Advanced Synthesis of Selective PDE IV Inhibiting Pyridine Derivatives for Respiratory Therapeutics
The pharmaceutical landscape for respiratory diseases, particularly asthma, has long sought agents that provide potent bronchodilation without the debilitating cardiac side effects associated with traditional treatments. Patent CN1127498C introduces a groundbreaking class of novel pyridine derivatives that exhibit selective inhibitory activity against phosphodiesterase IV (PDE IV). Unlike non-specific inhibitors, these compounds effectively suppress bronchoconstriction and airway inflammation while demonstrating a significantly improved safety profile regarding cardiac function. The core innovation lies in the specific structural arrangement where a pyridine ring is substituted with complex nitrogen-containing heterocyclic fused rings, creating a highly targeted therapeutic agent. As a reliable pharmaceutical intermediates supplier, understanding the depth of this chemical architecture is crucial for developing next-generation respiratory medications.
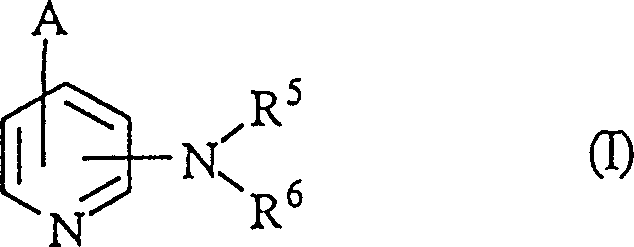
The development of these compounds addresses a critical gap in asthma therapy. Historically, agents like theophylline have been used as PDE inhibitors, but their lack of specificity leads to unwanted cardiotonic and central nervous system activities. This necessitates careful dosing and monitoring, limiting their clinical utility. The new derivatives described in the patent overcome these limitations by selectively targeting PDE IV, which is predominantly found in bronchial smooth muscle and inflammatory cells. This selectivity ensures that the therapeutic effect is concentrated where it is needed most, reducing the risk of systemic toxicity. For procurement managers, this represents a shift towards higher-value active pharmaceutical ingredients (APIs) that command better market positioning due to their superior efficacy and safety margins.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Conventional anti-asthmatic agents often suffer from a narrow therapeutic index, meaning the difference between an effective dose and a toxic dose is small. Theophylline, for instance, inhibits multiple PDE isozymes, leading to side effects such as hypertension and palpitations. Furthermore, the synthesis of older generation compounds often involves harsh conditions or generates significant impurities that are difficult to remove, complicating the purification process. These factors contribute to higher manufacturing costs and longer lead times for high-purity respiratory drug intermediates. The inability to finely tune the molecular structure to achieve isoform selectivity has been a persistent challenge in medicinal chemistry, resulting in drugs that treat symptoms but carry substantial risks for patients with comorbidities.
The Novel Approach
The novel approach detailed in the patent utilizes a sophisticated modular synthesis strategy that allows for precise control over the molecular architecture. By employing specific condensation and cyclization reactions, manufacturers can construct the complex heterocyclic cores, such as isoquinoline and naphthyridine systems, with high fidelity. This method enables the introduction of diverse substituents at key positions, optimizing the interaction with the PDE IV enzyme active site. The result is a compound that not only inhibits bronchoconstriction more effectively than theophylline but also possesses anti-inflammatory properties. This dual mechanism of action enhances the overall therapeutic value, making cost reduction in respiratory drug manufacturing achievable through streamlined processes that yield high-purity products with fewer by-products.
Mechanistic Insights into Copper-Catalyzed Coupling and Cyclization
The synthesis of these advanced pyridine derivatives relies heavily on robust transition metal catalysis and precise cyclization protocols. A key step in the process, often referred to as Method A in the patent documentation, involves the coupling of a halogenated pyridine intermediate with a nitrogen-containing heterocycle. This reaction is typically conducted in the presence of a copper catalyst, such as copper(I) iodide or copper(0) powder, and a base like potassium carbonate or sodium hydride. The reaction proceeds in polar aprotic solvents such as dimethylformamide (DMF) or dimethylsulfoxide (DMSO) at elevated temperatures ranging from 80°C to 160°C, with an optimal range often observed between 120°C and 150°C. This thermal energy is essential to overcome the activation barrier for the carbon-nitrogen bond formation, ensuring high conversion rates and minimizing the formation of unreacted starting materials.
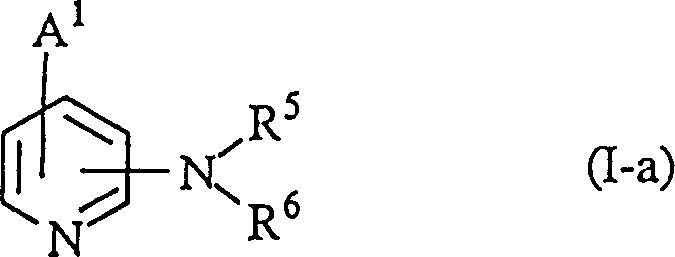
Another critical mechanistic pathway involves the construction of the isoquinoline or quinoline backbone, as illustrated in the synthesis of compound (II-a). This process begins with the condensation of an acetal compound with an amide derivative, followed by deprotection and subsequent cyclization. The use of protecting groups, such as lower alkyl or alkanoyl groups, is strategically employed to mask reactive hydroxyl or amino functionalities during intermediate steps, preventing unwanted side reactions. Once the core scaffold is established, these protecting groups are removed under mild acidic or basic conditions, revealing the active pharmacophore. This level of control over the synthetic pathway is vital for maintaining the integrity of the chiral centers often present in these molecules, which can significantly influence biological activity. Understanding these mechanistic details allows R&D teams to troubleshoot potential bottlenecks and optimize reaction parameters for commercial scale-up of complex heterocyclic intermediates.
