Advanced Non-C2 Symmetric Ruthenium Catalysts for Scalable Chiral Alcohol Production
The landscape of asymmetric synthesis is undergoing a significant transformation driven by the need for more versatile and efficient catalytic systems, as exemplified by the groundbreaking technology disclosed in Chinese Patent CN1356334A. This patent introduces a novel class of metal complexes featuring non-C2 symmetric axial chiral bisphosphine ligands, specifically derived from H4-BINAP architectures, coordinated with divalent ruthenium and chiral diamines. Unlike traditional catalysts that rely on rigid C2 symmetry, these new complexes exploit the subtle electronic and steric nuances of non-symmetric environments to achieve exceptional enantioselectivity in the hydrogenation of prochiral ketones. For R&D directors and process chemists, this represents a pivotal shift towards catalysts that can handle a wider array of substrate structures without compromising on optical purity. The ability to tune the ligand environment through variable R-groups on the phosphine and diamine moieties allows for precise optimization of the catalytic pocket, addressing long-standing challenges in the synthesis of complex chiral alcohols which are critical building blocks for active pharmaceutical ingredients (APIs).
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the field of asymmetric hydrogenation has been dominated by C2-symmetric ligands such as BINAP, DIOP, and DuPHOS, which were celebrated for reducing the number of possible transition state conformers and simplifying stereochemical outcomes.  However, this rigidity often becomes a liability when dealing with sterically demanding or electronically diverse ketone substrates that do not fit perfectly into the symmetric catalytic pocket. Conventional rhodium or ruthenium systems based on these ligands frequently require high catalyst loadings or extreme reaction conditions to drive conversions, leading to increased production costs and difficulties in downstream purification. Furthermore, the synthesis of highly pure C2-symmetric ligands can be labor-intensive and costly, creating bottlenecks in the supply chain for high-volume pharmaceutical intermediates. The lack of flexibility in these traditional systems often necessitates the development of entirely new ligand scaffolds for different substrate classes, fragmenting the process development workflow and delaying time-to-market for new drug candidates.
However, this rigidity often becomes a liability when dealing with sterically demanding or electronically diverse ketone substrates that do not fit perfectly into the symmetric catalytic pocket. Conventional rhodium or ruthenium systems based on these ligands frequently require high catalyst loadings or extreme reaction conditions to drive conversions, leading to increased production costs and difficulties in downstream purification. Furthermore, the synthesis of highly pure C2-symmetric ligands can be labor-intensive and costly, creating bottlenecks in the supply chain for high-volume pharmaceutical intermediates. The lack of flexibility in these traditional systems often necessitates the development of entirely new ligand scaffolds for different substrate classes, fragmenting the process development workflow and delaying time-to-market for new drug candidates.
The Novel Approach
The technology described in CN1356334A overcomes these limitations by introducing a non-C2 symmetric H4-BINAP ruthenium-diamine system that combines the robustness of the BINAP backbone with the tunable asymmetry required for broad substrate scope. By breaking the C2 symmetry, the catalyst creates a more differentiated chiral environment that can effectively distinguish between the prochiral faces of ketones that were previously difficult to reduce with high enantiomeric excess. This approach leverages the cooperative effect between the ruthenium center, the bulky phosphine ligand, and the chiral diamine, facilitating a bifunctional hydrogenation mechanism that operates efficiently under mild conditions. The result is a catalytic system that delivers conversion rates approaching 100% and enantiomeric excess values exceeding 98% for challenging substrates, as demonstrated in the patent examples. This versatility significantly reduces the need for substrate-specific catalyst screening, streamlining the process development phase and offering a unified solution for the cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Ru-H4-BINAP-Diamine Catalytic Hydrogenation
The core of this technological advancement lies in the unique structural arrangement of the non-C2 symmetric H4-BINAP ligand coordinated to the ruthenium center. 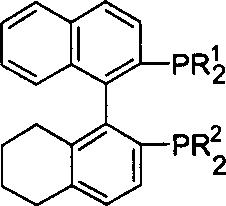 The ligand features a tetrahydro-binaphthyl backbone where the phosphine substituents (R1 and R2) can be varied from simple alkyl groups to aryl groups, allowing fine-tuning of the steric bulk around the metal center. Unlike symmetric ligands where both phosphine arms exert identical influence, the non-C2 architecture creates an asymmetric electronic density that stabilizes specific transition states more effectively. When combined with a chiral diamine co-ligand, the complex forms a highly organized six-membered pericyclic transition state during hydrogen transfer. The amine protons on the diamine ligand participate directly in the activation of the ketone substrate through hydrogen bonding, while the ruthenium hydride species delivers the hydride ion. This metal-ligand bifunctional mechanism lowers the activation energy barrier significantly, enabling reactions to proceed rapidly even at lower temperatures and hydrogen pressures, which is crucial for preserving sensitive functional groups on the substrate molecule.
The ligand features a tetrahydro-binaphthyl backbone where the phosphine substituents (R1 and R2) can be varied from simple alkyl groups to aryl groups, allowing fine-tuning of the steric bulk around the metal center. Unlike symmetric ligands where both phosphine arms exert identical influence, the non-C2 architecture creates an asymmetric electronic density that stabilizes specific transition states more effectively. When combined with a chiral diamine co-ligand, the complex forms a highly organized six-membered pericyclic transition state during hydrogen transfer. The amine protons on the diamine ligand participate directly in the activation of the ketone substrate through hydrogen bonding, while the ruthenium hydride species delivers the hydride ion. This metal-ligand bifunctional mechanism lowers the activation energy barrier significantly, enabling reactions to proceed rapidly even at lower temperatures and hydrogen pressures, which is crucial for preserving sensitive functional groups on the substrate molecule.
