creating a leaner cost structure that can withstand market fluctuations.
- Enhanced Supply Chain Reliability: The simplified workflow reduces the number of potential failure points in the manufacturing chain, thereby increasing the overall reliability of supply. With fewer steps involving solid handling and transfer, the risk of batch contamination or mechanical loss is minimized, leading to more consistent batch-to-batch quality. This stability is crucial for long-term supply agreements with major pharmaceutical companies, where consistency and on-time delivery are paramount. The ability to shorten the lead time for high-purity pharmaceutical intermediates ensures that downstream antibiotic production schedules can be met with greater confidence.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, as the liquid-phase operations are easier to manage in large reactors compared to the handling of massive quantities of wet solids. The reported reduction in water consumption by over 15% demonstrates a commitment to sustainable manufacturing practices, which is increasingly becoming a prerequisite for vendor qualification in the global pharmaceutical market. This eco-friendly profile not only reduces the environmental footprint but also mitigates the regulatory risks associated with wastewater discharge limits, ensuring uninterrupted operation even in regions with strict environmental enforcement.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis route. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this technology into their existing supply chains. The answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a factual basis for decision-making.
Q: How does the new composite catalyst improve the synthesis of DL-p-hydroxyphenylglycine?
A: The patent utilizes a composite catalyst of octadecyl benzyl ammonium chloride and sodium succinate. This specific combination enhances reaction orientation, significantly reducing ortho- and meta-position byproducts and increasing the para-isomer content by 1.0-1.5%.
Q: What are the primary efficiency gains of skipping the solid isolation step?
A: By resolving the racemic mixture directly from the sulfate solution rather than isolating the solid first, the process eliminates three operational steps (hydrolysis, solid-liquid separation, and redissolution). This reduces the production cycle by approximately 12 hours and cuts water consumption by 15-16%.
Q: What resolving agent is used in this optimized process?
A: The process employs phenylethanesulfonic acid (specifically the D-enantiomer) as the resolving agent to form a levo-p-hydroxyphenylglycine-phenylethanesulfonic acid complex salt, which is then hydrolyzed to release the pure L-enantiomer.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-p-Hydroxyphenylglycine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the streamlined synthesis process described in Patent CN110467537B for the global antibiotic market. As a premier CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this 6-step process are fully realized in a GMP-compliant environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of L-p-hydroxyphenylglycine meets the exacting standards required for beta-lactam antibiotic synthesis, providing our partners with peace of mind regarding quality and regulatory compliance.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cost-efficient technology for their supply chains. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to quantify the operational savings and supply security that this innovative manufacturing process can bring to your organization.
