Advanced Synthesis of Benzofuranone Derivatives for Commercial Scale-up and Supply Chain Stability
Advanced Synthesis of Benzofuranone Derivatives for Commercial Scale-up and Supply Chain Stability
The chemical industry is constantly seeking robust methodologies for synthesizing complex heterocyclic scaffolds that serve as critical building blocks for high-value agrochemicals and pharmaceuticals. Patent CN1096454C introduces a transformative process for the preparation of benzofuranone derivatives, specifically focusing on 3-(α-methoxy)methylenebenzofuran-2(3H)-one compounds and their subsequent conversion into phenoxypyrimidine structures. This technology addresses long-standing challenges in the field by replacing hazardous reagents with stable, commercially viable alternatives, thereby enhancing both operational safety and economic feasibility. For R&D directors and procurement specialists, understanding the nuances of this synthesis route is essential for securing a reliable agrochemical intermediate supplier capable of delivering consistent quality at scale. The patent outlines a versatile pathway that not only improves yield but also offers significant flexibility in managing isomeric ratios, a critical factor in downstream biological activity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-substituted benzofuran-2(3H)-ones relied heavily on methylation strategies involving diazomethane or sulfuric acid in methanol, as documented in earlier literature such as the Australian Journal of Chemistry (1973). These conventional approaches present severe drawbacks for modern industrial manufacturing, primarily due to the extreme toxicity and explosive nature of diazomethane, which necessitates expensive containment systems and rigorous safety protocols that drastically inflate operational costs. Furthermore, prior attempts to directly formylate benzofuran-2(3H)-one were largely unsuccessful, limiting the structural diversity accessible to chemists and forcing reliance on inefficient multi-step sequences. The inability to reliably produce high-purity intermediates without generating significant hazardous waste has traditionally constrained the supply chain, leading to volatility in availability and pricing for downstream manufacturers who depend on these specific scaffolds for herbicide and pharmaceutical production.
The Novel Approach
The methodology disclosed in CN1096454C represents a paradigm shift by utilizing trimethyl orthoformate in conjunction with acid anhydrides to achieve the desired methylation and functionalization under mild conditions. This novel approach eliminates the need for dangerous diazomethane, substituting it with stable reagents that are readily available in bulk quantities, thus facilitating cost reduction in agrochemical intermediate manufacturing. The process allows for the direct reaction of benzofuran-2(3H)-one or o-hydroxyphenylacetic acid derivatives to form the key 3-(α-methoxy)methylene intermediate with high efficiency. By adjusting reaction parameters such as temperature (preferably 90-130°C) and solvent choice, manufacturers can precisely control the formation of acetal versus acrylate isomers, ensuring the final product meets stringent specifications required for biological efficacy. This flexibility makes the process highly adaptable for commercial scale-up of complex heterocyclic intermediates.
Mechanistic Insights into Orthoformate-Mediated Cyclization and Coupling
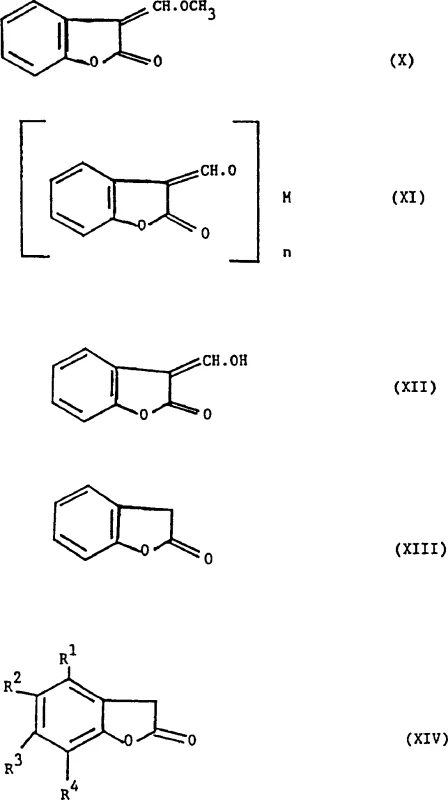
The core of this innovation lies in the mechanistic pathway where a compound of Formula (XIV) or (IX) reacts with trimethyl orthoformate, often in the presence of an activator like acetic anhydride, to generate the reactive 3-(α-methoxy)methylenebenzofuran-2(3H)-one (Formula X). This transformation likely proceeds through an activated ester intermediate that facilitates the introduction of the methoxymethylene group at the 3-position of the lactone ring. Subsequent reaction with a metal methoxide, such as sodium methoxide, generates a nucleophilic species that attacks electrophilic partners like 4,6-dichloropyrimidine (Formula III). This coupling step is critical for constructing the phenoxypyrimidine backbone found in many potent herbicides. The mechanism allows for the formation of a mixture of acetal and acrylate forms (Formula I), the ratio of which is kinetically and thermodynamically controlled by the solvent environment, providing a unique handle for process optimization that was previously unavailable in older synthetic routes.
