Advanced Synthesis of Tunable Quinoxaline OLED Materials for Next-Generation Display Technologies
Advanced Synthesis of Tunable Quinoxaline OLED Materials for Next-Generation Display Technologies
The rapid evolution of the organic semiconductor industry demands materials that offer not only high efficiency but also precise color tunability and robust thermal stability. Patent CN111909103A introduces a significant breakthrough in this domain by disclosing a novel class of quinoxaline-based organic light-emitting materials designed specifically for high-performance OLED applications. These materials feature a unique 2,3-diphenylquinoxaline core functionalized at the 6,7-positions, a structural motif that addresses critical challenges in exciton management and charge transport. By strategically modifying the steric and electronic properties of the substituents at these specific sites, the invention achieves a delicate balance between extending conjugation for red-shifting emission and disrupting planarity to prevent aggregation-caused quenching. This dual functionality makes these compounds highly attractive for manufacturers seeking reliable OLED material suppliers who can deliver components for next-generation full-color flat-panel displays.
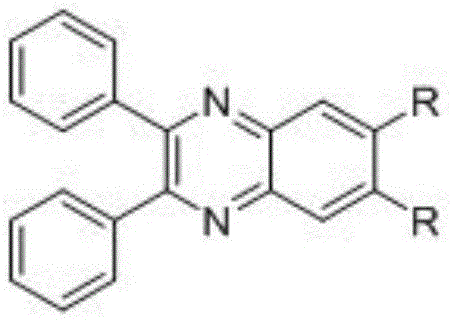
The commercial viability of any new electronic chemical hinges on the robustness of its synthetic pathway and the accessibility of its precursors. Conventional methods for synthesizing quinoxaline derivatives often suffer from limited scope in functionalization or require harsh conditions that degrade sensitive fluorophores. In contrast, the approach detailed in this patent leverages widely available commercial starting materials, specifically 1,2-diphenylethanedione and 4,5-dibromobenzene-1,2-diamine, to construct the core heterocyclic scaffold. This foundational step is followed by a versatile palladium-catalyzed cross-coupling reaction, allowing for the modular installation of diverse aromatic and heteroaromatic groups. This modularity is crucial for cost reduction in electronic chemical manufacturing, as it enables a single intermediate to be converted into a library of emitters with distinct optical properties without redesigning the entire synthesis from scratch.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional small-molecule organic light-emitting materials frequently encounter performance bottlenecks related to molecular packing and stability in the solid state. Many planar aromatic systems tend to undergo strong π-π stacking interactions when deposited as thin films, leading to non-radiative decay pathways that drastically reduce quantum yield and luminous efficiency. Furthermore, achieving precise color coordination across the RGB spectrum often requires complex, multi-step syntheses that are difficult to scale and purify. The reliance on rigid, fully planar structures in older generations of quinoxaline derivatives limits the ability to fine-tune energy levels and charge mobility independently. Consequently, device manufacturers often face trade-offs between color purity and operational lifetime, necessitating the development of new molecular architectures that can decouple these properties through intelligent structural design.
The Novel Approach
The methodology presented in CN111909103A overcomes these historical limitations by introducing bulky substituents at the 6,7-positions of the quinoxaline ring. This specific substitution pattern serves a dual purpose: it extends the π-conjugation system to modulate the bandgap for desired emission colors, while the steric bulk disrupts the coplanarity of the molecule. This disruption effectively inhibits intermolecular π-π stacking, thereby preserving high fluorescence quantum yields even in the solid state. The synthetic strategy employs a convergent route where a stable dibromo-intermediate is coupled with various boronic acids, ranging from simple phenyl groups to complex polycyclic aromatic hydrocarbons like pyrene and carbazole derivatives. This flexibility allows for the systematic optimization of thermal stability and film-forming ability, ensuring that the resulting materials meet the rigorous standards required for commercial scale-up of complex organic semiconductors.
