Advanced Vilsmeier-Haack Amidation for High-Purity Pharmaceutical Intermediates Manufacturing
Advanced Vilsmeier-Haack Amidation for High-Purity Pharmaceutical Intermediates Manufacturing
The chemical industry is constantly seeking more efficient pathways to synthesize essential amide structures, which serve as critical backbones in countless active pharmaceutical ingredients (APIs) and agrochemical agents. Patent CN100500646C introduces a groundbreaking chemical synthesis method for amide substances that fundamentally shifts the paradigm from traditional, harsh acid-catalyzed processes to a more controlled and environmentally benign Vilsmeier-mediated pathway. This technology leverages the in situ generation of Vilsmeier reagents from di-(trichloromethyl)-carbonate and N,N-disubstituted amides, creating a highly reactive intermediate that facilitates the conversion of ketoximes into amides with exceptional precision. For R&D directors and process chemists, this represents a significant opportunity to enhance purity profiles while mitigating the safety risks associated with handling corrosive acylating agents. The methodology outlined in this patent not only promises superior reaction yields but also offers a robust framework for the reliable pharma intermediate supplier looking to optimize their production lines for complex molecular architectures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of amides has relied heavily on the acylation of aniline derivatives using acyl chlorides or acid anhydrides, processes that are fraught with significant operational challenges and safety hazards. These traditional acylation reactions often require stringent temperature controls to manage exotherms and frequently result in undesirable side reactions that complicate downstream purification and reduce overall material throughput. Furthermore, alternative methods utilizing ketoximes as feedstock typically depend on strong mineral acids such as sulfuric acid or phosphoric acid to catalyze the Beckmann rearrangement, leading to severe equipment corrosion and the generation of large volumes of acidic wastewater that require costly neutralization and treatment. The reliance on these aggressive reagents not only increases the capital expenditure for corrosion-resistant reactors but also poses substantial environmental compliance burdens, making the conventional synthesis of amides increasingly unsustainable in the context of modern green chemistry mandates and regulatory scrutiny.
The Novel Approach
In stark contrast to these legacy techniques, the novel approach detailed in the patent utilizes a sophisticated two-step sequence initiated by the formation of a Vilsmeier reagent under mild cryogenic conditions ranging from -5°C to 5°C. This method employs di-(trichloromethyl)-carbonate (BTC) reacting with N,N-disubstituted amides, such as dimethylformamide (DMF) or dimethylacetamide (DMAc), to generate a potent electrophilic species without the need for external strong acid catalysts. 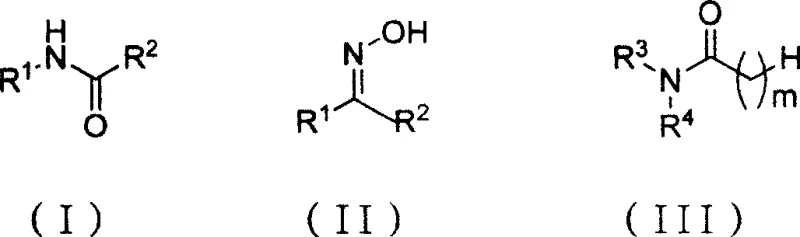 Once the Vilsmeier intermediate is established, ketoximes are introduced, and the reaction proceeds smoothly at elevated temperatures between 20°C and 150°C, typically optimized between 60°C and 120°C for maximum efficiency. This strategic shift allows for the precise manipulation of reaction kinetics, minimizing degradation pathways and ensuring that the resulting amide products, whether they are simple acetanilides or complex lactams, are obtained with high purity and minimal impurity burden, thereby streamlining the entire manufacturing workflow.
Once the Vilsmeier intermediate is established, ketoximes are introduced, and the reaction proceeds smoothly at elevated temperatures between 20°C and 150°C, typically optimized between 60°C and 120°C for maximum efficiency. This strategic shift allows for the precise manipulation of reaction kinetics, minimizing degradation pathways and ensuring that the resulting amide products, whether they are simple acetanilides or complex lactams, are obtained with high purity and minimal impurity burden, thereby streamlining the entire manufacturing workflow.
Mechanistic Insights into Vilsmeier-Mediated Amidation
The core of this technological advancement lies in the unique mechanistic pathway where di-(trichloromethyl)-carbonate acts as a dehydrating and activating agent to transform the tertiary amide into a chloroiminium salt, commonly known as the Vilsmeier reagent. This highly electrophilic intermediate then interacts with the nucleophilic oxygen of the ketoxime substrate, facilitating a rearrangement that effectively converts the oxime functionality into the desired amide bond. 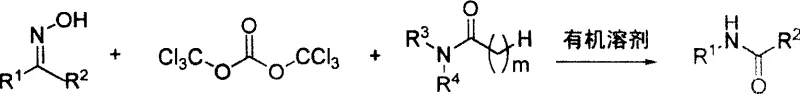 The stoichiometry is carefully balanced, with the patent specifying a molar ratio of ketoxime to di-(trichloromethyl)-carbonate to N,N-disubstituted amide at approximately 3:0.5-2:1.5-6, ensuring that there is sufficient activating agent to drive the reaction to completion without excessive waste. The use of BTC is particularly advantageous as it serves as a solid, easy-to-handle substitute for phosgene, releasing HCl in situ which is subsequently neutralized, thus avoiding the direct handling of toxic gases while maintaining the high reactivity required for difficult substrates like sterically hindered ketoximes.