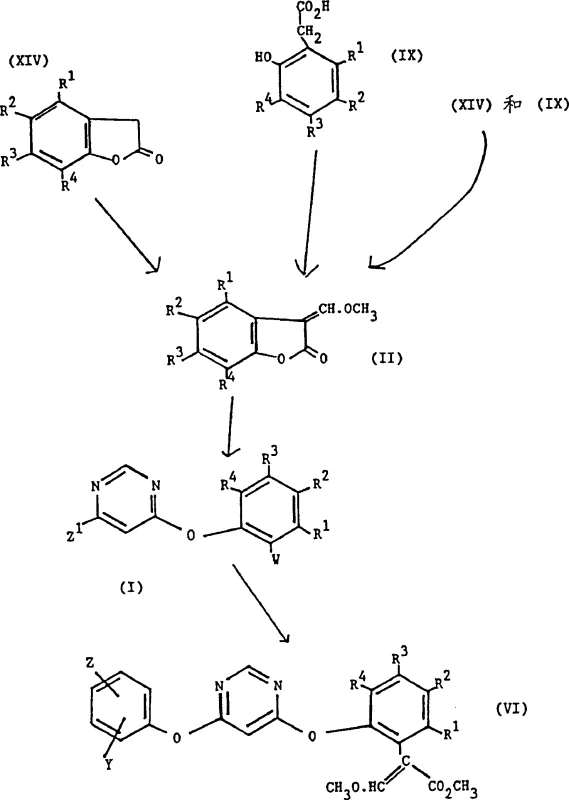
Impurity control is inherently built into this synthetic design through the ability to isolate and purify specific intermediates before the final coupling step. The patent describes methods to obtain substantially pure Formula (X) compounds from reaction mixtures containing acetals and acrylates by exploiting differences in solubility and reactivity towards aqueous base and acid. For instance, treating the mixture with aqueous sodium hydroxide converts the desired ketone into a water-soluble salt (Formula XI), separating it from neutral organic impurities, followed by acidification to regenerate the pure ketone. This purification strategy ensures that the final coupling reaction proceeds with minimal side reactions, resulting in a cleaner crude product that requires less intensive downstream processing. Such robust impurity management is vital for maintaining batch-to-batch consistency, a key metric for procurement managers evaluating potential suppliers for long-term contracts.
How to Synthesize 3-(α-methoxy)methylenebenzofuran-2(3H)-one Efficiently
Implementing this synthesis requires careful attention to reaction conditions to maximize yield and selectivity. The process generally involves heating the starting benzofuranone or acid precursor with trimethyl orthoformate and an anhydride at temperatures between 100-105°C, often utilizing a Dean-Stark apparatus to remove low-boiling by-products and drive the equilibrium forward. Following the formation of the key intermediate, it is reacted with a methoxide source and the appropriate halopyrimidine in solvents like tetrahydrofuran or methyl acetate. The detailed standardized synthesis steps see the guide below.