How to Synthesize High-Purity Pyridine Derivatives Efficiently
The efficient synthesis of these target molecules requires a disciplined approach to reaction engineering and purification. The process generally initiates with the preparation of key intermediates through condensation reactions, followed by ring-closing steps to form the fused heterocyclic systems. Subsequent functionalization involves coupling reactions that link the heterocyclic core to the pyridine moiety. Each step must be carefully monitored to ensure the removal of catalysts and by-products, which is essential for meeting stringent purity specifications required for pharmaceutical applications. The detailed standardized synthesis steps see the guide below.
- Condense acetal compounds with nitrogen-containing amides to form ketone intermediates.
- Perform intramolecular cyclization or hydrazine reaction to construct the heterocyclic core such as isoquinoline or naphthyridine.
- Execute copper-catalyzed coupling with halogenated pyridines at 80-160°C to finalize the target molecule.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this synthetic route offers distinct logistical and economic benefits. The reliance on readily available starting materials, such as substituted benzaldehydes and pyridine derivatives, ensures a stable supply chain that is less susceptible to raw material shortages. Furthermore, the reaction conditions, while requiring elevated temperatures, utilize standard industrial solvents and catalysts that are easily sourced and managed within existing manufacturing infrastructure. This compatibility reduces the need for specialized equipment investments, thereby lowering the barrier to entry for production. The ability to produce these intermediates with high consistency supports continuous manufacturing models, enhancing overall supply chain reliability and reducing the risk of production delays.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for exotic or prohibitively expensive reagents, relying instead on commodity chemicals and standard transition metal catalysts. By optimizing the stoichiometry and recycling solvents where possible, manufacturers can achieve substantial cost savings. Additionally, the high selectivity of the reactions minimizes the generation of difficult-to-separate impurities, reducing the burden on downstream purification processes like chromatography. This efficiency translates directly into a lower cost of goods sold (COGS), making the final therapeutic more accessible to the market while maintaining healthy profit margins for producers.
- Enhanced Supply Chain Reliability: The robustness of the chemical transformations described ensures that production schedules can be met with high predictability. The intermediates generated are stable and can be stored or transported without significant degradation, providing flexibility in inventory management. This stability is crucial for mitigating risks associated with global logistics disruptions. By establishing a reliable supply of these critical intermediates, pharmaceutical companies can secure their production pipelines for finished dosage forms, ensuring that patients have uninterrupted access to life-saving asthma medications.
- Scalability and Environmental Compliance: The processes outlined are inherently scalable, moving seamlessly from laboratory benchtop to pilot plant and full commercial production. The use of contained reaction systems and standard workup procedures facilitates compliance with environmental, health, and safety (EHS) regulations. Waste streams are manageable and can be treated using conventional methods, reducing the environmental footprint of the manufacturing operation. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the corporate social responsibility profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel compounds. The answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these aspects is key to making informed decisions about sourcing and development strategies.
Q: Why are selective PDE IV inhibitors preferred over traditional anti-asthmatics?
A: Traditional agents like theophylline exhibit non-specific PDE inhibition, leading to severe cardiac side effects. Selective PDE IV inhibitors target bronchial smooth muscle specifically, minimizing cardiac risks while maintaining potent bronchoconstriction inhibitory activity.
Q: What are the key structural features of these novel pyridine derivatives?
A: The core structure involves a pyridine ring substituted with nitrogen-containing fused heterocycles such as isoquinoline or naphthyridine moieties. Specific substitution patterns at the 4-position of the pyridine ring and the presence of hydroxymethyl or alkoxy groups are critical for activity.
Q: How does the synthesis process ensure scalability for commercial production?
A: The process utilizes robust reaction conditions, such as copper-catalyzed coupling in common solvents like DMF at manageable temperatures (80-160°C), and avoids extremely sensitive reagents, facilitating easier scale-up from laboratory to industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyridine Derivatives Supplier
The potential of these selective PDE IV inhibitors to transform asthma treatment is immense, offering a safer and more effective alternative to legacy therapies. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of intermediates meets the highest international standards, ready for immediate integration into your drug development pipeline. We understand the critical nature of respiratory therapeutics and are dedicated to supporting your mission with high-quality chemical solutions.
We invite you to engage with our technical procurement team to discuss your specific requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our manufacturing capabilities can optimize your budget without compromising quality. We encourage you to reach out for specific COA data and route feasibility assessments tailored to your project timelines. Together, we can accelerate the delivery of innovative asthma treatments to patients worldwide, leveraging our expertise in complex heterocyclic synthesis to drive your success.
