Advanced Synthesis of Indazole-Triazole Derivatives for Targeted Prostate Cancer Therapy
Advanced Synthesis of Indazole-Triazole Derivatives for Targeted Prostate Cancer Therapy
The pharmaceutical landscape for oncology treatments is constantly evolving, driven by the urgent need for agents that offer high specificity and reduced systemic toxicity. Patent CN113999211A introduces a novel class of indazole skeleton derivatives containing a 1,2,3-triazole moiety, specifically designed to combat prostate cancer. These compounds represent a significant breakthrough in medicinal chemistry, demonstrating potent antiproliferative activity against PC-3 prostate cancer cell lines while maintaining a favorable safety profile against normal prostate stromal cells. The structural versatility of this scaffold allows for extensive SAR (Structure-Activity Relationship) exploration, making it a highly attractive candidate for drug development programs focused on male reproductive health. 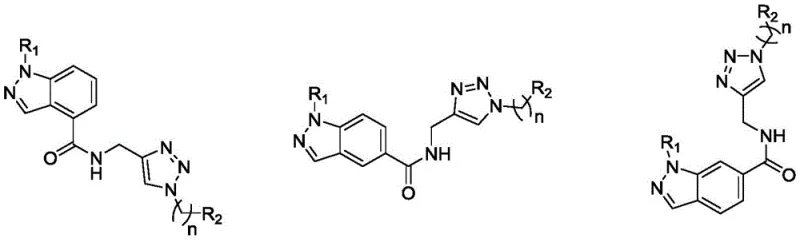 For procurement and supply chain leaders, the significance of this technology lies not just in its biological efficacy but in the robustness of its manufacturing process. The disclosed synthesis pathway utilizes mild reaction conditions and commercially accessible raw materials, which translates directly into potential cost efficiencies and reliable supply continuity. As a reliable pharmaceutical intermediate supplier, understanding the nuances of such patented routes is critical for securing long-term partnerships with innovator companies seeking to bring next-generation anti-tumor agents to clinical trials and eventual market approval.
For procurement and supply chain leaders, the significance of this technology lies not just in its biological efficacy but in the robustness of its manufacturing process. The disclosed synthesis pathway utilizes mild reaction conditions and commercially accessible raw materials, which translates directly into potential cost efficiencies and reliable supply continuity. As a reliable pharmaceutical intermediate supplier, understanding the nuances of such patented routes is critical for securing long-term partnerships with innovator companies seeking to bring next-generation anti-tumor agents to clinical trials and eventual market approval.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing heterocyclic anticancer agents often suffer from significant drawbacks that hinder commercial viability. Many legacy routes rely on harsh reaction conditions, such as extreme temperatures or the use of strong, corrosive acids and bases, which can degrade sensitive functional groups and lead to complex impurity profiles. Furthermore, conventional methods for constructing triazole rings frequently lack regioselectivity, resulting in mixtures of 1,4- and 1,5-disubstituted isomers that require difficult and yield-reducing separation processes. The reliance on expensive transition metal catalysts, such as palladium or rhodium, in older cross-coupling strategies also introduces substantial cost burdens and necessitates rigorous heavy metal removal steps to meet stringent regulatory limits for API intermediates. These factors collectively increase the lead time for high-purity pharmaceutical intermediates and elevate the overall cost of goods sold (COGS).
The Novel Approach
In contrast, the methodology described in CN113999211A offers a streamlined and efficient alternative that addresses these historical pain points. The synthesis employs a modular strategy centered around Copper-Catalyzed Azide-Alkyne Cycloaddition (CuAAC), widely recognized as a premier "click chemistry" reaction. This approach ensures excellent regioselectivity, predominantly yielding the desired 1,4-disubstituted 1,2,3-triazole products without the formation of unwanted isomers. The reaction conditions are remarkably mild, often proceeding at room temperature in benign solvent systems like tetrahydrofuran and water mixtures. This not only enhances operational safety but also simplifies downstream processing and purification. By eliminating the need for precious metal catalysts and avoiding extreme thermal stress, this novel route significantly reduces production costs and environmental impact, aligning perfectly with modern green chemistry principles and sustainable manufacturing goals.
