Advanced Synthetic Route for Cefepime Intermediates: Scaling High-Purity Beta-Lactams
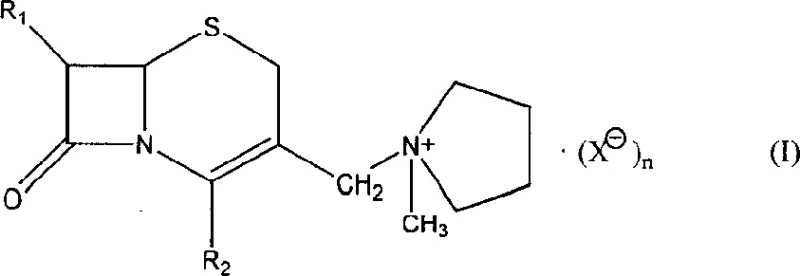
The pharmaceutical landscape for fourth-generation cephalosporins is undergoing a significant transformation driven by the need for more robust and economically viable synthetic pathways. Patent CN1159320C introduces a groundbreaking methodology for the preparation of 7 beta-(alkylamido)-3-(1-methyl-1-pyrrolidyl onium) methyl-3-cephem-4-carboxylate, a critical intermediate in the synthesis of Cefepime Hydrochloride. This innovation addresses the longstanding inefficiencies associated with traditional beta-lactam modification, specifically targeting the instability of 3-position substituents and the high costs of enzymatic deprotection. By shifting towards a chemically defined quaternization strategy, manufacturers can achieve superior control over impurity profiles while drastically simplifying the operational workflow. The technical implications of this patent extend beyond mere cost savings, offering a reliable cephalosporin intermediates supplier pathway that ensures consistent quality for large-scale antibiotic production. This report analyzes the mechanistic advantages and commercial scalability of this novel onium salt route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cefepime and related fourth-generation cephalosporins has been plagued by reliance on complex and fragile chemical transformations that hinder efficient commercial scale-up of complex polymer additives and pharmaceutical actives. Prior art, such as U.S. Pat 4,868,294, necessitates the formation of silyl compound intermediates using hazardous and expensive reagents like Iodotrimethylsilane and specialized refrigerants, creating significant supply chain vulnerabilities. Furthermore, alternative routes described in Chinese patent CN85101682 depend heavily on Penicillin G acylase for 7-position deprotection, a biological catalyst that requires stringent temperature control and complex recovery systems to maintain enzymatic activity. The failure to recover these enzymes effectively leads to substantial waste generation and unpredictable batch-to-batch variability in product purity. Additionally, the use of sodium iodide in substitution reactions, as seen in U.S. Pat 4,406,899, introduces heavy metal contamination risks that require additional purification steps, further inflating the cost reduction in pharmaceutical intermediates manufacturing. These conventional bottlenecks create a fragile production environment where minor deviations in reagent quality can compromise the entire batch yield.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN1159320C employs a direct quaternization strategy that bypasses the need for enzymatic cleavage or exotic silylating agents entirely. This novel approach utilizes N-methylpyrrolidine to directly displace the 3-chloromethyl leaving group under mild conditions, forming a stable quaternary ammonium salt that serves as a robust platform for subsequent acylation. The process operates within a moderate temperature range of -20°C to 40°C, utilizing tetrahydrofuran (THF) as a solvent, which eliminates the safety hazards associated with cryogenic reactions or high-pressure systems. By avoiding the use of Penicillin G acyltransferase, the method removes the biological variability inherent in enzymatic processes, ensuring a more predictable and reproducible reaction outcome for high-purity OLED material and pharma grade products alike. This chemical elegance translates directly into operational simplicity, allowing facilities to reduce lead time for high-purity cephalosporin intermediates by streamlining the workup and purification stages. The result is a streamlined synthesis that maintains high stereochemical integrity while significantly lowering the barrier to entry for commercial production.
