Advanced Total Synthesis of Estrone Using Tetralone for Scalable Pharmaceutical Production
Advanced Total Synthesis of Estrone Using Tetralone for Scalable Pharmaceutical Production
The pharmaceutical industry is constantly seeking robust, scalable, and cost-effective routes for the production of critical steroid hormones, and the total synthesis preparation method of estrone disclosed in patent CN110746477B represents a significant technological leap forward in this domain. Unlike traditional extraction or semi-synthetic methods that rely on scarce natural precursors, this innovative approach utilizes 6-methoxy tetralone as a readily available starting material to construct the steroid skeleton through a concise seven-step sequence. This patent details a fully synthetic pathway that integrates key organic transformations such as Grignard addition, acid-catalyzed cyclization, and selective catalytic hydrogenation to achieve high stereoselectivity and purity. For R&D directors and procurement managers alike, this methodology offers a compelling alternative to legacy processes, promising enhanced supply chain security and reduced dependency on biological fermentation substrates. The technical elegance of this route lies in its ability to assemble the complex tetracyclic core of estrone under relatively mild reaction conditions, thereby minimizing energy consumption and simplifying downstream purification protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of estrone has been heavily reliant on either direct extraction from biological sources or semi-synthetic routes starting from expensive steroidal intermediates derived from plant phytosterols. As illustrated in the prior art pathways, methods such as microbial conversion using Arthrobacter simplex or chemical aromatization of dehydroepiandrosterone (ADD) suffer from inherent structural bottlenecks. The microbial route, while green in principle, is plagued by long fermentation cycles ranging from 36 to 60 hours, sensitivity to dissolved oxygen levels, and the complex downstream separation required to meet USP standards. Furthermore, the chemical route starting from ADD faces severe supply chain vulnerabilities; ADD itself is obtained through secondary fermentation of phytosterols, making its price volatile and its availability limited by agricultural outputs. These conventional methods often involve toxic reagents, generate significant waste streams, and struggle with low overall yields due to the multiple steps required to modify the existing steroid nucleus rather than building it from scratch.
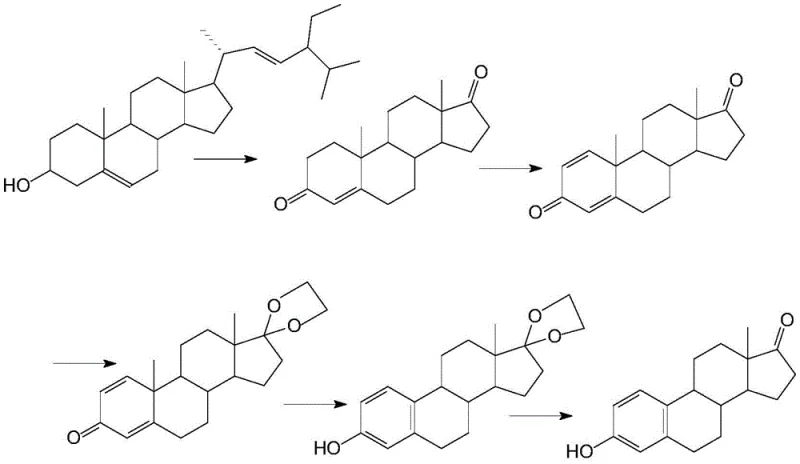
The Novel Approach
In stark contrast to these legacy constraints, the novel total synthesis route described in the patent leverages the accessibility of tetralone derivatives to construct the estrone framework de novo. This approach fundamentally shifts the paradigm from modifying a complex, expensive natural product to assembling the molecule from simple, commodity-grade chemicals. By initiating the synthesis with 6-methoxy tetralone, the process bypasses the need for costly fermentation-derived substrates entirely. The seven-step sequence is designed for operational simplicity, utilizing standard chemical reactors and avoiding the stringent sterility requirements of bioprocessing. This transition from a bio-based to a chemo-synthetic platform not only stabilizes the raw material supply but also allows for precise control over impurity profiles through well-defined chemical parameters. The result is a manufacturing process that is inherently more robust, scalable, and economically viable for meeting the growing global demand for estrogenic compounds.
