Optimizing Herbicide Intermediate Production Through Novel Oxazinone Cyclization Technology
The chemical landscape for agrochemical intermediate manufacturing is constantly evolving, driven by the need for more efficient and cost-effective synthetic routes. Patent CN1344259A introduces a significant technological breakthrough in the preparation of 6-(perfluoroalkyl)uracil compounds, which serve as critical active ingredients in modern herbicide formulations. This intellectual property outlines an improved process that circumvents the limitations of prior art methods, specifically targeting the synthesis of 2-(N,N-disubstituted)amino-4-(perfluoroalkyl)-1,3-oxazin-6-one compounds as key intermediates. By shifting away from problematic reagents, this technology offers a robust pathway for producing high-purity agrochemical intermediates. For R&D directors and procurement specialists, understanding the mechanistic advantages of this patent is essential for optimizing supply chains and reducing overall manufacturing costs in the competitive herbicide market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-(N,N-disubstituted)amino-4-(perfluoroalkyl)-1,3-oxazin-6-one compounds relied heavily on the use of imidoyl chloride hydrochloride compounds. As documented in earlier literature such as Bull.Soc.Chem.Belg., 101(4), pp. 313-321 (1992), these traditional reagents present substantial operational challenges. They are notoriously difficult to handle due to their instability and hygroscopic nature, which complicates storage and transportation logistics. Furthermore, the cost associated with sourcing high-quality imidoyl chloride hydrochloride compounds is relatively expensive, creating a bottleneck in the cost reduction in agrochemical manufacturing. The harsh reaction conditions often required to manage these unstable intermediates can also lead to lower yields and increased formation of by-products, necessitating complex purification steps that further erode profit margins and extend lead times.
The Novel Approach
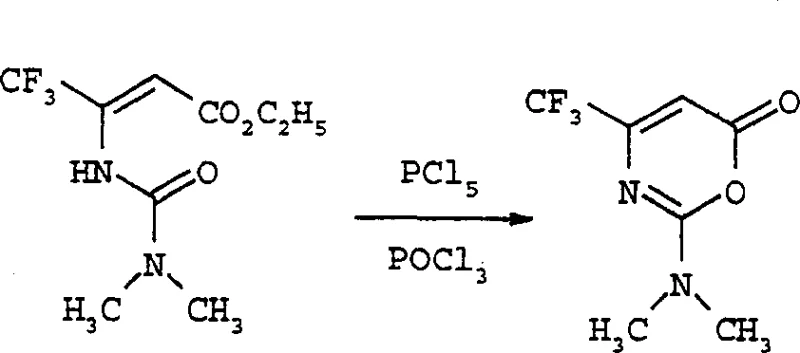
The methodology disclosed in CN1344259A represents a paradigm shift by utilizing beta-amino-beta-(perfluoroalkyl)acrylate compounds reacting with carbamoyl chloride compounds in the presence of a base. This novel approach effectively avoids the use of the problematic imidoyl chloride hydrochloride compounds entirely. The reaction proceeds through a stable urea intermediate, which is subsequently cyclized using phosphorus pentahalide or oxalyl halide. This strategic change in synthetic design not only simplifies the operational workflow but also enhances the safety profile of the manufacturing process. By employing readily available starting materials and more manageable reaction conditions, this method facilitates the commercial scale-up of complex herbicides. The ability to generate the core oxazinone structure with greater efficiency directly translates to improved supply chain reliability and substantial cost savings for downstream producers of perfluoroalkyl uracil derivatives.
