Advanced Chromium-Free Synthesis of Diacerein for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust manufacturing pathways for active pharmaceutical ingredients (APIs) that balance efficacy with environmental sustainability. Patent CN102826996A introduces a transformative synthetic methodology for Diacerein, a critical interleukin-1 beta inhibitor used in the treatment of osteoarthritis. This intellectual property details a novel two-step sequence that fundamentally shifts the oxidation paradigm from hazardous chromium-based reagents to a benign hydrogen peroxide system catalyzed by tungstates. By leveraging this technology, manufacturers can achieve superior control over reaction parameters while drastically mitigating the environmental footprint associated with heavy metal waste disposal. The strategic implementation of this process addresses long-standing pain points in the supply chain regarding toxicity management and regulatory compliance for anthraquinone derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Diacerein has relied heavily on stoichiometric amounts of chromium trioxide or similar hexavalent chromium oxidants to convert Aloe-emodin into Rhein. These traditional protocols present severe operational challenges, including the generation of vast quantities of toxic chromium-containing wastewater that requires complex and costly remediation before discharge. Furthermore, the aggressive nature of chromium oxidants often leads to over-oxidation side reactions, resulting in a complex impurity profile that necessitates rigorous and yield-diminishing purification steps. The difficulty in precisely controlling the oxidation state frequently leads to batch-to-batch variability, complicating the validation processes required for pharmaceutical grade materials. Additionally, the residual chromium levels in the final API must be strictly monitored and reduced to trace parts per million, adding significant downstream processing costs and analytical burdens to the manufacturing workflow.
The Novel Approach
In stark contrast, the methodology disclosed in the patent employs a catalytic oxidation system utilizing inexpensive sodium tungstate and aqueous hydrogen peroxide, offering a markedly cleaner and more controllable alternative. This green chemistry approach operates under mild reflux conditions in polar aprotic solvents, ensuring high selectivity for the carboxylic acid formation without degrading the sensitive anthraquinone core. The reaction kinetics are easily managed by controlling the addition rate of the oxidant, which prevents thermal runaways and ensures consistent conversion rates across large-scale batches. By eliminating the need for toxic heavy metals, the process inherently simplifies the workup procedure, as there is no requirement for specialized heavy metal scavenging resins or complex extraction protocols to meet regulatory limits. This streamlined workflow not only enhances the overall yield but also significantly reduces the ecological impact of the manufacturing facility.
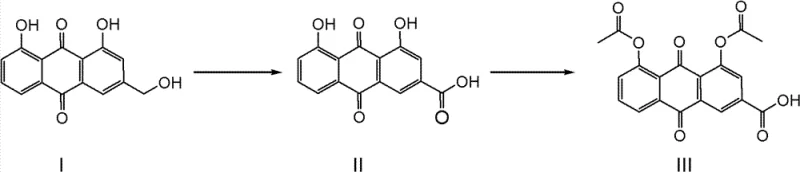
Mechanistic Insights into Tungstate-Catalyzed Oxidation
The core innovation lies in the activation of hydrogen peroxide by the tungstate catalyst to form a peroxotungstate species, which acts as the active oxygen transfer agent. This mechanism allows for the selective oxidation of the hydroxymethyl group on the Aloe-emodin scaffold to a carboxylic acid functionality while preserving the phenolic hydroxyl groups at the 1 and 8 positions. The catalytic cycle is highly efficient, requiring only minimal loading of the tungsten species, typically between 0.5wt% and 2wt% relative to the substrate, to drive the reaction to completion. The use of solvents like N,N-Dimethylformamide (DMF) or N-Methyl pyrrolidone (NMP) is critical, as they provide the necessary solubility for the starting material at elevated temperatures around 80°C to 100°C, facilitating homogeneous reaction conditions. This homogeneity is key to preventing localized hot spots that could lead to decomposition, thereby ensuring a narrow particle size distribution and consistent crystal morphology in the precipitated Rhein intermediate.
Impurity control is intrinsically built into this mechanistic pathway through the precise modulation of reaction temperature and oxidant stoichiometry. The patent data indicates that maintaining the reaction temperature at the solvent reflux point while slowly dripping the hydrogen peroxide solution minimizes the formation of over-oxidized byproducts or quinone degradation products. Analytical monitoring via HPLC confirms that the residual starting material, Aloe-emodin, can be consistently controlled below 0.5% in the crude isolate, a specification that is difficult to achieve with non-catalytic oxidants. This high level of crude purity means that the intermediate Rhein can often proceed directly to the acetylation step without intermediate recrystallization, saving both time and solvent consumption. The final acetylation step, utilizing acetic anhydride under acidic or basic catalysis, proceeds smoothly on this high-quality intermediate to yield Diacerein with a purity exceeding 99%, demonstrating the robustness of the entire synthetic sequence.
