Advanced Non-C2 Symmetric Ruthenium Complexes for High-Efficiency Asymmetric Ketone Hydrogenation
The landscape of asymmetric synthesis has been fundamentally transformed by the continuous evolution of chiral transition metal catalysts, with patent CN1356334A representing a significant milestone in the development of non-C2 symmetric axial chiral bisphosphine ligand metal complexes. This intellectual property discloses a sophisticated class of ruthenium-based catalysts formed by the complexation of R or S configured H4-BINAP ligands, divalent ruthenium compounds, and chiral diamines, specifically engineered to overcome the stereochemical limitations of earlier generations of catalysts. The invention addresses the critical need for highly efficient, selective, and robust catalytic systems capable of reducing a broad spectrum of prochiral ketones into optically active alcohols, which serve as indispensable building blocks in the manufacture of active pharmaceutical ingredients and fine chemicals. By breaking the traditional C2 symmetry constraint found in many classic ligands like BINAP or DIOP, this technology offers a unique chiral environment that enhances the differentiation of enantiotopic faces in substrate molecules, thereby driving higher enantiomeric purity in the final product.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the field of homogeneous asymmetric hydrogenation relied heavily on C2 symmetric ligands, such as the pioneering DIPAMP and BINAP systems, which, while effective for certain substrate classes like dehydroamino acids, often exhibited restricted scope when applied to simple aromatic ketones or functionally diverse carbonyl compounds. Traditional rhodium-based Wilkinson-type catalysts and early ruthenium-BINAP complexes frequently suffered from moderate activity or insufficient enantioselectivity when confronted with sterically demanding or electronically deactivated ketones, necessitating high catalyst loadings or extreme reaction conditions that are economically and environmentally undesirable. Furthermore, the rigid symmetry of C2 ligands limits the ability to fine-tune the steric quadrants around the metal center, creating a 'ceiling' on the achievable optical purity for substrates that do not fit perfectly into the symmetric chiral pocket, thus forcing process chemists to resort to costly resolution techniques or multi-step synthetic detours to obtain the desired chiral alcohols.
The Novel Approach
The methodology outlined in CN1356334A introduces a paradigm shift by utilizing non-C2 symmetric H4-BINAP ligands in conjunction with chiral diamines to create a ternary ruthenium complex that exhibits exceptional catalytic performance across a diverse array of ketone substrates. This novel approach leverages the structural flexibility and distinct steric properties of the non-symmetric ligand backbone to create a more adaptable active site, allowing for precise spatial arrangement of the substrate during the hydride transfer step. As demonstrated in the patent examples, this system achieves near-quantitative conversion and high enantiomeric excess (ee) values for substrates ranging from simple acetophenones to complex functionalized ketones containing amino, nitro, and halogen groups, all under relatively mild hydrogen pressures and temperatures. 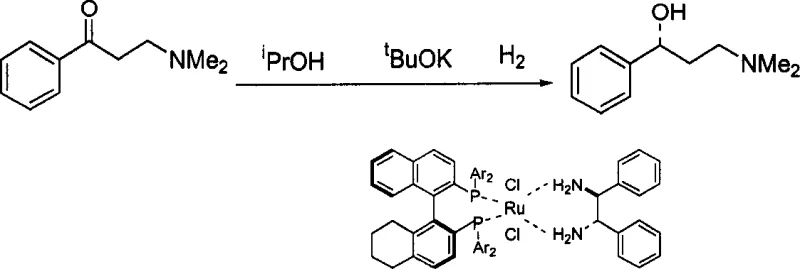
Mechanistic Insights into Non-C2 Symmetric Ruthenium-Diamine Catalysis
The superior performance of this catalyst system is rooted in the intricate cooperative mechanism between the ruthenium center, the non-C2 symmetric bisphosphine ligand, and the chiral diamine co-ligand, which functions through a metal-ligand bifunctional pathway. Unlike classical mechanisms where the metal solely activates the substrate, this system utilizes the NH protons of the diamine ligand to activate the carbonyl oxygen via hydrogen bonding while the ruthenium-hydride species simultaneously delivers a hydride to the carbonyl carbon, proceeding through a concerted six-membered pericyclic transition state. The absence of C2 symmetry in the H4-BINAP ligand, as structurally depicted below, allows for an asymmetric distribution of steric bulk around the ruthenium center, effectively creating four distinct quadrants that can be independently optimized to maximize repulsive interactions with the unwanted enantiomer transition state while stabilizing the desired one. 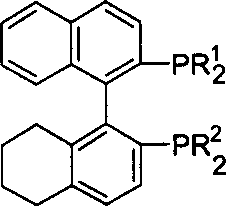
Furthermore, the electronic properties of the substituents on the phosphine aryl rings (R1 and R2) and the diamine backbone (R3 and R4) play a crucial role in modulating the electron density at the metal center, thereby influencing the rate of oxidative addition and reductive elimination steps within the catalytic cycle. This tunability ensures that the catalyst maintains high activity even in the presence of potentially coordinating functional groups on the substrate, such as amines or ethers, which might otherwise poison less robust catalytic systems. The rigorous control over the transition state geometry afforded by this non-C2 symmetric architecture minimizes the formation of byproduct impurities and racemic material, directly translating to simplified downstream purification processes and higher overall yields of the target chiral alcohol intermediate.