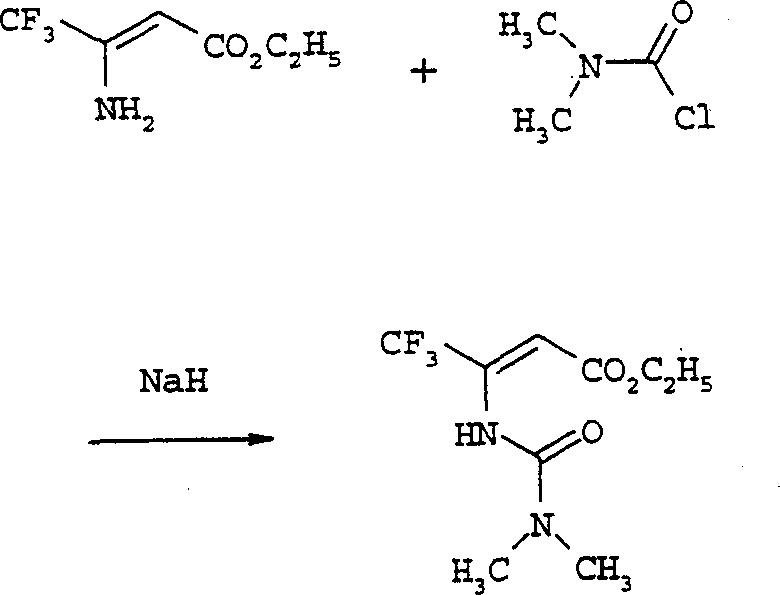
Mechanistic Insights into Carbamoyl Chloride Mediated Cyclization
The core of this technological advancement lies in the precise formation of the urea intermediate (Formula IV) and its subsequent cyclization. The process begins with the reaction of a beta-amino-beta-(perfluoroalkyl)acrylate (Formula II) with a carbamoyl chloride (Formula III). This step is typically conducted in a polar aprotic solvent such as N,N-dimethylformamide (DMF) at temperatures ranging from -20°C to 80°C. The use of bases like sodium hydride or potassium tert-butoxide ensures the deprotonation of the amino group, facilitating nucleophilic attack on the carbamoyl chloride. This mechanism is crucial for impurity control, as the mild conditions prevent the decomposition of the sensitive perfluoroalkyl chain. The resulting urea compound is isolated with high fidelity, setting the stage for the ring-closing step. The structural integrity of the perfluoroalkyl group is maintained throughout, which is vital for the biological activity of the final herbicide product.
Following the formation of the urea intermediate, the cyclization to the 1,3-oxazin-6-one (Formula I) is achieved using dehydrating agents such as phosphorus pentachloride or oxalyl chloride, often in a solvent like phosphorus oxychloride. This step involves the activation of the urea carbonyl and subsequent intramolecular nucleophilic attack by the ester oxygen. The reaction temperatures are carefully controlled, preferably between 20°C and 50°C, to maximize yield while minimizing side reactions. This specific mechanistic pathway allows for the introduction of diverse N,N-disubstituted groups, providing flexibility in tuning the physicochemical properties of the intermediate. For technical teams, understanding these parameters is key to replicating the high yields reported in the patent examples, such as the 93.8% yield observed in the cyclization of specific trifluoromethyl derivatives. This level of control ensures consistent quality for high-purity agrochemical intermediates.
How to Synthesize 6-(Perfluoroalkyl)uracil Efficiently
The synthesis of the final herbicide active ingredient involves converting the oxazinone intermediate into the target 6-(perfluoroalkyl)uracil (Formula V). This transformation is achieved by reacting the oxazinone with an appropriate amine compound in the presence of an acid or base catalyst. The versatility of this step allows for the creation of a wide library of derivatives by varying the amine component. Detailed standardized synthesis steps see the guide below.
- React beta-amino-beta-(perfluoroalkyl)acrylate with carbamoyl chloride in the presence of a base to form the urea intermediate.
- Cyclize the urea compound using phosphorus pentahalide or oxalyl halide to generate the 1,3-oxazin-6-one core structure.
