Advanced Sulfonium Salt Photoacid Generators for High-Resolution Lithography and Semiconductor Manufacturing
Introduction to Next-Generation Photoacid Generators
The relentless miniaturization of integrated circuits has placed unprecedented demands on lithography materials, specifically requiring photoacid generators (PAGs) that can deliver superior resolution with minimal line edge roughness. Patent CN111077731A introduces a groundbreaking class of sulfonium salt photoacid generators that incorporate a unique "trumpet-shaped tea alcohol" structural motif. This innovation addresses the critical challenge of acid diffusion, a phenomenon that often blurs the boundary between exposed and unexposed regions in chemically amplified resists. By integrating this bulky, naturally derived scaffold into the anion structure, the technology achieves a delicate balance between hydrophilicity and lipophilicity, essential for modern semiconductor processing.
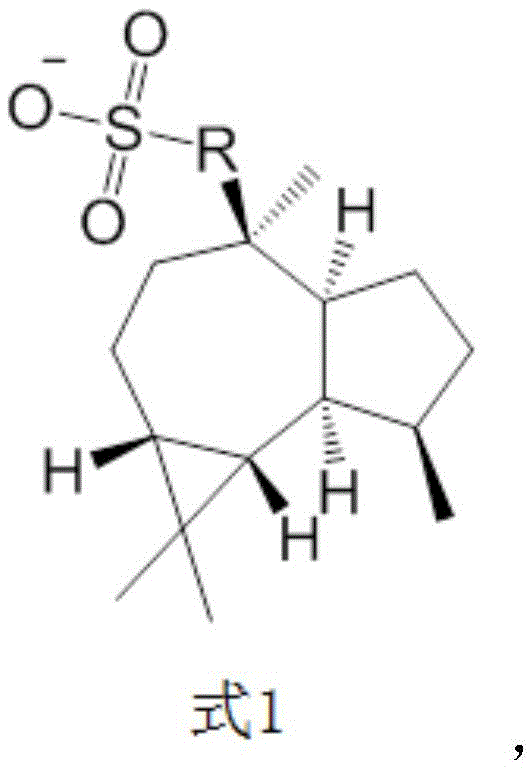
The core of this invention lies in the specific architectural design of the anion, which is covalently linked to the trumpet-shaped tea alcohol framework. This structural modification is not merely cosmetic; it fundamentally alters the physicochemical properties of the PAG. As the industry pushes towards smaller node sizes, the control of acid diffusion becomes paramount. The disclosed compounds offer a robust solution by physically restricting the mobility of the photogenerated acid, thereby enhancing the fidelity of the pattern transfer process. For procurement teams seeking a reliable photoacid generator supplier, this technology represents a significant leap forward in material performance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional onium salt photoacid generators, while commercially mature, suffer from inherent limitations regarding acid diffusion. In standard sulfonium or iodonium salts, the counter-anions are often small and highly mobile. Upon exposure to light, the generated super-strong acids can diffuse laterally beyond the intended exposure area. This uncontrolled diffusion leads to increased line edge roughness (LER) and line width roughness (LWR), which are critical defects in the manufacturing of large-scale and ultra-large-scale integrated circuits. Furthermore, conventional PAGs often struggle to maintain an optimal balance between solubility in the organic resin matrix and adhesion to the substrate, leading to coating defects or poor development contrast.
The Novel Approach
The novel approach detailed in the patent overcomes these deficiencies by engineering the anion to act as a diffusion barrier. By attaching the sulfonic acid group to the rigid and bulky trumpet-shaped tea alcohol skeleton, the resulting molecule possesses a significantly larger hydrodynamic radius. This steric bulk effectively suppresses the migration of the acid species after photolysis. Additionally, the presence of ester linkages within the structure enhances compatibility with organic solvents and polymer resins, ensuring a homogeneous film formation. This dual functionality—diffusion inhibition and solubility enhancement—makes these compounds ideal candidates for cost reduction in photoresist manufacturing by improving yield and reducing defect rates.
Mechanistic Insights into Esterification and Ion Exchange Synthesis
The synthesis of these advanced photoacid generators relies on a straightforward yet highly effective two-stage chemical strategy: functionalization of the natural product followed by salt formation. The process begins with the activation of the hydroxyl group on the trumpet-shaped tea alcohol. Through an esterification reaction, typically catalyzed by p-toluenesulfonic acid in a refluxing toluene system, the sulfonic acid derivative is covalently bonded to the core structure. This step is crucial as it installs the acid-generating moiety directly onto the diffusion-inhibiting scaffold. The reaction conditions are mild enough to preserve the stereochemical integrity of the chiral centers within the tea alcohol derivative, which may further influence the packing and solubility properties of the final salt.