The stoichiometry is carefully balanced, with the patent specifying a molar ratio of ketoxime to di-(trichloromethyl)-carbonate to N,N-disubstituted amide at approximately 3:0.5-2:1.5-6, ensuring that there is sufficient activating agent to drive the reaction to completion without excessive waste. The use of BTC is particularly advantageous as it serves as a solid, easy-to-handle substitute for phosgene, releasing HCl in situ which is subsequently neutralized, thus avoiding the direct handling of toxic gases while maintaining the high reactivity required for difficult substrates like sterically hindered ketoximes.
From an impurity control perspective, the mildness of the reaction conditions plays a pivotal role in defining the quality of the final product. Traditional strong acid methods often promote polymerization or hydrolysis side reactions due to the harsh acidic environment, leading to complex impurity profiles that are difficult to separate. By operating in organic solvents such as acetonitrile, dichloromethane, or toluene, and maintaining the reaction pH near neutral during the workup phase using saturated sodium carbonate, this new process effectively suppresses these degradation pathways. The ability to recrystallize the crude product directly from the reaction mixture, as demonstrated in the embodiments where purities exceed 98%, indicates that the mechanism favors the formation of a single dominant crystalline polymorph, significantly reducing the need for extensive chromatographic purification and enhancing the overall economic viability of the process for high-value intermediates.
How to Synthesize Acetanilide Efficiently
To implement this synthesis effectively, process engineers must adhere to a strict protocol that begins with the dissolution of di-(trichloromethyl)-carbonate in a suitable organic solvent, followed by the controlled addition of the N,N-disubstituted amide at low temperatures to ensure stable Vilsmeier reagent formation. The subsequent addition of the ketoxime must be managed carefully to prevent localized overheating, after which the reaction mixture is warmed to the target temperature range of 60°C to 120°C and maintained for a duration of 1 to 6 hours depending on the specific substrate reactivity.
- Dissolve di-(trichloromethyl)-carbonate in an organic solvent such as acetonitrile or dichloromethane and cool the mixture to between -5°C and 5°C.
- Slowly add the N,N-disubstituted amide (e.g., DMF or DMAc) to the cooled solution while stirring to generate the Vilsmeier reagent in situ.
- Add the ketoxime substrate to the reaction mixture, warm the temperature to between 60°C and 120°C, and maintain reaction for 1 to 6 hours before workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Vilsmeier-based synthesis route offers profound strategic advantages that extend far beyond simple yield improvements, fundamentally altering the cost structure of amide manufacturing. By eliminating the need for corrosive strong acids and the associated specialized infrastructure, facilities can significantly reduce their capital maintenance costs and extend the lifespan of their reactor vessels, leading to substantial long-term savings. The use of readily available and relatively inexpensive reagents like BTC and common solvents such as acetonitrile ensures a stable supply chain that is less susceptible to the volatility often seen with specialized acylating agents, thereby enhancing supply continuity and reducing the risk of production stoppages due to raw material shortages.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous acyl chlorides, coupled with the reduced need for corrosion-resistant equipment, drives down the overall cost of goods sold (COGS) significantly. Furthermore, the high selectivity of the reaction minimizes the formation of by-products, which reduces the consumption of solvents and energy required for purification steps like distillation or column chromatography, resulting in a leaner and more cost-effective production process that maximizes margin potential for high-volume commodities.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as ketoximes and DMF, which are produced on a massive global scale, ensures that the raw material supply is robust and resilient against market fluctuations. This stability allows for more accurate forecasting and inventory planning, enabling manufacturers to commit to tighter delivery schedules and fulfill large-scale orders with greater confidence, ultimately strengthening relationships with downstream pharmaceutical clients who demand just-in-time delivery capabilities.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste compared to traditional sulfuric acid methods, simplifying wastewater treatment protocols and reducing disposal fees. The mild reaction conditions and the use of standard organic solvents make the process inherently safer and easier to scale from pilot plant to commercial production, allowing facilities to ramp up capacity rapidly to meet surging market demand without encountering the engineering bottlenecks typical of highly exothermic or corrosive processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this patented amide synthesis technology, providing clarity on its practical application and benefits. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring that stakeholders have access to accurate and actionable information for decision-making.
Q: What are the typical yields for this amide synthesis method?
A: According to patent CN100500646C, the process consistently achieves high reaction yields, generally exceeding 80%, with specific examples demonstrating yields up to 97% for various substituted acetanilides.
Q: How does this method improve upon traditional Beckmann rearrangement?
A: Unlike traditional methods that rely on corrosive strong acids like sulfuric acid which cause severe pollution and equipment damage, this novel approach utilizes milder conditions and generates significantly less hazardous waste.
Q: What solvents are compatible with this Vilsmeier-based amidation?
A: The process is versatile regarding solvent choice, successfully operating in halohydrocarbons, esters, ethers, acetonitrile, benzene, toluene, or nitromethane, with acetonitrile and dichloromethane being particularly preferred.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amides Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced Vilsmeier-mediated synthesis route and possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring such innovations to the global market. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications that ensure every batch of amide intermediate meets the exacting standards of the international pharmaceutical industry, providing our partners with the confidence they need to accelerate their drug development pipelines. We are committed to leveraging this cutting-edge technology to deliver high-purity amides that enable faster time-to-market for our clients' critical therapeutic programs.
We invite you to engage with our technical procurement team to discuss how this novel synthesis method can be tailored to your specific project requirements, offering a Customized Cost-Saving Analysis that quantifies the potential efficiencies for your operations. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in complex organic synthesis can become a cornerstone of your supply chain strategy for sustainable and cost-effective chemical manufacturing.
