Advanced Catalytic Synthesis of Benzofuranone Derivatives for Industrial Scale-up
Advanced Catalytic Synthesis of Benzofuranone Derivatives for Industrial Scale-up
The pharmaceutical and agrochemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways to access complex heterocyclic scaffolds. A significant breakthrough in this domain is detailed in patent CN109438402B, which discloses a novel class of benzofuranone derivatives and their green catalytic synthesis method. This technology leverages a cross-dehydrogenative coupling (CDC) strategy, utilizing cuprous bromide as an economical catalyst and potassium persulfate as a robust oxidant. For R&D directors and procurement managers alike, this represents a pivotal shift away from expensive noble metal catalysis towards abundant earth-abundant copper systems. The method achieves high yields under mild conditions, specifically at 80°C in acetonitrile, offering a compelling value proposition for the manufacturing of high-purity pharmaceutical intermediates and agrochemical precursors such as azoxystrobin analogues.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of C-O bonds to form benzofuranone derivatives often relied on pre-functionalized starting materials, such as aryl halides or activated esters, which necessitate multi-step synthesis prior to the coupling event. These conventional routes frequently employ palladium or other precious metal catalysts that drive up the Bill of Materials (BOM) costs significantly and introduce stringent requirements for residual metal removal, a critical quality attribute for API intermediates. Furthermore, many classical methods require harsh reaction conditions, including high temperatures or strong bases, which can compromise the integrity of sensitive functional groups on the substrate. The generation of stoichiometric amounts of salt waste from leaving groups like halides also poses environmental challenges, conflicting with modern green chemistry principles and increasing the burden on waste treatment facilities for chemical manufacturers.
The Novel Approach
In stark contrast, the methodology described in CN109438402B utilizes a direct Cross-Dehydrogenative Coupling (CDC) reaction between benzofuranone and alcohols, bypassing the need for pre-functionalization entirely. This atom-economical approach directly activates C-H and O-H bonds to forge the new C-O linkage, drastically simplifying the synthetic route. The use of cuprous bromide (CuBr) as the catalyst and potassium persulfate (K2S2O8) as the terminal oxidant creates a highly efficient redox system that operates effectively in acetonitrile. As illustrated in the general reaction scheme below, this process accommodates a wide variety of substituents, allowing for the rapid generation of diverse derivative libraries.
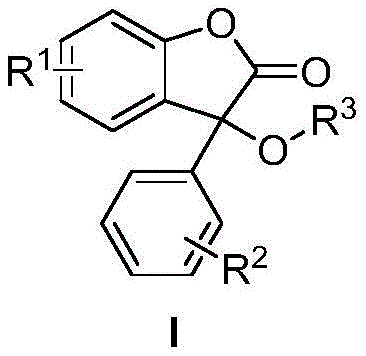
This structural versatility is crucial for medicinal chemists optimizing structure-activity relationships (SAR). By eliminating the need for halogenated precursors, the process not only reduces raw material costs but also minimizes the formation of inorganic salt byproducts, aligning perfectly with sustainability goals. The operational simplicity, requiring only standard heating at 80°C for 12 hours, makes this technology exceptionally attractive for reliable benzofuranone derivatives supplier networks aiming to streamline their production workflows.
Mechanistic Insights into Cu-Catalyzed Cross-Dehydrogenative Coupling
The success of this transformation hinges on the intricate interplay between the copper catalyst and the persulfate oxidant within the reaction medium. Mechanistically, the cycle likely initiates with the oxidation of Cu(I) to a higher valent Cu(II) or Cu(III) species by the persulfate radical anion generated in situ. This high-valent copper species facilitates the activation of the C-H bond at the C3 position of the benzofuranone ring, generating a reactive carbon-centered radical or a copper-bound organometallic intermediate. Simultaneously, the alcohol substrate undergoes coordination or hydrogen atom transfer, enabling the nucleophilic attack or radical recombination that forms the critical C-O ether bond. The presence of potassium dihydrogen phosphate (KH2PO4) acts as a mild buffer and base, stabilizing the reaction environment and preventing the decomposition of sensitive intermediates while promoting the turnover of the catalytic cycle.
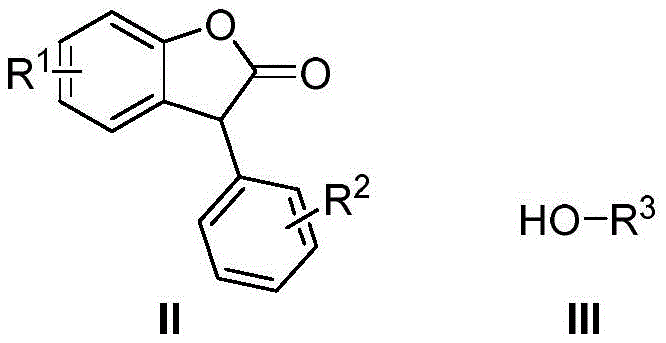
Understanding the substrate scope is vital for assessing the commercial viability of this route. The patent data indicates remarkable tolerance for electronic and steric variations. Electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as fluorine, chlorine, and nitro, are well-tolerated at the R1 and R2 positions of the benzofuranone core. Similarly, the alcohol component (R3) can range from simple methyl and ethyl groups to bulkier isopropyl and benzyl moieties without significant erosion in yield. This robustness suggests that impurity profiles can be tightly controlled, as the reaction conditions are specific enough to avoid non-selective oxidation of the alcohol or over-oxidation of the lactone ring, ensuring the delivery of high-purity OLED material or pharmaceutical intermediates with minimal downstream purification burden.