Mechanistic Insights into Quaternary Ammonium Salt Formation
The core of this technological advancement lies in the nucleophilic substitution mechanism at the 3-position of the cephem nucleus, which is meticulously optimized to prevent degradation of the sensitive beta-lactam ring. The reaction initiates with the attack of the lone pair electrons on the nitrogen atom of N-methylpyrrolidine onto the electrophilic carbon of the 3-chloromethyl group. This SN2-type displacement is facilitated by the polar aprotic nature of the THF solvent, which stabilizes the transition state without promoting unwanted side reactions such as beta-lactam ring opening. The formation of the positively charged onium center creates a strong electrostatic interaction with the counterion, typically a chloride or bromide, which enhances the crystallinity of the final intermediate. This structural rigidity is crucial for preventing the rearrangement reactions that often plague 3-unsubstituted cephalosporins during storage or subsequent processing steps. Understanding this mechanistic nuance is vital for R&D teams aiming to replicate the high purity standards required for regulatory approval in global markets.
Controlling the impurity profile in this synthesis requires precise management of the reaction stoichiometry and pH levels during the workup phases. The patent specifies the use of sodium bicarbonate to adjust the pH to 6-7 during the final acylation step, a critical parameter that ensures the amino group at the 7-position remains nucleophilic enough to react with the AE active ester without hydrolyzing the newly formed onium salt. Excessive acidity could lead to the protonation of the pyrrolidine nitrogen, deactivating the molecule or causing decomposition of the beta-lactam core. Conversely, overly basic conditions might trigger elimination reactions at the 3-position, regenerating the unstable double bond and compromising the structural integrity of the drug candidate. The detailed protocol involving slow addition of reaction mixtures into acetone for precipitation further demonstrates a focus on particle size control and polymorph selection. These fine-tuned parameters collectively ensure that the final Cefepime Hydrochloride meets the stringent purity specifications demanded by international pharmacopoeias.
How to Synthesize 7-beta-alkylamido-3-cephem Efficiently
Implementing this synthesis route requires a disciplined approach to reagent preparation and temperature control to maximize yield and minimize byproduct formation. The process begins with the protection of the 7-beta-amino group using formic acid and acetic anhydride, followed by the removal of the 4-position carboxyl protecting group using trifluoroacetic acid in methylene dichloride. The critical quaternization step involves dissolving the 3-chloromethyl intermediate in THF and adding N-methylpyrrolidine dropwise while maintaining the temperature between -20°C and 40°C to manage the exotherm. Detailed standardized synthetic steps see the guide below for specific molar ratios and stirring times that have been validated to produce the light yellow solid compound with high consistency. Adhering to these protocols allows manufacturers to transition from laboratory scale to pilot plant operations with confidence in the reproducibility of the chemical transformation.
- Protect the 7-beta-amino group of the cephalosporin nucleus via acylation to form the 7-beta-alkylamido-3-chloromethyl derivative.
- Remove the 4-position carboxyl protecting group under mild acidic conditions to yield the free acid intermediate.
- Perform quaternization using N-methylpyrrolidine in THF at controlled temperatures between -20°C and 40°C to generate the final onium salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this patent technology offers profound advantages by decoupling production capacity from the volatility of biological enzyme markets. Traditional methods that rely on Penicillin G acylase are susceptible to supply disruptions and batch inconsistencies inherent in fermentation-derived products, whereas this chemical route utilizes commodity chemicals that are readily available from multiple global vendors. This shift significantly enhances supply chain reliability by reducing dependency on single-source biological suppliers and mitigating the risk of production stoppages due to enzyme deactivation. Furthermore, the elimination of expensive silylating reagents and specialized refrigerants directly contributes to substantial cost savings in raw material expenditure, improving the overall margin profile for generic antibiotic manufacturers. The simplified workflow also reduces the requirement for specialized containment equipment needed for hazardous reagents, lowering capital expenditure requirements for facility upgrades. These factors combine to create a more resilient and cost-effective manufacturing ecosystem capable of withstanding market fluctuations.