- Cyclize o-hydroxyphenylacetic acid derivatives or react benzofuran-2(3H)-one with trimethyl orthoformate and an acid anhydride at 90-130°C to form the key intermediate.
- React the resulting 3-(α-methoxy)methylenebenzofuran-2(3H)-one with a metal methoxide and a halopyrimidine derivative in a suitable solvent to form the coupled product.
- Control the acetal to acrylate ratio by selecting specific solvents like methyl acetate or toluene, and purify via crystallization or distillation.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this patent-protected methodology offers substantial strategic advantages over traditional manufacturing routes. By removing the dependency on diazomethane, facilities can operate with significantly reduced safety overheads, eliminating the need for specialized explosion-proof reactors and the associated high insurance premiums. This simplification of the safety profile translates directly into lower capital expenditure for new production lines and reduced operational risk for existing plants, ensuring greater supply continuity even during periods of strict regulatory scrutiny. Furthermore, the use of commodity chemicals like trimethyl orthoformate and acetic anhydride insulates the supply chain from the volatility associated with specialized, low-volume reagents, providing procurement teams with more predictable pricing and lead times.
- Cost Reduction in Manufacturing: The elimination of hazardous reagents removes the costly requirements for specialized containment and waste disposal associated with explosive materials, leading to substantial cost savings in overall production. Additionally, the ability to tune the isomer ratio through solvent selection reduces the need for expensive chromatographic separations, streamlining the purification process and improving overall material throughput. The one-pot potential of combining cyclization and orthoformate reaction steps further minimizes solvent usage and energy consumption, driving down the variable cost per kilogram of the final intermediate.
- Enhanced Supply Chain Reliability: Because the raw materials required for this process are bulk commodity chemicals produced by multiple global suppliers, the risk of raw material shortage is drastically minimized compared to processes relying on bespoke reagents. The robustness of the reaction conditions, which tolerate a range of temperatures and pressures, allows for flexible manufacturing scheduling and easier technology transfer between different production sites. This resilience ensures that downstream customers face fewer disruptions, making the supplier a more dependable partner for critical just-in-time manufacturing schedules.
- Scalability and Environmental Compliance: The process is designed for scalability, with examples demonstrating successful execution from gram scale to multi-kilogram batches without loss of efficiency. The avoidance of heavy metal catalysts in the initial steps and the use of recoverable solvents align with modern green chemistry principles, simplifying environmental permitting and waste treatment. This environmental compatibility future-proofs the supply chain against tightening global regulations on chemical emissions and hazardous waste, ensuring long-term viability of the manufacturing asset.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this benzofuranone synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for technical teams evaluating this route.
Q: Why is this new process safer than conventional diazomethane methods?
A: Conventional methods often rely on diazomethane, which is highly toxic and explosive. This patent utilizes trimethyl orthoformate and acid anhydrides, which are stable, commercially available reagents that eliminate the need for specialized explosion-proof infrastructure.
Q: Can the ratio of acetal to acrylate isomers be controlled?
A: Yes, the patent demonstrates that the ratio of acetal to acrylate forms depends heavily on the solvent nature. For instance, using methanol favors the acetal form, while solvents like pyridine or acetonitrile can shift the equilibrium, allowing manufacturers to tune the product profile.
Q: What represents the key quality metric for these intermediates?
A: High purity is critical, specifically achieving substantially pure states of the benzofuranone ketone (Formula X) with purity levels exceeding 85%, often achieved through specific base-acid workup sequences described in the purification protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzofuranone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your final products. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in CN1096454C can be translated into reliable industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of benzofuranone derivatives meets the exacting standards required for agrochemical and pharmaceutical applications, providing you with the confidence needed to plan your long-term production cycles.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how adopting this safer, more efficient methodology can optimize your total cost of ownership. Contact us today to obtain specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for complex heterocyclic intermediates.