Mechanistic Insights into CuAAC Click Chemistry and Amide Coupling
The core of this synthetic strategy involves a sophisticated yet practical sequence of transformations that build molecular complexity with high precision. The process initiates with the N-alkylation of the indazole core, where methyl 1H-indazole-4-carboxylate reacts with various iodo-compounds in the presence of an inorganic base like cesium carbonate. This step is crucial for introducing diversity at the R1 position, allowing for the tuning of physicochemical properties. Following this, a standard hydrolysis converts the ester to a carboxylic acid, setting the stage for amide bond formation. 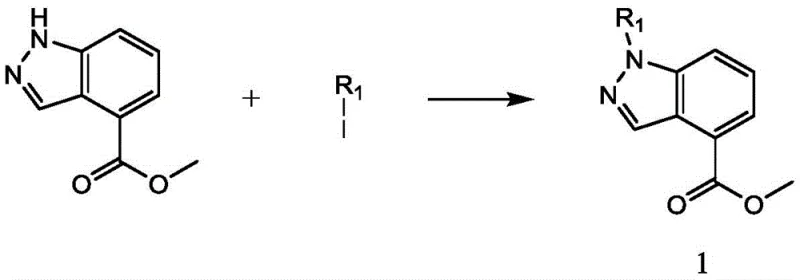 The subsequent amidation with propargylamine introduces the alkyne handle necessary for the final cyclization. This step typically utilizes efficient coupling reagents such as HATU or EDCI/HOBt, ensuring high conversion rates and minimal racemization. The culmination of the synthesis is the CuAAC reaction, where the alkyne-functionalized intermediate reacts with an organic azide. In the presence of a catalytic system comprising anhydrous copper sulfate and sodium ascorbate, a concerted cycloaddition occurs to form the stable 1,2,3-triazole ring. This mechanism is highly tolerant of various functional groups, enabling the incorporation of diverse R2 substituents including halogens, alkyl chains, and electron-withdrawing groups without compromising yield or purity.
The subsequent amidation with propargylamine introduces the alkyne handle necessary for the final cyclization. This step typically utilizes efficient coupling reagents such as HATU or EDCI/HOBt, ensuring high conversion rates and minimal racemization. The culmination of the synthesis is the CuAAC reaction, where the alkyne-functionalized intermediate reacts with an organic azide. In the presence of a catalytic system comprising anhydrous copper sulfate and sodium ascorbate, a concerted cycloaddition occurs to form the stable 1,2,3-triazole ring. This mechanism is highly tolerant of various functional groups, enabling the incorporation of diverse R2 substituents including halogens, alkyl chains, and electron-withdrawing groups without compromising yield or purity.
From an impurity control perspective, this route offers distinct advantages. The high regioselectivity of the click reaction minimizes the generation of structural isomers, which are often the most challenging impurities to remove. Additionally, the use of water-soluble copper salts facilitates easy removal of the catalyst during the aqueous workup phase, reducing the risk of metal contamination in the final product. The intermediates formed at each stage, such as the carboxylic acid and the alkyne-amide, are generally stable solids that can be purified via recrystallization or column chromatography, ensuring that only high-quality material proceeds to the next step. This stepwise control over purity is essential for meeting the rigorous specifications required for clinical-grade pharmaceutical intermediates and supports the development of a robust quality control framework.
How to Synthesize Indazole-Triazole Derivatives Efficiently
The synthesis of these potent anticancer candidates follows a logical four-step progression that balances chemical efficiency with operational simplicity. The process begins with the alkylation of the indazole nitrogen, followed by ester hydrolysis to reveal the reactive acid functionality. This acid is then coupled with propargylamine to install the alkyne moiety, which serves as the dipolarophile for the final cycloaddition. The detailed standardized synthesis steps, including specific molar ratios, solvent choices, and reaction times optimized for scale-up, are outlined below to guide process chemists in replicating this high-yielding route.
