Industrial Scale-Up of High-Purity Diacerein via Novel Zinc-Catalyzed Acetylation
The pharmaceutical landscape for osteoarthritis treatment has long relied on Interleukin-1 (IL-1) inhibitors, with Diacerein standing out as a critical slow-acting drug. However, the manufacturing of this anthraquinone derivative has historically been plagued by complex multi-step syntheses and the use of hazardous heavy metal oxidants. Patent CN103360250A introduces a transformative approach to Diacerein synthesis, shifting the paradigm from toxic oxidation protocols to a streamlined, high-yield acetylation strategy. By utilizing domestic high-purity Rhein (often referred to as Rhubarb Acid) as the starting material and employing anhydrous Zinc Chloride as a Lewis acid catalyst, this method achieves exceptional conversion rates under moderate thermal conditions. For global procurement teams and R&D directors, this patent represents a significant opportunity to secure a reliable diacerein supplier capable of delivering material with superior purity profiles and a drastically simplified environmental footprint compared to legacy processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Diacerein has been hindered by two primary synthetic bottlenecks that compromise both cost efficiency and environmental compliance. The first conventional route involves a total chemical synthesis starting from 3-nitrophthalic anhydride and meta-cresol. As illustrated in the reaction scheme below, this pathway requires a tedious sequence of Friedel-Crafts acylation, reduction, cyclization, diazotization, and finally oxidation. This multi-step trajectory not only extends the production cycle significantly but also accumulates impurities at every stage, resulting in lower overall yields and higher purification costs.
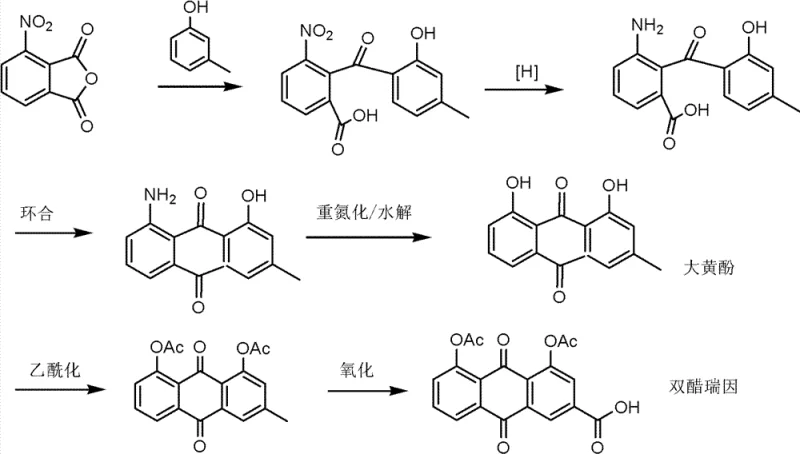
The second prevalent method, a semi-synthetic approach, attempts to shorten the route by starting from natural anthraquinone derivatives like Rhabarberone or Chrysophanol. However, as depicted in the following figure, this method critically depends on Chromium-based reagents to oxidize the methyl group to a carboxylic acid functionality. The reliance on hexavalent chromium introduces severe toxicity risks, necessitating expensive waste treatment protocols to prevent environmental contamination. Furthermore, chromium residues are notoriously difficult to remove completely from the final API, posing a significant regulatory risk for pharmaceutical manufacturers aiming for strict heavy metal limits.
