Industrial Scale Menthone Production: Overcoming Conventional Limitations with Novel Heterogeneous Catalysis
The global demand for high-purity terpenes continues to drive innovation in synthetic methodology, particularly for key aroma compounds like menthone. Patent CN109704944B introduces a transformative approach to synthesizing menthone directly from citronellal, bypassing the inefficiencies of legacy oxidation technologies. This technical breakthrough leverages a sophisticated heterogeneous catalyst system comprising ruthenium and zinc supported on alumina, combined with a trace auxiliary agent to facilitate intramolecular cyclization. For R&D directors and procurement specialists, this represents a pivotal shift away from toxic stoichiometric oxidants toward a safer, more sustainable catalytic cycle. The reported conversion rates exceeding 90% and chemical selectivity above 96% underscore the viability of this route for commercial adoption. By addressing the critical pain points of waste generation and catalyst recovery, this technology offers a compelling value proposition for manufacturers seeking to optimize their supply chain resilience and environmental compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of menthone has relied heavily on the oxidation of menthol or the hydrogenation of thymol, processes fraught with significant operational and environmental challenges. Traditional oxidation routes often employ stoichiometric amounts of hazardous reagents such as chromates, permanganates, or hypochlorites, which generate substantial quantities of heavy metal waste and byproduct salts. Furthermore, alternative pathways involving the hydrogenation of isopulegol frequently suffer from low turnover numbers (TON) and short catalyst lifespans, necessitating frequent catalyst replacement and driving up operational costs. Some existing methods require complex multi-step sequences, such as hydrogenating isopulegol to menthol followed by a separate oxidation step, which introduces safety risks associated with switching between hydrogen and oxygen atmospheres. These conventional methodologies not only inflate production costs through expensive raw materials and waste treatment but also complicate the purification process, making it difficult to achieve the stringent purity specifications required by top-tier flavor and fragrance houses.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the novel method disclosed in the patent utilizes a direct intramolecular cyclization of citronellal driven by a robust heterogeneous catalyst system. This approach streamlines the synthesis into a single continuous step, eliminating the need for intermediate isolation and the handling of dangerous oxidizing agents. The core of this innovation lies in the use of a fixed-bed reactor loaded with a bimetallic Ru-Zn catalyst, which facilitates the reaction under mild conditions while maintaining exceptional stability. By operating in a downflow mode with quantitative feeding, the process ensures consistent product quality and minimizes downtime associated with batch processing. This technological leap not only simplifies the plant layout but also drastically reduces the environmental footprint by avoiding the generation of volatile organic solvents and toxic salt waste. The ability to achieve high conversion and selectivity in a continuous flow setup marks a significant advancement in the commercial scale-up of complex flavor intermediates.
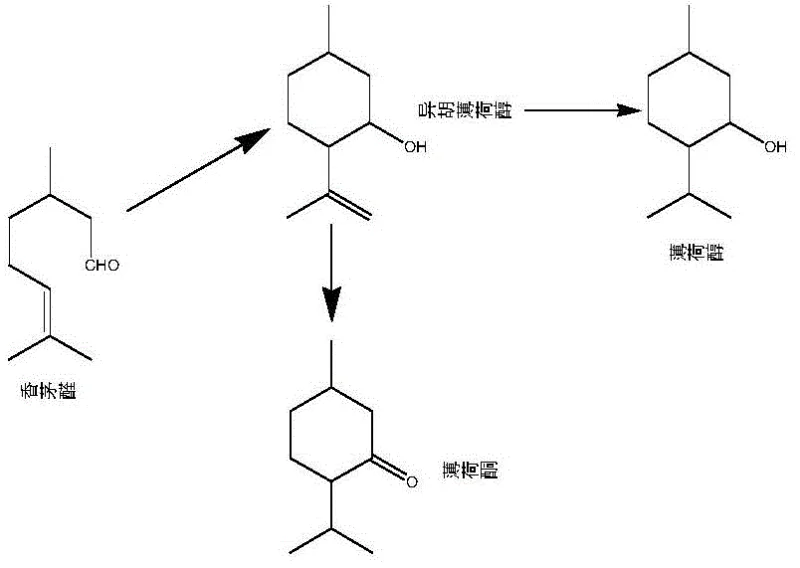
Mechanistic Insights into Ru-Zn Catalyzed Cyclization
The efficacy of this synthesis route is rooted in the synergistic interaction between the ruthenium and zinc metal centers dispersed on the alumina support. The ruthenium component acts as the primary active site for the activation of the carbon-carbon double bond in citronellal, promoting the necessary electronic rearrangement for cyclization. Meanwhile, the zinc promoter modifies the electronic environment of the ruthenium, enhancing its Lewis acidity and stabilizing the transition state during the ring-closing step. This bimetallic cooperation is critical for suppressing competing side reactions, such as over-hydrogenation or polymerization, which typically plague single-metal catalyst systems. The heterogeneous nature of the catalyst ensures that the active sites remain accessible throughout the reaction duration, preventing the leaching of metals into the product stream and thereby safeguarding the purity of the final menthone. This mechanistic precision allows the process to maintain high performance even under the rigorous demands of continuous industrial operation.