- Perform N-alkylation of methyl 1H-indazole-4-carboxylate with an iodo compound using cesium carbonate in acetonitrile under reflux.
- Hydrolyze the resulting ester intermediate using sodium hydroxide in methanol to obtain the corresponding carboxylic acid.
- Couple the carboxylic acid with propargylamine using HATU or EDCI/HOBt condensing agents to form the alkyne-amide intermediate.
- Execute copper-catalyzed azide-alkyne cycloaddition (CuAAC) with organic azides using copper sulfate and sodium ascorbate to finalize the triazole ring formation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route presents compelling economic and logistical benefits. The reliance on commodity chemicals and the avoidance of exotic reagents create a resilient supply chain that is less susceptible to market volatility. The simplicity of the workup procedures, particularly the aqueous extraction steps involved in the click chemistry reaction, reduces solvent consumption and waste disposal costs, contributing to a more sustainable and cost-effective manufacturing process. These factors collectively enhance the commercial attractiveness of partnering with suppliers who have mastered this specific technology.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts like palladium represents a direct saving in raw material costs, which is significant when scaling to multi-kilogram production. Furthermore, the high yields reported in the patent examples, often exceeding 70-80% for key steps, minimize material loss and maximize throughput. The ability to perform reactions at room temperature also reduces energy consumption associated with heating and cooling, leading to lower utility costs. By streamlining the purification process through high-selectivity reactions, the need for extensive chromatographic separation is reduced, further lowering labor and consumable expenses associated with production.
- Enhanced Supply Chain Reliability: The starting materials, including methyl 1H-indazole-4-carboxylate and various substituted anilines or benzyl bromides, are widely available from global chemical vendors, ensuring a stable supply of inputs. The robustness of the reaction conditions means that the process is less sensitive to minor variations in temperature or moisture, reducing the risk of batch failures and ensuring consistent delivery schedules. This reliability is critical for maintaining continuous production lines and meeting the demanding timelines of pharmaceutical clients who depend on just-in-time delivery of critical intermediates for their drug development pipelines.
- Scalability and Environmental Compliance: The use of green solvents like ethanol, methanol, and water mixtures aligns with increasingly strict environmental regulations regarding volatile organic compound (VOC) emissions. The synthetic route is inherently scalable, as demonstrated by the successful preparation of numerous derivatives in the patent data, suggesting that translation from gram-scale laboratory synthesis to ton-scale commercial production is feasible with minimal process re-engineering. The generation of benign byproducts and the ease of catalyst removal simplify waste treatment protocols, facilitating compliance with environmental standards and reducing the overall environmental footprint of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these indazole-triazole derivatives. The answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing accurate insights for stakeholders evaluating this technology for potential integration into their supply chains or R&D portfolios.
Q: What is the specific biological activity of these indazole-triazole derivatives?
A: These derivatives exhibit specific antiproliferative activity against prostate cancer cells (PC-3) with IC50 values significantly lower than those for breast, liver, or gastric cancer cells, indicating high selectivity.
Q: Are the starting materials for this synthesis commercially available?
A: Yes, key starting materials such as methyl 1H-indazole-4-carboxylate, propargylamine, and various aromatic amines or benzyl bromides required for azide synthesis are readily available from standard chemical suppliers.
Q: Does the synthesis require expensive transition metal catalysts?
A: The final step utilizes a copper-catalyzed click chemistry reaction involving anhydrous copper sulfate and sodium ascorbate, which are cost-effective and easily removable compared to precious metal catalysts like palladium.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indazole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the indazole-triazole scaffold in the fight against prostate cancer. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to industrial manufacturing is seamless. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity and assay.
We invite you to engage with our technical procurement team to discuss how we can support your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of optimizing this synthesis route. We encourage you to contact us today to obtain specific COA data for related intermediates and comprehensive route feasibility assessments tailored to your volume requirements and timeline constraints.