Mechanistic Insights into Pd-Catalyzed Suzuki Coupling and Cyclization
The synthesis begins with a condensation reaction between 1,2-diphenylethanedione and 4,5-dibromobenzene-1,2-diamine in an acidic medium, typically acetic acid, at elevated temperatures around 140°C. This step constructs the central quinoxaline ring through a double nucleophilic attack of the amine groups on the diketone carbonyls, followed by dehydration. The resulting 6,7-dibromo-2,3-diphenylquinoxaline serves as a pivotal electrophilic building block. The bromine atoms at the 6,7-positions are strategically placed to activate the ring for subsequent transition-metal catalyzed cross-coupling reactions without interfering with the integrity of the quinoxaline nitrogen atoms. This intermediate exhibits excellent stability, allowing it to be isolated and stored, which is a significant logistical advantage for supply chain management compared to unstable reactive intermediates.
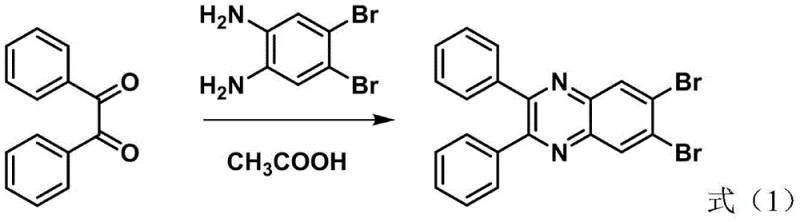
The second stage involves a palladium-catalyzed Suzuki-Miyaura coupling, a cornerstone reaction in modern organic synthesis known for its tolerance of functional groups and mild conditions. In this specific application, the dibromo-intermediate reacts with arylboronic acids in the presence of a palladium catalyst such as Pd(PPh3)4 and a base like potassium carbonate. The reaction mechanism proceeds through the oxidative addition of the palladium species into the carbon-bromine bond, followed by transmetallation with the boronate complex formed in situ, and finally reductive elimination to forge the new carbon-carbon bond. The use of a mixed solvent system comprising toluene, ethanol, and water facilitates the solubility of both organic substrates and inorganic bases, ensuring homogeneous reaction kinetics. This robust catalytic cycle allows for the efficient introduction of diverse R groups, including electron-rich carbazoles and electron-deficient aldehydes, enabling precise tuning of the HOMO-LUMO gap for specific RGB emission profiles.
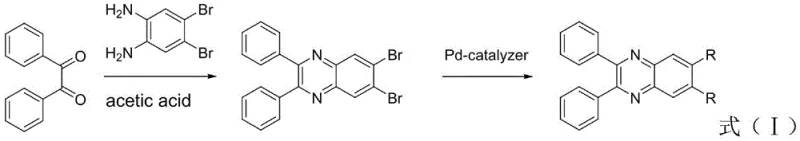
How to Synthesize 6,7-Disubstituted-2,3-diphenylquinoxaline Efficiently
The preparation of these high-performance emitting materials follows a streamlined two-step protocol that balances yield with purity. The initial condensation establishes the heterocyclic core with high efficiency, while the subsequent coupling step offers modularity. For R&D teams looking to replicate or adapt this chemistry, controlling the stoichiometry of the boronic acid and the activity of the palladium catalyst is critical to minimizing homocoupling byproducts. Detailed standardized synthesis steps are provided below to guide process development and optimization efforts.
- Condense 1,2-diphenylethanedione and 4,5-dibromobenzene-1,2-diamine in acetic acid at 140°C under inert atmosphere to form 6,7-dibromo-2,3-diphenylquinoxaline.
- Perform Suzuki coupling by reacting the dibromo-intermediate with specific boronic acids using Pd(PPh3)4 catalyst and K2CO3 base in a toluene/ethanol/water system at 90-100°C.