How to Synthesize H4-BINAP Ruthenium Complexes Efficiently
The preparation of these high-performance catalysts is designed to be operationally straightforward, involving the sequential or one-pot complexation of commercially available or easily synthesized precursors in standard polar organic solvents. The process typically begins with the reaction of a divalent ruthenium source, such as [RuCl2(C6H6)]2, with the chiral H4-BINAP ligand in a solvent like DMF or toluene at elevated temperatures to ensure complete ligand exchange and coordination. Following the formation of the ruthenium-bisphosphine intermediate, the chiral diamine is introduced, often at lower temperatures, to complete the assembly of the active ternary complex, which can then be isolated as a stable solid or used in situ for immediate hydrogenation campaigns.
- Prepare the reaction vessel under inert atmosphere by charging a Schlenk flask with the divalent ruthenium precursor and the non-C2 symmetric H4-BINAP ligand in a polar organic solvent.
- Heat the mixture to facilitate the initial coordination between the ruthenium center and the bisphosphine ligand, ensuring complete dissolution and complex formation.
- Cool the solution to room temperature and introduce the chiral diamine co-ligand, stirring for an extended period to finalize the ternary catalytic complex structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this non-C2 symmetric ruthenium catalyst technology presents a compelling value proposition centered on cost efficiency, supply reliability, and process scalability. The shift from expensive and supply-constrained rhodium catalysts to robust ruthenium-based systems significantly reduces the raw material cost burden associated with precious metal usage, while the high turnover numbers demonstrated in the patent data imply that lower catalyst loadings can be utilized without sacrificing reaction throughput. Additionally, the ability of this catalyst to tolerate a wide range of functional groups eliminates the need for extensive protecting group strategies, thereby shortening the overall synthetic route and reducing the consumption of auxiliary reagents and solvents, which contributes to substantial cost reduction in pharmaceutical intermediate manufacturing.
- Cost Reduction in Manufacturing: The implementation of this catalytic system drives down production costs by enabling the use of base metals like ruthenium instead of scarcer platinum group metals, while the high selectivity minimizes waste generation and the need for costly chiral chromatography purification steps. The mild reaction conditions, often requiring only moderate hydrogen pressure and ambient to slightly elevated temperatures, further decrease energy consumption and reduce the wear and tear on high-pressure reactor equipment, leading to lower capital expenditure and operational overheads over the lifecycle of the manufacturing process.
- Enhanced Supply Chain Reliability: By utilizing ligand scaffolds that are synthetically accessible and stable, manufacturers can mitigate the risks associated with supply chain disruptions common to exotic or highly specialized reagents. The robustness of the catalyst against air and moisture, relative to more sensitive alternatives, simplifies logistics and storage requirements, ensuring consistent quality and availability of the catalytic system for continuous production runs, which is critical for maintaining the supply continuity of high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The protocol described in the patent is inherently scalable, having been validated in batch reactors with straightforward workup procedures involving filtration and solvent evaporation, making it ideal for commercial scale-up of complex chiral alcohols. The use of green solvents like isopropanol and the generation of minimal hazardous waste align with modern environmental, health, and safety (EHS) standards, facilitating regulatory approval and reducing the environmental footprint of the chemical manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this non-C2 symmetric ruthenium catalyst technology, derived directly from the experimental data and claims within the patent documentation. These insights are intended to clarify the operational parameters and potential benefits for R&D and production teams evaluating this technology for integration into their existing synthetic platforms.
Q: What is the primary advantage of using non-C2 symmetric ligands over traditional C2 symmetric ligands?
A: Non-C2 symmetric ligands, such as the H4-BINAP derivatives described in the patent, provide a more differentiated steric environment around the metal center. This lack of symmetry allows for finer tuning of the chiral pocket, often resulting in superior enantioselectivity for challenging prochiral ketones that traditional C2 symmetric ligands struggle to differentiate effectively.
Q: Can this catalyst system handle functionalized ketones commonly found in API synthesis?
A: Yes, the patent data demonstrates excellent compatibility with a wide range of functionalized ketones, including those containing amino, nitro, halo, and alkoxy groups. The cooperative catalysis between the ruthenium center, the bisphosphine, and the chiral diamine ensures high conversion rates and enantiomeric excess even in the presence of sensitive functional groups.
Q: What are the typical reaction conditions required for this hydrogenation process?
A: The process operates under relatively mild conditions, typically utilizing hydrogen pressures around 10 to 50 atmospheres and temperatures ranging from room temperature to 60°C. The use of common alcoholic solvents like isopropanol and simple bases like potassium tert-butoxide further simplifies the operational requirements for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable H4-BINAP Ruthenium Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced asymmetric hydrogenation technologies in accelerating the development of next-generation therapeutics and fine chemicals. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of chiral intermediate meets the exacting standards required by global regulatory bodies and end-user applications.
We invite you to collaborate with our technical team to explore how this non-C2 symmetric catalyst technology can optimize your specific synthetic routes and deliver tangible economic benefits. Please contact our technical procurement team to request a Customized Cost-Saving Analysis, along with specific COA data and route feasibility assessments tailored to your project's unique requirements and timeline.
