Advanced Semi-Synthetic Routes for Paclitaxel and Docetaxel: Scalable Manufacturing Solutions
Advanced Semi-Synthetic Routes for Paclitaxel and Docetaxel: Scalable Manufacturing Solutions
The global demand for potent antineoplastic agents continues to drive innovation in the manufacturing of taxane derivatives, specifically Paclitaxel and Docetaxel. As natural extraction from Taxus species remains limited by ecological constraints and low abundance, semi-synthetic approaches have become the industry standard for securing a reliable pharmaceutical intermediates supplier pipeline. A pivotal advancement in this domain is detailed in patent CN100586940C, which outlines a highly efficient semi-synthetic method utilizing protected 10-deacetylbaccatin III (10-DAB) and novel chiral side chains. This technology addresses critical bottlenecks in stereochemical control and process scalability, offering a robust pathway for the production of high-purity oncology APIs. By leveraging specific condensation and stereoinversion techniques, this method ensures consistent quality and supply continuity for downstream drug manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional total synthesis of taxanes is often plagued by excessive step counts, low overall yields, and the formidable challenge of constructing the complex taxane core with the correct stereochemistry. Even earlier semi-synthetic routes faced significant hurdles, particularly regarding the sourcing and coupling of the C-13 side chain. The synthesis of the requisite cis-side chain is notoriously difficult and expensive, often requiring resolution of racemates which halves the theoretical yield. Furthermore, conventional coupling methods sometimes suffer from epimerization at the C-2' position of the side chain, leading to impurities that are difficult to separate and detrimental to the biological activity of the final drug. These inefficiencies translate directly into higher costs and longer lead times, creating volatility in the supply chain for these life-saving medications.
The Novel Approach
The methodology described in CN100586940C introduces a strategic shift by utilizing a trans-chiral side chain, which is synthetically more accessible and stable than its cis-counterpart. The core innovation lies in a post-coupling stereochemical inversion step that corrects the configuration at the 2'-hydroxyl position only after the side chain is attached to the baccatin core. This decoupling of side chain synthesis from stereochemical perfection allows for higher yields in the early stages. The process involves a streamlined sequence: condensation of protected 10-DAB with the side chain, selective deprotection, stereoinversion (via oxazoline or Mitsunobu reaction), and final global deprotection. This logical flow minimizes handling of sensitive intermediates and maximizes atom economy.
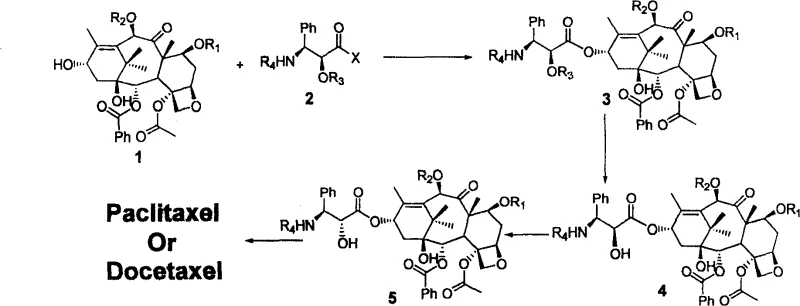
As illustrated in the reaction scheme above, the versatility of this approach allows for the divergence towards either Paclitaxel or Docetaxel by simply altering the protecting groups (R1, R2) and the specific inversion reagent used. This modularity is a significant advantage for contract development and manufacturing organizations (CDMOs) looking to optimize their production lines for multiple taxane products simultaneously without retooling entire facilities.
Mechanistic Insights into Stereoselective Inversion and Coupling
The success of this semi-synthetic route hinges on the precise control of stereochemistry during the coupling and inversion phases. The initial condensation reaction between the C-13 hydroxyl of the protected 10-DAB and the carboxylic acid of the chiral side chain is typically mediated by carbodiimide coupling agents such as DCC (dicyclohexylcarbodiimide) in the presence of nucleophilic catalysts like DMAP. This activation strategy ensures rapid ester formation under mild thermal conditions (30-40°C), preserving the integrity of the sensitive oxetane ring and other functional groups on the baccatin core. The use of mild temperatures is critical for preventing thermal degradation and minimizing the formation of elimination byproducts, which are common impurities in taxane chemistry.
Following the coupling and subsequent removal of the 2'-hydroxyl protecting group, the critical stereoinversion takes place. For Paclitaxel synthesis, the patent describes an oxazoline-mediated pathway using Vilsmeier reagent. This involves the formation of an oxazoline intermediate which locks the stereochemistry, followed by hydrolysis to reveal the inverted 2'-hydroxyl group with high fidelity. Alternatively, for Docetaxel, a classic Mitsunobu reaction employing triphenylphosphine (PPh3) and diethyl azodicarboxylate (DEAD) is utilized. This SN2-type displacement effectively flips the stereocenter with inversion of configuration. The ability to switch between these two proven mechanistic pathways within the same general framework provides exceptional flexibility for manufacturing different taxane analogues.