- Purify the final crude product through dichloromethane extraction, brine washing, rotary evaporation, and column chromatography to isolate the high-purity light-emitting material.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the synthetic route described in this patent offers substantial strategic benefits regarding raw material security and process scalability. The starting materials, benzil and dibromophenylenediamine, are commodity chemicals available from multiple global suppliers, reducing the risk of supply chain bottlenecks associated with exotic or proprietary precursors. Furthermore, the reliance on the Suzuki coupling reaction, a well-understood industrial process, means that existing manufacturing infrastructure can often be utilized with minimal retrofitting. This compatibility significantly lowers the barrier to entry for commercial production and ensures that lead times for high-purity OLED materials can be kept competitive. The ability to generate a library of derivatives from a single common intermediate also simplifies inventory management and reduces the overall working capital required to support a diverse product portfolio.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for expensive transition metals other than the catalytic amount of palladium, which can potentially be recovered and recycled in large-scale operations. By avoiding complex multi-step sequences that require protecting group strategies or cryogenic conditions, the overall process mass intensity is reduced. The use of common solvents like acetic acid, toluene, and ethanol further drives down operational expenditures compared to processes requiring specialized fluorinated solvents or supercritical fluids. These factors collectively contribute to a more economical manufacturing profile without compromising the high quality of the final electronic grade material.
- Enhanced Supply Chain Reliability: The robustness of the condensation and coupling steps ensures consistent batch-to-batch reproducibility, a critical factor for long-term supply agreements with display panel manufacturers. The intermediate 6,7-dibromo-2,3-diphenylquinoxaline is chemically stable, allowing it to be produced in bulk and stored as a strategic reserve to buffer against fluctuations in downstream demand. Additionally, the modular nature of the second step means that switching between different product variants (e.g., from blue to green emitters) can be achieved rapidly by simply changing the boronic acid feedstock, providing exceptional agility in responding to market trends.
- Scalability and Environmental Compliance: The reaction conditions operate at moderate temperatures (90-140°C) and atmospheric pressure, which simplifies reactor design and safety protocols for scale-up from kilogram to tonne quantities. The aqueous workup procedures involving brine and organic extraction are standard in the fine chemical industry, facilitating efficient waste stream management and solvent recovery. By designing molecules that inherently possess high thermal and air stability, the end-products reduce the need for stringent encapsulation in the final device, indirectly contributing to the sustainability and longevity of the consumer electronics in which they are deployed.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the structural properties and application potential of these quinoxaline derivatives. Understanding these nuances is essential for integrating these materials into existing device architectures and maximizing their performance benefits.
Q: What is the primary advantage of the 6,7-substitution pattern in these quinoxaline derivatives?
A: The specific introduction of substituents at the 6,7-positions increases conjugation length while simultaneously destroying molecular coplanarity. This structural modification effectively inhibits π-π stacking interactions, which significantly enhances luminous efficiency, quantum yield, and color purity compared to unsubstituted analogs.
Q: Can this synthesis route support full-color RGB display applications?
A: Yes, the patent demonstrates that by varying the R substituent groups—such as pyrene, carbazole, or aldehyde-functionalized aromatics—the emission spectrum can be precisely tuned. This flexibility allows for the generation of a series of organic fluorescent materials capable of covering the red, green, and blue primary colors required for full-color flat-panel displays.
Q: What are the key reaction conditions for the final coupling step?
A: The final functionalization step utilizes a standard Suzuki-Miyaura cross-coupling protocol. It requires a palladium catalyst like tetrakis(triphenylphosphine)palladium, potassium carbonate as a base, and a mixed solvent system of toluene, ethanol, and water. The reaction is typically conducted at 90-100°C for 24 to 48 hours under an inert nitrogen or argon atmosphere.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoxaline Derivatives Supplier
As the demand for high-efficiency OLED displays continues to surge, partnering with a chemically proficient manufacturer is essential for securing a competitive edge. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications required for electronic grade materials, including trace metal analysis and particle size distribution. We understand that the transition from laboratory synthesis to industrial manufacturing involves complex engineering challenges, and our team is dedicated to navigating these hurdles to deliver cost-effective solutions.
We invite you to engage with our technical procurement team to discuss how our capabilities align with your specific material requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data to validate material compatibility, we are prepared to provide comprehensive support. Contact us today to request route feasibility assessments and discover how our advanced synthesis platforms can accelerate your product development cycles while optimizing total cost of ownership.