- Convert the oxazinone to the final 6-(perfluoroalkyl)uracil herbicide by reacting with an appropriate amine compound under acidic or basic conditions.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of the process described in CN1344259A offers distinct advantages for procurement managers and supply chain heads. The primary benefit stems from the substitution of expensive and hazardous reagents with more commodity-grade chemicals. This shift fundamentally alters the cost structure of the manufacturing process, leading to significant cost savings without compromising on quality. Moreover, the stability of the intermediates reduces the risk of batch failures, thereby enhancing supply chain reliability. For organizations focused on reducing lead time for high-purity agrochemical intermediates, this streamlined process eliminates several purification bottlenecks associated with older technologies. The robustness of the reaction conditions also supports scalability, ensuring that production can be ramped up to meet global demand for effective herbicides.
- Cost Reduction in Manufacturing: The elimination of imidoyl chloride hydrochloride compounds removes a major cost driver from the bill of materials. These legacy reagents often require specialized handling and storage infrastructure, adding indirect costs to the production budget. By switching to carbamoyl chlorides, which are more stable and widely available, manufacturers can achieve substantial cost savings. Furthermore, the higher yields reported in the patent examples mean less raw material waste per kilogram of final product. This efficiency gain compounds over large production runs, directly improving the gross margin for the final herbicide product. The qualitative improvement in process economics makes this route highly attractive for cost-sensitive agrochemical manufacturing.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the availability of niche reagents. The reagents utilized in this novel process, such as beta-amino-acrylates and common phosphorus halides, are produced by multiple suppliers globally. This diversification of the supply base mitigates the risk of shortages that can halt production lines. Additionally, the stability of the urea intermediate allows for potential stockpiling or transport between different manufacturing sites if necessary. This flexibility is crucial for maintaining consistent delivery schedules to downstream formulators. By adopting this method, companies can secure a more resilient supply chain for their agrochemical intermediate needs, ensuring that market demand is met without interruption.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new environmental and safety challenges. This methodology operates under relatively mild conditions and avoids the generation of excessive hazardous waste associated with unstable chlorinating agents. The use of standard solvents like DMF and ethyl acetate simplifies waste treatment and solvent recovery protocols. This alignment with environmental compliance standards reduces the regulatory burden on manufacturing facilities. Furthermore, the straightforward workup procedures, often involving simple aqueous washes and crystallization, are easily adaptable to large-scale reactors. This ease of scale-up ensures that the transition from laboratory bench to commercial plant is smooth, enabling rapid deployment of new herbicide products to the market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation. They provide clarity on the practical aspects of adopting this route for industrial production. Understanding these details helps stakeholders make informed decisions about integrating this chemistry into their existing portfolios.
Q: What is the primary advantage of this synthesis method over conventional routes?
A: The primary advantage is the avoidance of imidoyl chloride hydrochloride compounds, which are difficult to handle and expensive. This new process utilizes readily available carbamoyl chlorides, significantly simplifying the supply chain and reducing raw material costs.
Q: How does this process ensure high purity for agrochemical applications?
A: The process employs specific solvent systems like N,N-dimethylformamide and phosphorus oxychloride which facilitate clean cyclization. Additionally, the intermediate urea compounds can be purified via standard extraction and chromatography, ensuring stringent purity specifications for the final herbicide active ingredients.
Q: Is this methodology scalable for commercial production volumes?
A: Yes, the reaction conditions operate within moderate temperature ranges (e.g., 0°C to 100°C) and utilize common industrial solvents. The elimination of hazardous and unstable reagents enhances operational safety, making the commercial scale-up of complex herbicides more feasible and reliable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-(Perfluoroalkyl)uracil Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise to leverage advanced synthetic routes like CN1344259A for the production of high-value agrochemical intermediates. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 6-(perfluoroalkyl)uracil meets the highest industry standards. Our commitment to quality assurance means that you receive intermediates that are ready for immediate formulation, minimizing your internal testing overhead.
We invite you to collaborate with us to optimize your herbicide supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term business goals. Let us be your partner in delivering high-performance agrochemical solutions to the global market.