How to Synthesize Benzofuranone Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized stoichiometry and reaction parameters defined in the intellectual property. The process is designed to be operationally simple, avoiding the need for inert atmosphere gloveboxes or specialized high-pressure reactors, which lowers the barrier to entry for contract development and manufacturing organizations (CDMOs). The standardized protocol involves charging the benzofuranone and alcohol substrates into a reaction vessel followed by the addition of the catalytic system and solvent. Maintaining the temperature at 80°C is critical to ensure complete conversion within the 12-hour timeframe while preventing thermal degradation of the product. For detailed standard operating procedures and safety data sheets regarding the handling of persulfates and copper salts, please refer to the technical documentation provided by your chemical partner.
- Charge a reaction vessel with benzofuranone substrate and alcohol reactant in a 2: 1 molar ratio.
- Add cuprous bromide catalyst (10mol%), potassium persulfate oxidant (2 equiv), and potassium dihydrogen phosphate base (2 equiv) in acetonitrile solvent.
- Heat the mixture to 80°C and stir for 12 hours, then isolate the product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, the adoption of this copper-catalyzed CDC technology offers transformative benefits that extend beyond mere technical feasibility. The shift from precious metal catalysts to copper-based systems represents a fundamental change in cost structure, removing the volatility associated with palladium and rhodium pricing. Furthermore, the use of commodity chemicals like acetonitrile, potassium persulfate, and potassium dihydrogen phosphate ensures that the supply chain remains resilient against shortages of exotic reagents. This stability is paramount for supply chain heads who must guarantee continuity of supply for critical drug substances. The simplified workflow also translates to reduced manufacturing cycle times, as fewer unit operations are required for precursor synthesis and metal scavenging, thereby enhancing overall plant throughput and asset utilization rates.
- Cost Reduction in Manufacturing: The elimination of expensive pre-functionalized starting materials and noble metal catalysts leads to substantial cost savings in pharmaceutical intermediates manufacturing. By utilizing direct C-H activation, the process reduces the number of synthetic steps, which cumulatively lowers labor, energy, and solvent consumption costs. Additionally, the avoidance of heavy metal catalysts removes the need for costly and time-consuming metal scavenging resins or complex purification protocols, further driving down the Cost of Goods Sold (COGS) and improving margin potential for high-volume products.
- Enhanced Supply Chain Reliability: The reliance on widely available, bulk commodity chemicals such as cuprous bromide and simple alcohols mitigates the risk of supply disruptions often associated with specialized fine chemical reagents. This accessibility ensures that production schedules can be maintained consistently, reducing lead time for high-purity benzofuranone derivatives. The robustness of the reaction conditions also means that the process is less susceptible to minor variations in raw material quality, providing a more forgiving and stable manufacturing window that supports just-in-time inventory strategies.
- Scalability and Environmental Compliance: The mild reaction temperature of 80°C and the use of standard organic solvents facilitate easy scale-up from gram to ton quantities without requiring significant engineering modifications or capital investment in specialized high-pressure equipment. Moreover, the atom-economic nature of the CDC reaction minimizes waste generation, aligning with increasingly stringent environmental regulations and corporate sustainability mandates. This green chemistry profile simplifies waste disposal logistics and reduces the environmental footprint of the manufacturing site, enhancing the company's ESG (Environmental, Social, and Governance) standing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the fit of this technology within their existing portfolios and identify potential areas for process optimization or further development.
Q: What are the key advantages of this copper-catalyzed CDC method over traditional coupling?
A: This method eliminates the need for pre-functionalized substrates, utilizing direct C-H/O-H activation which significantly reduces raw material costs and waste generation compared to traditional halogenated coupling partners.
Q: Is this synthesis route scalable for commercial production of pharmaceutical intermediates?
A: Yes, the process operates at mild temperatures (80°C) using common solvents like acetonitrile and inexpensive inorganic salts, making it highly suitable for large-scale manufacturing without specialized high-pressure equipment.
Q: What is the substrate scope for R1, R2, and R3 groups in this reaction?
A: The protocol demonstrates excellent tolerance for various substituents including alkyl, alkoxy, halogens (F, Cl, Br), and nitro groups on the benzofuranone ring, as well as primary, secondary, and benzylic alcohols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzofuranone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of adopting innovative synthetic methodologies to maintain a competitive edge in the global fine chemicals market. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which are equipped with state-of-the-art analytical instrumentation to verify identity, assay, and impurity profiles according to international pharmacopeial standards.
We invite you to collaborate with us to leverage this advanced copper-catalyzed synthesis for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your supply chain resilience and your bottom line.