Mechanistic Insights into the Seven-Step Synthetic Sequence
The core of this technological advancement lies in the meticulously orchestrated reaction sequence that builds the steroid skeleton with high fidelity. The process begins with a Grignard reaction where vinyl magnesium chloride adds to 6-methoxy tetralone at a controlled temperature of 50-60°C, forming a magnesium alkoxide intermediate that is subsequently hydrolyzed to yield the vinyl-substituted tetralone. This intermediate then undergoes a condensation reaction with 2-methyl-1,3-cyclopentanedione (the methyl D-ring) to forge the critical carbon-carbon bond connecting the A/B rings to the D-ring. Following this, an acid-catalyzed cyclization at 70-80°C closes the C-ring, establishing the tetracyclic gonane skeleton characteristic of steroids. The subsequent steps involve protecting the C17 ketone as an ethylene glycol ketal to prevent unwanted side reactions during the crucial hydrogenation phase. This hydrogenation step is particularly noteworthy, employing a specialized composite catalyst comprising palladium, zinc, and manganese on a calcium carbonate support to selectively reduce the double bonds while preserving the aromatic A-ring integrity. Finally, acidic hydrolysis removes the ketal protecting group to reveal the C17 ketone, yielding the final estrone product with high purity.
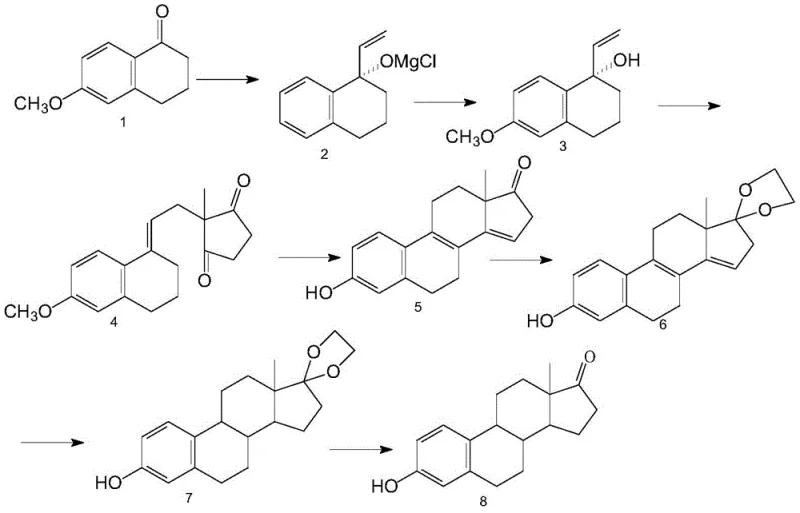
From an impurity control perspective, this synthetic route offers distinct advantages over semi-synthetic alternatives. The use of a custom-formulated hydrogenation catalyst ensures high stereoselectivity, minimizing the formation of diastereomeric impurities that are notoriously difficult to separate in steroid chemistry. Furthermore, the intermediate purification steps, such as the crystallization of the condensate and the ketal, serve as effective checkpoints to remove byproducts before they can propagate through the synthesis. The final purification in glacial acetic acid provides an additional layer of quality assurance, ensuring that the resulting estrone meets stringent pharmaceutical specifications. This level of control over the chemical trajectory allows manufacturers to consistently produce high-purity material, reducing the risk of batch failures and ensuring compliance with regulatory standards for active pharmaceutical ingredients.
How to Synthesize Estrone Efficiently
The synthesis of estrone via this total synthesis route requires precise adherence to reaction conditions and stoichiometry to maximize yield and purity. The process involves a series of interconnected chemical transformations, starting from the Grignard addition and culminating in the final hydrolytic deprotection. Each step has been optimized to balance reaction kinetics with selectivity, ensuring that the complex polycyclic structure is assembled efficiently. While the general workflow is outlined below, successful implementation requires careful attention to temperature control, reagent quality, and workup procedures to mitigate the formation of side products. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis protocol provided in the technical documentation.
- Perform Grignard addition of vinyl magnesium chloride to 6-methoxy tetralone at 50-60°C, followed by alkaline hydrolysis to obtain the vinyl tetralone intermediate.
