Advanced Semi-Synthetic Route for High-Purity Taxane Derivatives and Paclitaxel Production
The pharmaceutical industry's relentless pursuit of efficient oncology therapeutics has long centered on the optimization of Paclitaxel synthesis. Patent CN100432064C introduces a transformative semi-synthetic methodology that fundamentally alters the landscape of taxane derivative production. This innovation pivots away from traditional, often inefficient pathways by leveraging a unique activated oxazole strategy. By reacting phenylisoserine with suitably protected baccatin III derivatives, this process generates versatile taxane substrates capable of being further modified into Paclitaxel and other potent analogs. For R&D directors and procurement specialists, this represents a critical evolution in API intermediate manufacturing, offering a pathway that mitigates the historical challenges of low yields and structural instability associated with earlier generations of semi-synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the semi-synthesis of taxanes from 10-deacetylbaccatin III (10-DAB III) has been fraught with significant chemical and economic hurdles. The pioneering work by Greene et al., while foundational, was plagued by inefficiency due to the requirement for large excesses of expensive side chains, resulting in suboptimal yields and problematic epimerization at the 2'-position. Subsequent improvements by Holton et al. utilizing cyclic amide derivatives offered better efficiency but introduced complexity in side chain formation. Furthermore, methods employed by major pharmaceutical entities like Bristol-Myers Squibb and Rhone-Poulenc Rorer, while commercially viable, often locked manufacturers into specific synthetic routes that lacked flexibility. These conventional pathways typically necessitate the formation of specific side chain amides at the very beginning of the synthesis, limiting the ability to generate a diverse library of novel taxane derivatives from a common intermediate without restarting the entire process.
The Novel Approach
The methodology disclosed in CN100432064C breaks these constraints by introducing a modular approach centered on activated oxazoles. Instead of committing to a final side chain structure immediately, this process generates a unique phenylisoserine derivative that serves as a highly reactive yet stable precursor. The key innovation lies in the use of activated benzaldehydes, specifically those containing electron-donating methoxy groups such as 3,4-dimethoxybenzaldehyde. This activation facilitates a subsequent hydrolysis step that is gentle enough to preserve the integrity of the sensitive taxane core while effectively removing the protecting group. This novel approach not only streamlines the production of Paclitaxel but also opens the door for the rapid synthesis of various novel taxane derivatives from a single, versatile intermediate, significantly enhancing the strategic flexibility for generic API manufacturers.
Mechanistic Insights into Activated Oxazole-Mediated Coupling
The core of this technological breakthrough resides in the precise manipulation of the phenylisoserine side chain. The process begins with the esterification of 2R,3S-phenylisoserine hydrochloride followed by amidation with α-methylcinnamoyl chloride to form a crystalline amide. Under acid catalysis, this amide undergoes cyclization with an activated benzaldehyde dimethyl acetal. The choice of the activating group is mechanistically critical; it must provide sufficient electron density to promote the formation of the oxazolidine ring while remaining labile enough for later removal. We observe that benzaldehydes with alkyl ether groups, particularly at the 4-position, are suitable, but 3,4-dimethoxy substitution proves optimal. This electronic tuning ensures that the subsequent hydrolysis with bases like lithium hydroxide proceeds without inducing epimerization at the chiral 2-position, a common failure point in less refined syntheses.
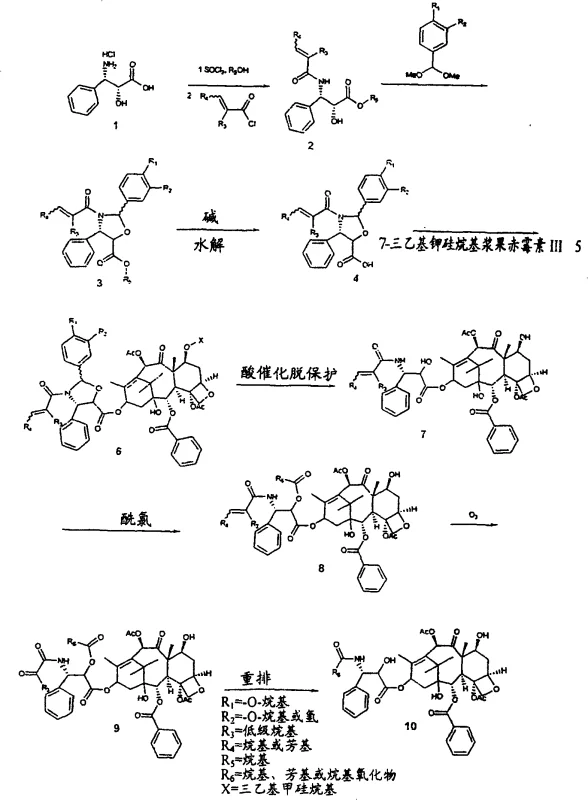
Following the formation of the activated oxazole acid, the coupling with the baccatin core is executed using standard carbodiimide chemistry, specifically dicyclohexylcarbodiimide (DCC) with DMAP catalysis. This step forms the crucial ester linkage at the C-13 position of the baccatin III scaffold. The subsequent deprotection phase is where the true elegance of the mechanism is revealed. Unlike non-activated oxazoles which resist cleavage or cause decomposition under harsh acidic conditions, the activated oxazole in this patent allows for clean removal using strong acid catalysts like gaseous hydrogen chloride or trifluoroacetic acid at moderate temperatures (0°C to 40°C). This controlled deprotection yields the free hydroxyl groups necessary for the final oxidative rearrangement steps, ensuring high purity and minimizing the formation of difficult-to-remove impurities that often plague commercial batches.