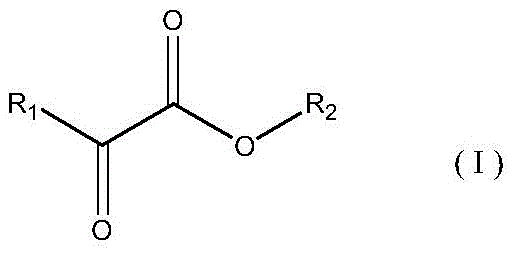
Furthermore, the inclusion of a specific auxiliary agent, typically an alpha-keto ester like methyl pyruvate, plays a pivotal role in fine-tuning the reaction selectivity. Although used in trace amounts ranging from 0.01% to 1% by mass, this additive interacts with the catalyst surface to create a microenvironment that favors the formation of the desired ketone structure. The auxiliary agent likely coordinates with the metal centers to modulate the adsorption strength of the reactant, ensuring that the cyclization proceeds with minimal formation of isomeric byproducts. This subtle yet powerful modification of the catalytic landscape is what enables the system to achieve chemical selectivity figures exceeding 96%. Understanding this mechanism is vital for process engineers aiming to replicate these results, as the precise control of auxiliary dosage and catalyst pretreatment conditions determines the overall success of the synthesis.
How to Synthesize Menthone Efficiently
Implementing this advanced synthesis route requires careful attention to catalyst preparation and reactor configuration to fully realize its commercial potential. The process begins with the co-precipitation of ruthenium and zinc salts onto an alumina carrier, followed by a controlled calcination and reduction sequence to activate the metal sites. Once the fixed-bed reactor is charged with the reduced catalyst, the system operates by continuously pumping a mixture of citronellal and the auxiliary agent through the heated catalyst bed. Detailed standard operating procedures regarding temperature gradients, flow rates, and catalyst regeneration cycles are essential for maintaining long-term stability. The following guide outlines the critical standardized synthesis steps required to transition this laboratory-scale innovation into a robust manufacturing protocol.
- Preparation of the bimetallic Ru-Zn/Al2O3 heterogeneous catalyst via co-precipitation and calcination at 400-600°C.
- Reduction of the catalyst precursor in a hydrogen stream at 150-200°C to activate the metal sites.
- Continuous flow reaction of citronellal with a trace auxiliary agent over the fixed bed catalyst at 100-120°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this heterogeneous catalytic process offers profound strategic advantages beyond mere technical performance. The elimination of stoichiometric oxidants and the shift to a continuous flow regime fundamentally alter the cost structure of menthone manufacturing. By removing the need for expensive and hazardous reagents, companies can significantly reduce raw material expenditures and lower the costs associated with waste disposal and regulatory compliance. The robustness of the fixed-bed catalyst system also implies longer operational cycles with less frequent shutdowns for catalyst replacement, directly translating to improved asset utilization and production throughput. These factors combine to create a more resilient supply chain capable of meeting fluctuating market demands without the volatility associated with batch-based chemical synthesis.
- Cost Reduction in Manufacturing: The transition to a heterogeneous catalyst system eliminates the recurring cost of purchasing stoichiometric oxidants and the subsequent expense of treating heavy metal waste streams. By utilizing a fixed-bed reactor, the process minimizes solvent consumption and energy usage compared to traditional batch oxidation methods. The high selectivity of the reaction reduces the burden on downstream purification units, lowering the overall cost of goods sold. Furthermore, the extended lifespan of the supported catalyst reduces the frequency of catalyst procurement, providing substantial long-term savings.
- Enhanced Supply Chain Reliability: Continuous flow processing inherently offers greater predictability and consistency than batch operations, ensuring a steady output of high-purity product. The use of readily available raw materials like citronellal, combined with a stable catalyst system, mitigates the risk of supply disruptions caused by reagent shortages. The simplified process flow reduces the number of unit operations, decreasing the likelihood of mechanical failures or operational bottlenecks. This reliability is crucial for maintaining just-in-time delivery schedules with major international clients in the flavor and pharmaceutical sectors.
- Scalability and Environmental Compliance: The design of this process aligns perfectly with modern green chemistry principles, facilitating easier permitting and regulatory approval in stringent jurisdictions. The absence of toxic chromium or manganese waste simplifies effluent treatment and reduces the environmental liability of the manufacturing site. Scalability is inherent to the fixed-bed design, allowing production capacity to be increased by numbering up reactors rather than building massive new vessels. This modular approach supports rapid expansion to meet growing global demand while maintaining a minimal environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and process descriptions found in the patent documentation. They are intended to clarify the operational parameters and benefits for stakeholders evaluating this synthesis route for potential licensing or production partnerships. Understanding these details is essential for making informed decisions about integrating this technology into existing manufacturing portfolios.
Q: What distinguishes this menthone synthesis method from traditional oxidation routes?
A: Unlike traditional methods that rely on toxic stoichiometric oxidants like chromates or complex multi-step hydrogenation-oxidation sequences, this patent utilizes a direct intramolecular cyclization of citronellal. This approach eliminates hazardous waste streams and simplifies the process flow significantly.
Q: How does the auxiliary agent improve reaction selectivity?
A: The addition of specific alpha-keto ester auxiliary agents, such as methyl pyruvate, interacts with the heterogeneous catalyst surface to enhance chemical selectivity. This interaction suppresses side reactions, ensuring conversion rates exceed 90% with selectivity above 96%.
Q: Is this process suitable for large-scale continuous manufacturing?
A: Yes, the technology is explicitly designed for fixed-bed reactor operations. The robustness of the heterogeneous catalyst allows for continuous quantitative feeding, making it highly scalable for industrial production compared to batch-wise homogeneous catalysis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Menthone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced catalytic technology for the global flavor and fragrance market. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovation to life. Our facilities are equipped with state-of-the-art fixed-bed reactor systems and rigorous QC labs capable of meeting stringent purity specifications for high-value terpenes. We are committed to leveraging our technical expertise to optimize this synthesis route, ensuring that our clients receive a consistent, high-quality supply of menthone that meets the highest industry standards.
We invite forward-thinking organizations to collaborate with us to unlock the full commercial value of this efficient synthesis method. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this technology can enhance your supply chain efficiency. Let us help you navigate the complexities of modern chemical manufacturing and secure a competitive advantage in the marketplace.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