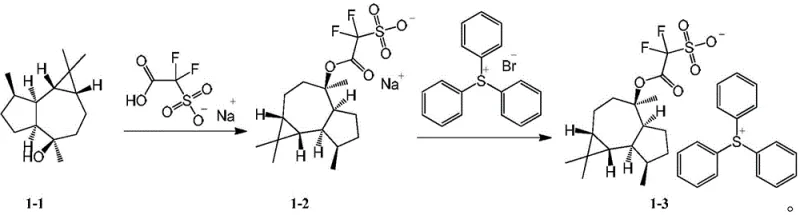
Following the isolation of the intermediate sulfonate, the final step involves a classic ion exchange reaction. The sodium salt of the functionalized tea alcohol is reacted with a halogenated sulfonium salt, such as triphenylsulfonium bromide or tris(4-methylphenyl)sulfonium chloride. This metathesis reaction occurs in a biphasic solvent system, typically dichloromethane and water, allowing for the efficient exchange of the sodium cation for the bulky sulfonium cation. The resulting product precipitates or can be crystallized out, yielding the final photoacid generator with high purity. This mechanism ensures that the final product is free from metal contaminants, a critical requirement for high-purity electronic chemicals used in semiconductor fabrication.
How to Synthesize Sulfonium Salt Photoacid Generator Efficiently
The preparation of these specialized photoacid generators is designed for operational simplicity and high yield, making it accessible for both laboratory scale-up and industrial production. The process leverages standard organic synthesis techniques such as reflux, filtration, and recrystallization, avoiding the need for exotic catalysts or high-pressure equipment. The initial esterification step drives the reaction to completion through the removal of water, ensuring high conversion of the valuable trumpet-shaped tea alcohol starting material. Subsequent purification steps, including washing with acetonitrile and recrystallization from methyl tert-butyl ether, effectively remove unreacted starting materials and by-products.
- React trumpet-shaped tea alcohol with sulfonic acid derivatives (e.g., sodium difluorosulfoacetate) in toluene under reflux to form the sulfonate intermediate.
- Purify the intermediate solid via filtration, washing with acetonitrile, and recrystallization from methyl tert-butyl ether.
- Perform ion exchange by reacting the purified sulfonate intermediate with a sulfonium halide (e.g., triphenylsulfonium bromide) in a dichloromethane/water biphasic system.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the adoption of this new synthetic route offers distinct strategic advantages rooted in process efficiency and raw material availability. The utilization of trumpet-shaped tea alcohol as a starting material is particularly noteworthy; as a derivative of a natural product, it represents a renewable and potentially cost-effective feedstock compared to fully synthetic petrochemical precursors. The synthetic pathway eliminates the need for transition metal catalysts, which not only simplifies the purification process but also removes the regulatory and environmental burdens associated with heavy metal residue testing. This translates directly into streamlined quality control protocols and faster batch release times.
- Cost Reduction in Manufacturing: The synthetic route is characterized by high atom economy and simple workup procedures. The reactions proceed with high yields, as evidenced by the experimental data where yields exceed 90% in the ion exchange step. By minimizing the number of purification steps and avoiding expensive chromatographic separations, the overall cost of goods sold (COGS) is significantly optimized. Furthermore, the use of common solvents like toluene, dichloromethane, and methyl tert-butyl ether ensures that solvent recovery and recycling can be easily implemented, further driving down operational expenses.
- Enhanced Supply Chain Reliability: The reliance on readily available reagents such as sodium difluorosulfoacetate and triphenylsulfonium bromide mitigates the risk of supply chain disruptions. These materials are commodity chemicals with established global supply networks. The robustness of the reaction conditions—reflux temperatures and atmospheric pressure—means that the process can be transferred to multiple manufacturing sites with minimal re-validation effort. This flexibility ensures a continuous supply of high-purity sulfonium salts even in the face of regional logistical challenges.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory experiments to multi-kilogram pilot runs. The absence of hazardous reagents and the generation of benign by-products (such as sodium halides) simplify waste treatment and disposal. The "green" nature of using a natural product derivative aligns with increasingly stringent environmental regulations and corporate sustainability goals. This compliance reduces the administrative overhead related to environmental permitting and waste management, facilitating smoother commercial scale-up of complex electronic chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel photoacid generator technology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and performance characteristics of the material. Understanding these details is crucial for R&D teams evaluating the material for next-generation resist formulations.
Q: How does the trumpet-shaped structure improve photoresist performance?
A: The bulky trumpet-shaped tea alcohol structure increases the molecular weight and steric hindrance of the anion, which physically inhibits the diffusion of the generated acid during exposure, thereby reducing line edge roughness (LER).
Q: What are the solubility characteristics of this new photoacid generator?
A: The inclusion of ester groups within the structure enhances lipid solubility in organic resins and solvents, ensuring uniform dissolution, while the sulfonate group maintains sufficient hydrophilicity for adhesion to silicon wafers.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the synthesis utilizes simple unit operations such as reflux, filtration, and recrystallization without requiring complex chromatography or extreme conditions, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfonium Salt Photoacid Generator Supplier
As the semiconductor industry continues to evolve, the demand for specialized materials that enable finer patterning capabilities is growing exponentially. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the precise synthesis and purification required for high-purity electronic chemicals, ensuring that every batch meets the stringent purity specifications demanded by leading photoresist manufacturers. Our rigorous QC labs employ advanced analytical techniques to verify the absence of metallic impurities and confirm the structural integrity of the final product.
We invite you to collaborate with our technical team to explore how this innovative trumpet-shaped tea alcohol-based PAG can enhance your formulation portfolio. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring a seamless integration of this advanced material into your supply chain.