How to Synthesize Taxane Derivatives Efficiently
Implementing this synthesis requires strict adherence to the reaction parameters defined in the patent to ensure stereochemical fidelity and high yield. The process is designed to be robust, utilizing common organic solvents and reagents that are readily available in a GMP environment. From the initial preparation of the 7-TES-Baccatin III to the final oxidative cleavage with ozone, each step is optimized for scalability. The detailed standardized synthesis steps, including specific molar ratios, temperature controls, and workup procedures required to replicate this high-efficiency route, are outlined below for technical reference.
- Preparation of Phenylisoserine Derivative: React 2R,3S-phenylisoserine hydrochloride with methanol and thionyl chloride to form the methyl ester, followed by amidation with alpha-methylcinnamoyl chloride.
- Oxazole Cyclization: Cyclize the amide with activated benzaldehyde dimethyl acetal (preferably 3,4-dimethoxybenzaldehyde) under acid catalysis in toluene to form the activated oxazolidine carboxylic acid methyl ester.
- Coupling and Deprotection: Hydrolyze the ester to the acid, couple with 7-TES-Baccatin III using DCC/DMAP, and subsequently remove protecting groups under acidic conditions to yield the taxane substrate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this activated oxazole route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the substantial cost reduction in API manufacturing driven by improved material efficiency. By eliminating the need for the vast excesses of side chain reagents required in older Greene-type syntheses, the overall material cost per kilogram of finished product is significantly lowered. Furthermore, the ability to isolate key intermediates, such as Compound 6, as solid materials simplifies the purification workflow. This reduces the reliance on extensive chromatographic separations, which are often the bottleneck in terms of both time and solvent consumption in large-scale production facilities.
- Cost Reduction in Manufacturing: The process inherently lowers production costs by optimizing reagent stoichiometry and improving overall yield through reduced decomposition. The use of activated protecting groups means that deprotection can be achieved under milder conditions, reducing energy consumption and minimizing the degradation of the high-value baccatin core. Additionally, the avoidance of expensive transition metal catalysts in favor of standard organic reagents like DCC and DMAP eliminates the need for costly and time-consuming heavy metal scavenging steps, further driving down the operational expenditure associated with waste management and quality control testing.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of widely available starting materials. The reliance on 10-DAB derived from renewable Taxus needles and common chemical building blocks like phenylisoserine ensures that the supply chain is not vulnerable to the shortages of exotic or proprietary reagents. The robustness of the reaction conditions, which tolerate a range of temperatures and solvents, also means that production is less susceptible to minor fluctuations in utility availability or environmental controls, ensuring consistent delivery schedules for downstream formulation partners.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this route is highly favorable. The solvents employed, such as toluene, ethyl acetate, and dichloromethane, are standard industrial solvents with well-established recovery and recycling protocols. The process avoids the generation of complex heavy metal waste streams, simplifying effluent treatment and ensuring compliance with increasingly stringent environmental regulations. The ability to crystallize intermediates rather than relying solely on chromatography makes the transition from pilot plant to multi-ton commercial production seamless, reducing the lead time for high-purity taxane intermediates to reach the market.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and validation of this semi-synthetic pathway. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on critical process parameters and quality attributes.
Q: How does the activated oxazole method improve upon conventional Greene or Holton syntheses?
A: Unlike the Greene method which suffers from low yields and epimerization at the 2'-position due to excessive side chain usage, this novel approach utilizes an activated oxazole intermediate. This allows for milder hydrolysis conditions that prevent significant breakdown of the sensitive taxane structure while avoiding the rigidity of previous cyclic amide derivatives.
Q: What are the critical control points for maintaining stereochemical integrity during synthesis?
A: Maintaining the 2R,3S configuration is paramount. The process strictly controls hydrolysis and acidification conditions (pH ~2 using HCl or citric acid) to avoid epimerization at the 2-position of the phenylisoserine moiety. Additionally, the use of specific activating groups like 3,4-dimethoxybenzaldehyde ensures the oxazole ring opens cleanly without racemization.
Q: Is this process suitable for large-scale commercial manufacturing of Paclitaxel?
A: Yes, the process is designed for scalability. It employs robust reagents like dicyclohexylcarbodiimide (DCC) for coupling and standard solvents like toluene and ethyl acetate. The intermediates, such as Compound 6, can be isolated as solid materials, facilitating purification and quality control essential for GMP production environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Paclitaxel Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the competitive oncology market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the complexities of the activated oxazole method are managed with precision. We maintain stringent purity specifications and operate rigorous QC labs equipped to handle the analytical challenges of complex taxane derivatives, guaranteeing that every batch meets the exacting standards required for global regulatory submissions.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a secure and efficient supply of high-quality pharmaceutical intermediates.