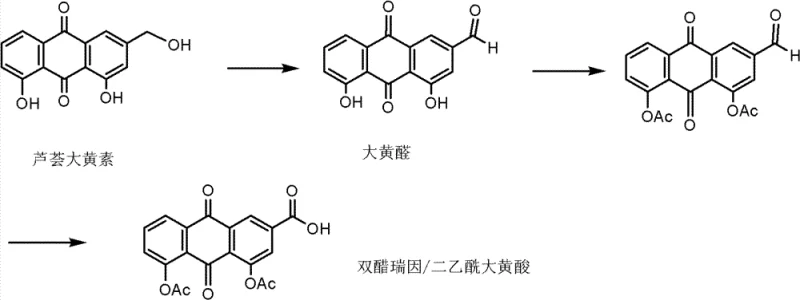
The Novel Approach
In stark contrast to these legacy methods, the novel process disclosed in the patent bypasses the need for oxidation entirely by selecting Rhein as the direct precursor. The core innovation lies in a one-step acetylation reaction where acetic anhydride serves a dual role as both the solvent and the acylating agent. Catalyzed by a mere stoichiometric fraction of anhydrous Zinc Chloride, the reaction proceeds smoothly at temperatures between 85°C and 90°C. This approach, visualized in the schematic below, eliminates the generation of toxic chromium waste and reduces the number of unit operations. The result is a robust process that delivers crude Diacerein with high content, which is subsequently refined to pharmaceutical grade through a sophisticated recrystallization technique.

Mechanistic Insights into ZnCl2-Catalyzed Acetylation
The efficacy of this synthesis hinges on the precise activation of the acetic anhydride by the Zinc Chloride catalyst. As a strong Lewis acid, anhydrous ZnCl2 coordinates with the carbonyl oxygen of the acetic anhydride, increasing the electrophilicity of the carbonyl carbon. This activation facilitates a nucleophilic attack by the phenolic hydroxyl groups located at the 1 and 8 positions of the Rhein anthraquinone nucleus. Unlike harsher acidic conditions that might degrade the sensitive quinone structure or cause polymerization, the ZnCl2 system provides a controlled environment that favors O-acylation over C-acylation or ring degradation. The reaction temperature is maintained strictly between 85°C and 90°C for approximately 6 hours, ensuring complete conversion of the di-hydroxy starting material into the di-acetoxy product without inducing thermal decomposition.
Impurity control in this mechanism is inherently superior due to the absence of oxidative side reactions. In chromium-mediated pathways, over-oxidation often leads to ring-opened byproducts or quinone-hydrate impurities that share similar polarity with the target molecule, making separation arduous. In the ZnCl2-catalyzed route, the primary impurities are typically mono-acetylated intermediates or unreacted Rhein. These impurities exhibit distinct solubility profiles compared to the di-acetylated Diacerein, particularly in the specific NMP/MEK solvent system used for refining. This chemical orthogonality allows for the efficient exclusion of related substances during the crystallization phase, ensuring the final product meets stringent pharmacopoeial standards for single impurities (typically <0.5%) and total impurities (<1.5%).
How to Synthesize Diacerein Efficiently
The operational simplicity of this patent makes it highly attractive for technology transfer. The process begins with the precise weighing of high-purity Rhein, anhydrous Zinc Chloride, and acetic anhydride into a glass-lined or stainless steel reactor equipped with mechanical stirring. The mixture is heated gradually to the target range of 85-90°C and held for 6 hours to ensure reaction completion. Upon cooling to ambient temperature (25-30°C), the product precipitates and is isolated via suction filtration. The resulting filter cake is then subjected to vacuum drying at 75-80°C to yield the crude intermediate. For the critical refining step, the crude material is dissolved in a heated mixture of NMP and MEK, filtered while hot to remove particulates, and then slowly cooled to induce crystallization of the pure API.
- Acetylation Reaction: React high-purity Rhein with acetic anhydride using anhydrous Zinc Chloride as a catalyst at 85-90°C for approximately 6 hours.
- Crude Isolation: Cool the reaction mixture to 25-30°C, perform suction filtration, and vacuum dry the filter cake at 75-80°C to obtain crude Diacerein.
- Purification: Recrystallize the crude product using a mixed solvent system of NMP and MEK, involving hot filtration followed by controlled cooling crystallization to achieve >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ZnCl2-catalyzed route offers tangible strategic benefits that extend beyond simple yield improvements. The elimination of chromium reagents fundamentally alters the cost structure of Diacerein manufacturing by removing the need for specialized hazardous waste disposal and complex heavy metal scavenging steps. This translates to a significantly reduced operational expenditure (OPEX) per kilogram of produced API. Furthermore, the use of acetic anhydride as a reaction solvent simplifies the supply chain, as it is a commodity chemical available globally in bulk quantities, reducing the risk of raw material shortages that often plague specialty reagent-dependent processes.
