Advanced Two-Step Oxidation Strategy for High-Purity Diacerein Production and Commercial Scale-Up
The pharmaceutical industry constantly seeks robust synthetic routes that balance high purity with environmental sustainability, particularly for active pharmaceutical ingredients (APIs) used in chronic conditions like osteoarthritis. Patent CN101104583A introduces a transformative methodology for the preparation of diacerein, a potent interleukin-1 inhibitor, by employing a strategic two-step oxidation sequence. This innovation addresses the critical limitations of prior art, which relied heavily on violent oxidation conditions using toxic chromium reagents that were difficult to control and prone to generating complex impurity profiles. By decoupling the oxidation of the benzyl alcohol and the protection of the phenolic hydroxyl groups, this new process allows for the use of milder, more selective oxidants in the initial stages. This technical breakthrough not only enhances the chemical integrity of the anthraquinone core but also paves the way for a more sustainable manufacturing protocol that is highly attractive to modern regulatory frameworks. For stakeholders evaluating reliable pharmaceutical intermediates supplier partnerships, understanding this shift towards controlled, step-wise functionalization is crucial for ensuring long-term supply stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of diacerein has been plagued by the reliance on harsh oxidizing agents such as potassium bichromate or chromic anhydride to directly convert the benzyl hydroxyl group of aloe-emodin into a carboxylic acid. As illustrated in the traditional pathway, this direct oxidation often proceeds under violent conditions that lack selectivity, leading to the undesirable over-oxidation of the sensitive hydroxyl groups located on the anthraquinone ring. 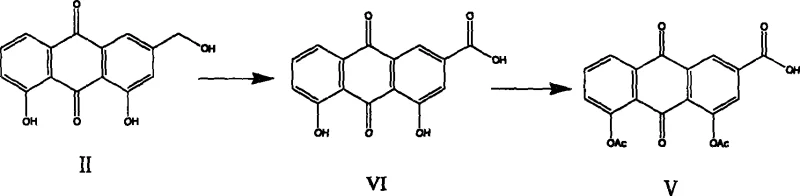 The lack of control in these reactions necessitates the use of large excesses of toxic chromium reagents, creating significant challenges in waste management and labor safety. Furthermore, the resulting crude products typically contain a complex mixture of over-oxidized byproducts and unreacted starting materials, making the subsequent purification steps arduous and yield-limiting. The environmental burden of disposing of chromium-containing waste streams has become a major bottleneck for manufacturers aiming to scale up production while adhering to green chemistry principles. Consequently, the overall process efficiency is compromised, and the final product quality often struggles to meet the stringent purity specifications required for therapeutic applications without extensive and costly recrystallization procedures.
The lack of control in these reactions necessitates the use of large excesses of toxic chromium reagents, creating significant challenges in waste management and labor safety. Furthermore, the resulting crude products typically contain a complex mixture of over-oxidized byproducts and unreacted starting materials, making the subsequent purification steps arduous and yield-limiting. The environmental burden of disposing of chromium-containing waste streams has become a major bottleneck for manufacturers aiming to scale up production while adhering to green chemistry principles. Consequently, the overall process efficiency is compromised, and the final product quality often struggles to meet the stringent purity specifications required for therapeutic applications without extensive and costly recrystallization procedures.
The Novel Approach
In stark contrast to the brute-force methods of the past, the novel approach detailed in the patent utilizes a sophisticated two-step oxidation strategy that fundamentally alters the reaction landscape. The process begins by gently oxidizing the benzyl alcohol of aloe-emodin to an aldehyde intermediate using mild oxidants, thereby preserving the integrity of the phenolic hydroxyls on the aromatic rings. Once the aldehyde is formed, these sensitive hydroxyl groups are protected via acetylation, creating a stable intermediate that shields the core structure from degradation during the final oxidation step. This sequential logic ensures that when the aldehyde is finally oxidized to the carboxylic acid to yield diacerein, the reaction environment is far more controlled and selective. By avoiding the initial exposure of the unprotected anthraquinone ring to strong oxidizers, the generation of impurities is drastically minimized. This methodological shift represents a significant advancement in cost reduction in API manufacturing, as it simplifies downstream processing and improves the overall mass balance of the synthesis.
Mechanistic Insights into Two-Step Oxidation and Protection Strategy
The core mechanistic advantage of this process lies in the precise selection of oxidants tailored to specific functional group transformations. In the first step, the conversion of aloe-emodin to rheinaldehyde is achieved using reagents such as Pyridinium Chlorochromate (PCC), Manganese Dioxide, or even chrome-free alternatives like IBX and Dess-Martin periodinane. These reagents operate under much milder conditions, typically at temperatures ranging from room temperature to reflux in solvents like dichloroethane or acetone, preventing the radical-mediated degradation of the anthraquinone skeleton. The ability to substitute toxic chromium(VI) reagents with manganese(IV) or hypervalent iodine species in this step is a critical design feature that reduces the heavy metal load early in the synthesis. Following this, the acetylation of the phenolic hydroxyls using acetic anhydride and a base like sodium acetate creates an electron-withdrawing environment that further stabilizes the molecule. This protection is essential because it prevents the competing oxidation of the phenolic groups during the final step, where the aldehyde is converted to the carboxylic acid using oxidants like potassium permanganate or sodium chlorite.