Impurity control in this system is inherently managed by the high stereoselectivity of the catalyst, which minimizes the formation of the unwanted enantiomer and reduces the burden on downstream chiral separation processes. The robust nature of the Ru-P-N coordination sphere ensures that the catalyst remains active throughout the reaction cycle without significant decomposition or leaching of the metal, which is a common issue with less stable homogeneous catalysts. The patent data indicates that the catalyst can be prepared in high yields (up to 89%) through a straightforward complexation process involving divalent ruthenium precursors like [RuCl2(C6H6)]2, the H4-BINAP ligand, and the diamine in polar solvents such as DMF or toluene. This synthetic simplicity translates directly to supply chain reliability, as the precursors are commercially available and the preparation does not require exotic reagents or cryogenic conditions. The resulting catalyst solutions are stable and can be handled with standard Schlenk techniques, facilitating their integration into existing manufacturing infrastructure without the need for specialized equipment upgrades.
How to Synthesize H4-BINAP Ruthenium Complexes Efficiently
The preparation of these high-performance catalysts is designed for operational simplicity and scalability, making it accessible for both laboratory research and industrial production teams. The process typically involves the sequential addition of the ruthenium precursor and the H4-BINAP ligand in a polar organic solvent, followed by heating to facilitate coordination, and finally the introduction of the chiral diamine at controlled temperatures to complete the complex formation. Detailed standardized synthesis steps see the guide below.
- Complexation of divalent ruthenium precursor with H4-BINAP ligand in polar solvent at elevated temperatures.
- Addition of chiral diamine co-ligand at controlled temperatures to form the active catalytic species.
- Isolation of the catalyst complex via precipitation and vacuum drying for immediate use in hydrogenation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this non-C2 symmetric catalyst technology offers compelling economic and logistical benefits that extend beyond mere technical performance. The primary advantage lies in the significant simplification of the synthetic route for chiral alcohols, which eliminates the need for resolution steps or the use of stoichiometric chiral auxiliaries that generate substantial waste. By achieving high enantiomeric excess directly through catalysis, manufacturers can drastically reduce the consumption of raw materials and solvents, leading to substantial cost savings in overall production. Furthermore, the mild reaction conditions (typically 25-60°C and moderate hydrogen pressure) reduce energy consumption and enhance operational safety, lowering the total cost of ownership for the manufacturing facility. The robustness of the catalyst system also implies longer catalyst life and potentially lower catalyst loading requirements, which directly impacts the cost of goods sold (COGS) for high-value pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal scavengers and the reduction in downstream purification steps due to high selectivity contribute to a leaner manufacturing process. The ability to use simpler, non-C2 symmetric ligands which may be easier to synthesize than their C2 counterparts further drives down the input costs for the catalyst itself. This efficiency allows for a more competitive pricing structure for the final chiral alcohol products, enhancing margin potential for the end manufacturer.
- Enhanced Supply Chain Reliability: The reliance on commercially available ruthenium salts and adaptable ligand structures mitigates the risk of supply disruptions associated with proprietary or single-source reagents. The synthetic protocol is robust against minor variations in reaction parameters, ensuring consistent batch-to-batch quality which is critical for regulatory compliance in the pharmaceutical sector. This reliability reduces the lead time for high-purity pharmaceutical intermediates by minimizing the need for re-processing or batch rejection due to quality deviations.
- Scalability and Environmental Compliance: The process is inherently scalable from gram to multi-ton quantities without fundamental changes to the reaction engineering, supporting the commercial scale-up of complex pharmaceutical intermediates. The use of standard solvents and the generation of minimal hazardous waste align with green chemistry principles, simplifying environmental permitting and waste disposal logistics. This environmental compatibility is increasingly important for maintaining social license to operate and meeting the sustainability goals of global corporate partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic technology in industrial settings. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making.
Q: What is the primary advantage of non-C2 symmetric ligands over traditional BINAP?
A: Non-C2 symmetric ligands offer greater conformational flexibility and reduced transition state degeneracy, allowing for superior enantioselectivity across a broader range of sterically diverse ketone substrates compared to rigid C2-symmetric analogs.
Q: Can this catalyst system handle functionalized ketones sensitive to harsh conditions?
A: Yes, the Ru-H4-BINAP-diamine system operates under mild conditions (20-60°C, 10-50 atm H2) and tolerates various functional groups including amines, halogens, and ethers without requiring protective group strategies.
Q: Is the catalyst synthesis scalable for industrial production?
A: The synthesis involves simple mixing of commercially available precursors in standard polar solvents with high yields (up to 89%), making it highly amenable to multi-kilogram scale-up for commercial API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable H4-BINAP Catalyst Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the non-C2 symmetric Ru-H4-BINAP-diamine catalyst system in advancing the synthesis of chiral pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent technology are fully realized in your supply chain. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications and enantiomeric excess values, guaranteeing that every batch of catalyst or intermediate meets the highest international standards. We understand that consistency is key in API manufacturing, and our process engineers are adept at optimizing reaction parameters to maximize yield and selectivity while minimizing impurity profiles.
We invite you to collaborate with us to evaluate the feasibility of integrating this advanced hydrogenation technology into your current production lines. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific substrate portfolio, demonstrating how this catalyst system can optimize your manufacturing economics. Please contact us to request specific COA data for our catalyst offerings and comprehensive route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to cutting-edge catalytic solutions backed by a commitment to quality, reliability, and continuous innovation in fine chemical manufacturing.