- Condense the vinyl tetralone with 2-methyl-1,3-cyclopentanedione (methyl D-ring) and perform acidic cyclization at 70-80°C to form the steroid backbone.
- Protect the ketone as a ketal, hydrogenate using a specialized Pd/C/Zn/Mn catalyst, and finally hydrolyze under acidic conditions to yield high-purity estrone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift to this total synthesis route translates into tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the decoupling of production from the volatile agricultural markets that dictate the price of phytosterol-based starting materials. By utilizing tetralone, a commodity chemical produced on a massive scale for other industries, manufacturers can secure a stable and predictable raw material supply. This stability is crucial for long-term planning and contract negotiations, as it eliminates the risk of sudden price spikes or shortages associated with crop failures or fermentation bottlenecks. Additionally, the chemical nature of the process allows for continuous optimization and scale-up without the biological lag times inherent in fermentation processes, enabling faster response to market demand fluctuations.
- Cost Reduction in Manufacturing: The economic impact of this synthetic route is profound, driven primarily by the elimination of expensive fermentation-derived substrates like ADD. By replacing these high-cost inputs with inexpensive tetralone derivatives, the overall cost of goods sold is significantly reduced. Furthermore, the process operates under mild conditions that do not require specialized high-pressure or cryogenic equipment, leading to lower capital expenditure and reduced energy consumption. The streamlined seven-step sequence also minimizes solvent usage and waste generation, contributing to lower disposal costs and a smaller environmental footprint. These cumulative efficiencies result in a highly competitive cost structure that allows for substantial margin improvement or aggressive pricing strategies in the marketplace.
- Enhanced Supply Chain Reliability: Supply chain resilience is markedly improved by adopting a fully synthetic pathway that relies on a diverse portfolio of petrochemical feedstocks rather than a single biological source. The availability of tetralone and other reagents is supported by a mature global chemical infrastructure, ensuring consistent access regardless of seasonal or geopolitical factors affecting agricultural production. Moreover, the synthetic process is less susceptible to biological contamination or strain degeneration, which can halt production in fermentation-based facilities for extended periods. This reliability ensures uninterrupted supply to downstream customers, fostering stronger partnerships and reducing the need for excessive safety stock inventory.
- Scalability and Environmental Compliance: The scalability of this process is exceptional, as it utilizes standard unit operations such as reflux, distillation, and filtration that are easily replicated from pilot plant to commercial scale. The absence of complex biological media simplifies wastewater treatment, as the effluent consists primarily of organic solvents and salts that can be managed through established recovery and neutralization protocols. The use of a heterogeneous catalyst in the hydrogenation step further facilitates easy separation and recycling, minimizing heavy metal discharge. These features align perfectly with modern green chemistry principles and regulatory requirements, positioning manufacturers as responsible stewards of the environment while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the total synthesis of estrone. These answers are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical rationale behind this advanced manufacturing method. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this technology into their existing production portfolios.
Q: What are the advantages of the tetralone route over microbial fermentation for estrone?
A: The tetralone route avoids the complexities of biological fermentation, such as strain stability issues and long cycle times. It utilizes readily available chemical starting materials, offering a more consistent and scalable supply chain compared to the limited availability of fermentation substrates like ADD.
Q: How does the novel catalyst improve the hydrogenation step?
A: The process employs a self-made composite catalyst containing palladium, zinc, and manganese on calcium carbonate. This specific formulation enhances stereoselectivity during the hydrogenation of the double bonds, ensuring the correct stereochemistry for the final estrone product while operating under mild pressure and temperature conditions.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the method is designed for industrial scalability. It uses common solvents like ethanol and toluene, avoids extreme cryogenic conditions, and relies on standard unit operations like reflux and filtration, making it highly adaptable for multi-ton manufacturing facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Estrone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this total synthesis technology for the global steroid market. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this patented laboratory methodology into a robust commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop to plant floor is seamless and efficient. We are committed to delivering high-purity estrone that meets stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality assurance guarantees that every batch conforms to the highest industry standards, providing our partners with the confidence they need to bring their formulations to market.
We invite you to explore how this innovative synthesis route can optimize your supply chain and reduce your manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate the viability of this approach for your business. Let us collaborate to secure a sustainable and cost-effective future for your estrone supply needs.