From an impurity control perspective, this stepwise approach effectively isolates the reactive sites, ensuring that the oxidation potential is directed solely at the benzylic position. In conventional one-pot or direct oxidation methods, the simultaneous presence of free phenols and benzylic alcohols creates a competitive scenario where the oxidant attacks multiple sites, leading to quinone formation or ring cleavage byproducts. By masking the phenols as acetates before the final oxidation, the electronic density of the ring is modulated, rendering it less susceptible to electrophilic attack by the oxidant. This results in a crude product profile that is significantly cleaner, with the major impurity being primarily unreacted aldehyde rather than structurally degraded anthraquinone derivatives. Such purity profiles are vital for meeting the rigorous standards of high-purity diacerein required for clinical use, as it reduces the burden on analytical quality control and minimizes the risk of genotoxic impurities carrying through to the final drug substance.
How to Synthesize Diacerein Efficiently
The implementation of this synthesis route requires careful attention to reaction monitoring and solvent selection to maximize yield and purity. The process is designed to be flexible, allowing for the use of various solvents such as DMF, DMSO, or toluene depending on the solubility of the intermediates. Detailed standard operating procedures for the oxidation, acetylation, and final purification steps are critical for reproducibility. For a comprehensive breakdown of the specific reaction conditions, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below.
- Oxidize aloe-emodin (rhabarberone) to rheinaldehyde using mild oxidants like PCC, MnO2, or IBX to prevent over-oxidation of the anthraquinone ring.
- Protect the phenolic hydroxyl groups of rheinaldehyde via acetylation using acetic anhydride and a base like sodium acetate to form the aldehyde intermediate.
- Oxidize the protected aldehyde intermediate to the final carboxylic acid (diacerein) using oxidants such as potassium permanganate or sodium chlorite.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers substantial strategic benefits beyond mere technical elegance. The primary advantage lies in the drastic simplification of the waste treatment infrastructure required for production. By enabling the use of chrome-free oxidants in the initial steps and reducing the overall consumption of heavy metals, the facility's environmental compliance burden is significantly lightened. This translates directly into lower operational expenditures related to hazardous waste disposal and wastewater treatment, which are often hidden costs in traditional chemical manufacturing. Furthermore, the improved selectivity of the reaction means that fewer raw materials are wasted on forming byproducts, leading to a more efficient utilization of starting materials like aloe-emodin. This efficiency gain supports a more resilient supply chain by reducing the dependency on bulk quantities of hazardous reagents that may face regulatory scrutiny or supply volatility.
- Cost Reduction in Manufacturing: The elimination of excessive chromium reagents and the simplification of purification steps lead to significant cost optimizations. Traditional methods require large excesses of expensive and toxic oxidants, along with complex downstream processing to remove heavy metal residues. By switching to milder, more selective oxidants and a protective group strategy, the process reduces the consumption of auxiliary chemicals and solvents. Additionally, the higher purity of the crude product minimizes the number of recrystallization cycles needed, saving both time and energy. This streamlined workflow ensures that the commercial scale-up of complex pharmaceutical intermediates becomes more economically viable, offering a competitive pricing structure for the final API.
- Enhanced Supply Chain Reliability: The flexibility in oxidant selection provides a robust buffer against raw material shortages. Since the process can utilize a range of oxidants including manganese dioxide, IBX, or sodium chlorite, manufacturers are not locked into a single supply source for critical reagents. This diversification mitigates the risk of production stoppages due to supply chain disruptions. Moreover, the milder reaction conditions reduce the wear and tear on reactor equipment and lower the safety risks associated with handling violent exothermic reactions. This operational stability ensures consistent batch-to-batch quality and reliable delivery schedules, which is essential for reducing lead time for high-purity pharmaceutical intermediates in a just-in-time manufacturing environment.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, but this two-step oxidation method is inherently safer for large-scale production. The avoidance of violent oxidation conditions reduces the risk of thermal runaways, making the process easier to control in large reactors. From an environmental standpoint, the potential to operate a fully chrome-free or low-chromium process aligns with increasingly stringent global environmental regulations. This proactive compliance future-proofs the manufacturing site against tightening emission standards and avoids potential fines or shutdowns. The ability to produce high-quality diacerein with a reduced environmental footprint enhances the corporate sustainability profile, a key factor for modern pharmaceutical partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced diacerein synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits for potential partners.
Q: Why is the two-step oxidation method superior to direct oxidation for diacerein synthesis?
A: Direct oxidation using strong agents like potassium bichromate often leads to over-oxidation of the anthraquinone ring hydroxyls and generates significant toxic chromium waste. The two-step method first converts the benzyl alcohol to an aldehyde under mild conditions, protecting the sensitive ring hydroxyls via acetylation before the final oxidation, resulting in higher purity and easier control.
Q: What oxidants are recommended for the initial oxidation step in this novel process?
A: The patent suggests using mild oxidants such as Pyridinium Chlorochromate (PCC), Manganese Dioxide, IBX (2-Iodoxybenzoic acid), or Dess-Martin periodinane. Chrome-free options like IBX and Manganese Dioxide are particularly preferred for environmental compliance and waste reduction.
Q: How does this process impact the environmental footprint of diacerein manufacturing?
A: By enabling the use of chrome-free oxidants and avoiding the massive excess of chromic anhydride required in traditional methods, this process significantly reduces toxic heavy metal waste. This simplifies wastewater treatment and aligns with stricter environmental regulations for pharmaceutical production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diacerein Supplier
The technological advancements outlined in patent CN101104583A represent a significant leap forward in the manufacturing of anti-arthritis therapeutics, offering a pathway to higher purity and greater environmental sustainability. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate these innovative laboratory protocols into robust industrial realities. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the nuances of mild oxidation and protection chemistry are perfectly maintained at scale. We operate with stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of diacerein meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this superior synthetic route for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this greener, more efficient process. Please contact us to request specific COA data and route feasibility assessments tailored to your volume requirements, and let us demonstrate how our commitment to innovation can drive value for your organization.