How to Synthesize Diacerein Efficiently
The synthesis of Diacerein via this patented route involves a straightforward sequence that begins with the suspension of Aloe-emodin in a suitable high-boiling polar solvent. Following the addition of the tungstate catalyst, the mixture is heated to initiate dissolution before the controlled introduction of the oxidant. The reaction progress is monitored to ensure full conversion before isolating the Rhein intermediate, which is then subjected to standard acetylation conditions. For detailed operational parameters, specific stoichiometric ratios, and safety protocols, please refer to the standardized synthesis guide below.
- Suspend Aloe-emodin in a polar aprotic solvent such as DMF or NMP and add a catalytic amount of sodium tungstate.
- Heat the mixture to 80°C and slowly add 30% hydrogen peroxide while maintaining the temperature at reflux until conversion is complete.
- Filter the resulting Rhein crude product, wash, and subject it to acetylation using acetic anhydride to obtain high-purity Diacerein.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this chromium-free synthesis route offers substantial strategic benefits that extend beyond mere regulatory compliance. The replacement of expensive and hazardous chromium reagents with commodity chemicals like hydrogen peroxide and sodium tungstate drastically reduces the raw material cost base and mitigates supply risk associated with regulated heavy metals. Furthermore, the elimination of toxic waste streams significantly lowers the operational expenditure related to environmental health and safety (EHS) management, waste treatment, and disposal fees. This process intensification allows for faster batch turnover times due to simplified workup procedures, thereby enhancing the overall throughput capacity of existing manufacturing assets without the need for major capital investment in new equipment.
- Cost Reduction in Manufacturing: The economic advantages are driven primarily by the substitution of high-cost oxidants and the removal of downstream purification steps. By avoiding chromium, manufacturers eliminate the need for expensive metal scavengers and the associated labor for handling hazardous materials. The high selectivity of the reaction minimizes yield losses typically incurred during extensive recrystallization processes required to remove chromium traces. Additionally, the use of recyclable solvents and the potential to skip intermediate purification steps for high-quality crude Rhein contribute to a leaner cost structure. These factors collectively result in a more competitive pricing model for the final API, providing a distinct margin advantage in the global market.
- Enhanced Supply Chain Reliability: The reliance on widely available and stable reagents such as hydrogen peroxide and sodium tungstate ensures a resilient supply chain that is less susceptible to geopolitical disruptions or regulatory bans on specific chemicals. Unlike chromium trioxide, which faces increasing global restrictions and transportation hurdles due to its toxicity classification, the reagents in this process are standard industrial commodities with robust supply networks. This stability guarantees continuous production capabilities and reduces the risk of unplanned shutdowns caused by raw material shortages. Moreover, the simplified logistics of handling non-hazardous oxidants streamline the inbound supply chain operations and reduce insurance and storage costs.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction conditions that are easily translated from laboratory to multi-ton production scales without significant engineering challenges. The absence of heavy metal contamination simplifies the environmental permitting process for new manufacturing lines and facilitates easier renewal of existing permits in regions with strict effluent standards. Waste streams generated are primarily aqueous and organic solvents which are easier to treat biologically or recover compared to heavy metal sludge. This alignment with green chemistry principles not only future-proofs the manufacturing site against tightening environmental regulations but also enhances the corporate sustainability profile appealing to eco-conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. These insights are derived directly from the experimental data and claims within the patent documentation to assist technical teams in evaluating feasibility. Understanding these nuances is crucial for successful technology transfer and process validation.
Q: How does this process eliminate chromium contamination?
A: Unlike conventional methods using chromium trioxide, this patent utilizes a hydrogen peroxide and tungstate catalyst system, completely avoiding toxic heavy metal residues and simplifying purification.
Q: What is the purity profile of the intermediate Rhein?
A: The oxidation step yields Rhein crude with a purity exceeding 98%, with residual Aloe-emodin controlled below 0.5%, allowing for direct use in the subsequent acetylation step without extensive recrystallization.
Q: Which solvents are preferred for the oxidation step?
A: Polar aprotic solvents such as N,N-Dimethylformamide (DMF), N-Methyl pyrrolidone (NMP), or DMSO are preferred due to their ability to dissolve the starting material effectively at elevated temperatures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diacerein Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate this advanced patent technology into commercial reality for our global partners. Our R&D team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Diacerein meets the highest pharmacopoeial standards. Our commitment to quality assurance ensures that the impurity profiles are tightly controlled, specifically monitoring for residual starting materials and oxidation byproducts.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific volume requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this greener manufacturing process. We encourage you to contact us to obtain specific COA data and route feasibility assessments that demonstrate our capability to deliver high-purity Diacerein with reliable lead times and competitive commercial terms.