- Cost Reduction in Manufacturing: The economic advantage of this process is driven by the drastic simplification of the post-reaction workup. Traditional methods requiring chromium oxidation necessitate expensive quenching agents and multiple extraction steps to ensure metal clearance, which consumes significant labor and utility resources. By contrast, the novel method relies on a straightforward filtration and a recyclable solvent system for purification. The ability to recover and reuse solvents like MEK and NMP further drives down the variable cost of goods sold (COGS), allowing for more competitive pricing in the generic pharmaceutical market without compromising margin.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the regulatory scrutiny surrounding toxic reagents. Facilities handling hexavalent chromium face stricter environmental audits and potential shutdowns, creating volatility in the supply of Diacerein produced via older routes. The ZnCl2-catalyzed method utilizes reagents with a much lower regulatory burden, ensuring consistent production uptime. Additionally, the starting material, Rhein, is readily available from natural sources or established synthetic suppliers, providing a stable upstream foundation that mitigates the risk of production delays caused by raw material scarcity.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen thermal or mixing challenges, but this acetylation reaction is thermally benign and operates at atmospheric pressure. The exotherm is manageable, and the viscosity of the reaction mixture remains low enough for efficient agitation in large-scale reactors (e.g., 2000L to 5000L). From an environmental perspective, the process aligns with Green Chemistry principles by avoiding persistent bio-accumulative toxins. This compliance facilitates easier permitting for new manufacturing lines and reduces the long-term liability associated with environmental remediation, making it a sustainable choice for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following technical queries address common concerns regarding the implementation and quality assurance of this synthesis route. Understanding these details is crucial for R&D teams evaluating the feasibility of adopting this technology for their own production lines or for qualifying new vendors. The answers are derived directly from the experimental data and stability studies presented in the patent documentation.
Q: How does the new Zinc Chloride method improve upon traditional Chromium-based oxidation routes?
A: The traditional semi-synthetic routes often rely on toxic Chromium reagents for oxidizing intermediates like Rhabarberone, leading to severe environmental pollution and difficult-to-remove heavy metal residues in the final API. The novel method described in patent CN103360250A bypasses oxidation entirely by starting directly from Rhein (Rhubarb Acid) and utilizing a mild Lewis Acid (ZnCl2) catalyzed acetylation. This eliminates the risk of Chromium contamination, simplifies waste treatment, and significantly reduces the production cycle time.
Q: What represents the critical quality attribute in the purification of Diacerein?
A: The critical quality attribute is the removal of unreacted mono-acetylated intermediates and residual solvents while maintaining the integrity of the anthraquinone nucleus. The patent specifies a dual-solvent recrystallization system using N-Methyl-2-pyrrolidone (NMP) and Methyl Ethyl Ketone (MEK). This specific solvent pairing allows for effective hot filtration to remove insoluble impurities, followed by a controlled cooling profile (from 55-60°C down to 0-5°C) that ensures the formation of large, high-purity yellow crystals with content exceeding 99.5%.
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the process is explicitly designed for industrial scalability. It utilizes common, commercially available solvents like acetic anhydride, NMP, and MEK, which can be recovered and recycled to lower costs. The reaction conditions (85-90°C) are moderate and do not require extreme pressure or cryogenic temperatures. Furthermore, the isolation steps involve standard filtration and vacuum drying operations, making the transition from laboratory gram-scale to multi-ton annual production straightforward and robust.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diacerein Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a superior synthetic route requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the ZnCl2-catalyzed process are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with HPLC and GC systems to monitor every batch, guaranteeing that our Diacerein consistently meets the high standards required for global regulatory submissions.
We invite pharmaceutical companies seeking to optimize their osteoarthritis drug supply chain to engage with us for a Customized Cost-Saving Analysis. Our technical procurement team is ready to provide specific COA data from recent pilot batches and detailed route feasibility assessments tailored to your volume requirements. By partnering with us, you secure not just a chemical intermediate, but a strategic alliance focused on long-term cost efficiency, supply security, and uncompromised quality.