Impurity control is inherently built into this mechanism. By performing the inversion on the coupled intermediate rather than on the free side chain, the process avoids the isolation of unstable free side chain acids with the wrong stereochemistry. Furthermore, the final deprotection step using Zinc powder in acetic acid/methanol is highly chemoselective, removing Troc or Boc protecting groups without affecting the sensitive ester linkages or the oxetane ring, ensuring a clean final product profile that meets stringent pharmacopeial standards.
How to Synthesize Paclitaxel and Docetaxel Efficiently
The synthesis protocol outlined in the patent provides a clear, step-by-step guide for producing these complex molecules with high reproducibility. The process begins with the preparation of the ester intermediate, followed by sequential deprotection and inversion steps that are amenable to standard reactor setups. The use of common solvents like toluene, methanol, and ethyl acetate simplifies solvent recovery and waste management. Detailed operational parameters, including reagent equivalents and reaction times, are optimized to balance reaction rate with product purity. For a comprehensive breakdown of the specific experimental conditions and stoichiometry required for each stage of this transformation, please refer to the standardized synthesis guide below.
- Condense protected 10-deacetylbaccatin III (10-DAB) with a novel chiral side chain using condensing agents like DCC and DMAP at 30-40°C to form the ester linkage.
- Remove the protecting group from the 2'-hydroxyl of the side chain using acidic conditions (e.g., dilute HCl or HF/Pyridine) to expose the hydroxyl group.
- Invert the configuration of the 2'-hydroxyl group: use Vilsmeier reagent for Paclitaxel (oxazoline method) or Mitsunobu reaction (PPh3/DEAD) for Docetaxel.
- Perform final deprotection of the 7 and 10 positions (and side chain amine) using Zinc powder in acetic acid/methanol to yield the final active pharmaceutical ingredient.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this semi-synthetic method offers distinct advantages over legacy technologies. The primary benefit is the substantial reduction in raw material costs driven by the use of trans-chiral side chains. Since the trans-isomer is easier to synthesize and does not require costly chiral resolution steps that discard 50% of the material, the starting material costs are significantly lower. Additionally, the high yields reported in the patent embodiments—such as 91.6% for the initial condensation and over 73% for the final deprotection—indicate a process with excellent mass balance. This efficiency translates directly into reduced waste disposal costs and higher throughput per batch, allowing suppliers to offer more competitive pricing structures for bulk API intermediates.
- Cost Reduction in Manufacturing: The elimination of complex resolution steps for the side chain precursor drastically lowers the cost of goods sold (COGS). By utilizing a trans-side chain and correcting the stereochemistry later in the sequence, the process avoids the inherent 50% yield loss associated with resolving racemic mixtures. Furthermore, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to overall operational expenditure (OPEX) savings without compromising on the quality of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The reliance on readily available reagents such as DCC, DMAP, Zinc powder, and standard protecting group reagents ensures that the supply chain is not vulnerable to shortages of exotic catalysts. The robustness of the chemistry, demonstrated by its tolerance to standard workup procedures like column chromatography and crystallization, means that production schedules are less likely to be disrupted by batch failures. This reliability is crucial for maintaining continuous supply to downstream formulation partners who depend on just-in-time delivery of high-value oncology drugs.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial suitability, utilizing solvents and reagents that are manageable on a multi-kilogram to metric-ton scale. The final deprotection step using Zinc in acetic acid is a well-established, scalable reduction method that generates manageable waste streams compared to heavy metal catalyzed hydrogenolysis. This alignment with green chemistry principles facilitates easier regulatory approval and environmental compliance, reducing the risk of production halts due to environmental audits and ensuring long-term sustainability of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this specific semi-synthetic pathway. Understanding the nuances of the stereochemical inversion and the protecting group strategy is essential for partners evaluating this technology for their own supply chains. The answers provided are derived directly from the experimental data and claims within the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: How is the stereochemistry at the 2'-position controlled in this semi-synthesis?
A: The process utilizes a trans-chiral side chain which is easier to synthesize. The correct stereochemistry is achieved in a dedicated step where the 2'-hydroxyl configuration is inverted. For Paclitaxel, this is done via an oxazoline intermediate using Vilsmeier reagent, while for Docetaxel, a Mitsunobu reaction is employed to flip the stereocenter efficiently.
Q: What are the typical yields reported for this synthetic route?
A: The patent data indicates high efficiency across all stages. Embodiments report condensation yields up to 91.6%, deprotection yields around 85%, inversion yields near 80-86%, and final deprotection yields exceeding 73%. This cumulative high yield significantly impacts the cost of goods sold (COGS) favorably.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method is explicitly designed for industrial suitability. It employs mild reaction conditions (30-40°C for condensation), avoids extremely hazardous reagents where possible, and uses standard workup procedures like column chromatography and crystallization, making it robust for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Paclitaxel and Docetaxel Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the production of life-saving oncology therapeutics. Our technical team has extensively analyzed the semi-synthetic methodologies described in CN100586940C and possesses the expertise to implement these high-yielding strategies at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Paclitaxel or Docetaxel intermediate meets the highest global regulatory standards.
We invite you to collaborate with us to optimize your taxane supply chain. By leveraging our process chemistry expertise, we can help you navigate the complexities of stereochemical control and protecting group management to achieve superior cost efficiencies. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis. We are ready to provide specific COA data and route feasibility assessments tailored to your project requirements, ensuring a seamless transition from development to commercial manufacturing.