- Cost Reduction in Manufacturing: The economic benefits of this process are primarily derived from the complete removal of enzymatic deprotection steps, which traditionally account for a significant portion of processing costs due to enzyme purchase and recovery infrastructure. By replacing biological catalysis with a straightforward chemical quaternization, manufacturers eliminate the need for complex filtration and recycling systems designed to preserve enzyme activity. Additionally, the use of common solvents like THF and ethanol instead of specialized fluorinated refrigerants reduces both material costs and environmental compliance fees associated with volatile organic compound emissions. The higher purity of the intermediate also minimizes the need for extensive recrystallization cycles, thereby reducing solvent consumption and energy usage per kilogram of product. These cumulative efficiencies drive a drastic simplification of the cost structure without compromising the quality of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Operational continuity is significantly improved by the robustness of the chemical reagents employed in this synthetic route, which possess long shelf lives and do not require cold-chain logistics for storage. Unlike enzymatic processes that demand strict temperature controls to prevent denaturation, the chemical intermediates described in this patent can be stored under ambient conditions, reducing warehousing complexity and energy costs. The availability of N-methylpyrrolidine and standard cephalosporin nuclei from established chemical suppliers ensures that production schedules are not held hostage by niche reagent shortages. This stability allows supply chain managers to forecast production timelines with greater accuracy, ensuring consistent delivery of high-purity cephalosporin intermediates to downstream formulation partners. The reduction in process sensitivity also means that minor variations in raw material quality are less likely to cause batch failures, further securing the supply line.
- Scalability and Environmental Compliance: The environmental footprint of this manufacturing process is markedly lower than conventional methods due to the absence of heavy metal catalysts and halogenated solvents that pose disposal challenges. The reaction generates fewer toxic byproducts, simplifying wastewater treatment requirements and aligning with increasingly stringent global environmental regulations regarding pharmaceutical effluent. Scalability is enhanced by the homogeneous nature of the reaction mixture, which facilitates efficient heat transfer and mixing in large-scale reactors, allowing for seamless translation from 100 kgs to 100 MT annual commercial production volumes. The solid-state stability of the onium salt intermediate also permits flexible production scheduling, as batches can be held in inventory without significant degradation, decoupling synthesis from immediate packaging demands. This combination of green chemistry principles and engineering scalability makes the process highly attractive for modern, compliant manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route for cefepime intermediates. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation to ensure accuracy and relevance for industry stakeholders. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production lines. The responses highlight the specific advantages over prior art methods while acknowledging the critical process parameters that must be maintained for success.
Q: How does this patent method improve upon traditional enzymatic deprotection?
A: Unlike prior art requiring Penicillin G acylase which needs complex recovery systems to maintain activity, this method utilizes chemical acylation and deprotection steps that eliminate biological variability and enzyme costs.
Q: What are the storage stability benefits of the onium salt intermediate?
A: The formation of the quaternary ammonium salt at the 3-position significantly enhances the crystallinity and thermal stability of the molecule compared to unstable chloromethyl precursors, facilitating safer long-term storage.
Q: Does this route require expensive silylating reagents like Iodotrimethylsilane?
A: No, the disclosed process avoids the use of specialized silylating agents such as Iodotrimethylsilane or Refrigerant R 113, relying instead on standard organic solvents like THF and common amines.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefepime Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN1159320C are fully realized in practical manufacturing environments. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch against international standards. Our capability to handle complex beta-lactam chemistry positions us as a strategic partner for companies seeking to optimize their antibiotic supply chains with high-quality, cost-effective intermediates. We understand the nuances of regulatory compliance and are prepared to support your documentation needs for FDA or EMA filings.
We invite you to engage with our technical procurement team to discuss how this innovative onium salt route can be tailored to your specific production requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic impact of switching to this enzymatic-free process. We encourage you to contact us to obtain specific COA data and route feasibility assessments that demonstrate our commitment to transparency and technical excellence. Let us collaborate to enhance your supply chain resilience and drive down manufacturing costs through superior chemical engineering.
