Advanced Anhydrous Acylation Technology for High-Purity Cefepime Antibiotic Production
Advanced Anhydrous Acylation Technology for High-Purity Cefepime Antibiotic Production
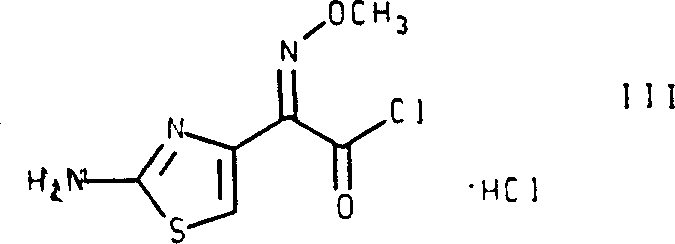
The pharmaceutical industry constantly seeks robust synthetic routes that ensure high stereochemical purity while minimizing processing complexity. Patent CN1059438C introduces a groundbreaking anhydrous acylation process specifically designed for the preparation of the antibiotic Cefepime dihydrochloride hydrate. This technology addresses a critical challenge in beta-lactam synthesis: the control of geometric isomerism in the methoxyimino side chain. Traditional methods often struggle with the formation of unwanted trans-isomers and Delta-2 isomers, which compromise the efficacy and stability of the final drug substance. The invention provides a method to prepare a stable, crystalline cis-isomer of 2-(2-aminothiazol-4-yl)-2-methoxyiminoacetyl chloride hydrochloride that is substantially free of the trans-isomer. This intermediate serves as a pivotal building block, enabling the direct acylation of the cephalosporin nucleus without the need for cumbersome amino protection and deprotection steps. By leveraging this specific chemical pathway, manufacturers can achieve superior impurity profiles and enhanced process efficiency, marking a significant evolution in the commercial production of fourth-generation cephalosporins.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing Cefepime and related cephalosporins have been plagued by significant chemical and operational inefficiencies. Prior art methods, such as those described in earlier patents, frequently relied on the use of protected amino groups and complex activating agents to facilitate the acylation of the 7-amino position on the cephalosporin nucleus. These protecting groups necessitate additional synthetic steps for installation and subsequent removal, which inherently lowers the overall yield and increases the consumption of raw materials. Furthermore, many conventional processes utilized toxic leaving groups, such as 2-mercaptobenzothiazole, which pose environmental hazards and require rigorous purification protocols to ensure they do not contaminate the final active pharmaceutical ingredient. Another major drawback of older technologies was the tendency to produce mixtures of cis and trans isomers, often requiring expensive and time-consuming chromatographic separation to isolate the biologically active cis-form. The instability of intermediate zwitterionic forms at room temperature further complicated storage and logistics, creating bottlenecks in the supply chain for large-scale manufacturing operations.
The Novel Approach
The methodology disclosed in the patent represents a paradigm shift by utilizing an unprotected, stable crystalline acid chloride hydrochloride salt as the acylating agent. This novel approach eliminates the need for amino protection on the thiazole ring, thereby removing two entire reaction steps from the synthesis tree. The process employs a specific chlorination protocol using oxalyl chloride and dimethylformamide under strictly controlled low-temperature conditions to preserve the desired cis-geometry. By avoiding the use of toxic organic leaving groups and relying instead on a simple chloride ion, the process significantly reduces the chemical burden on downstream purification systems. The resulting intermediate is not only stable but also crystalline, allowing for easy isolation, storage, and quality control testing before it is committed to the final acylation reaction. This direct route facilitates the production of Cefepime dihydrochloride hydrate that is substantially free of both trans-isomers and Delta-2 isomers, achieving high purity directly from the reaction mixture without the need for chromatographic intervention. This streamlined workflow translates directly into reduced operational costs and a more reliable manufacturing timeline for pharmaceutical producers.
Mechanistic Insights into Stereoselective Chlorination and Acylation
The core innovation of this technology lies in the precise control of reaction conditions during the conversion of the methoxyiminoacetic acid derivative into its corresponding acid chloride. The patent highlights that standard chlorinating agents, such as phosphorus pentachloride, often induce isomerization, leading to the formation of the undesirable trans-isomer. To circumvent this, the inventors discovered that using a combination of oxalyl chloride and dimethylformamide (DMF) at temperatures below -10°C, preferably between -15°C and -40°C, is critical. The mechanism involves the formation of a Vilsmeier-Haack type reagent in situ, which activates the carboxylic acid moiety. Crucially, the molar ratio of DMF to oxalyl chloride must be carefully managed; the patent specifies that the molar amount of DMF should exceed that of oxalyl chloride to prevent instability and isomerization. This delicate balance ensures that the methoxyimino double bond retains its Z-configuration (cis) throughout the activation process. The use of inert solvents like dichloromethane further supports this stereochemical integrity by providing a non-polar environment that minimizes side reactions. This mechanistic precision allows for the isolation of the acid chloride hydrochloride salt as a stable solid, which is a rare achievement for such reactive intermediates.
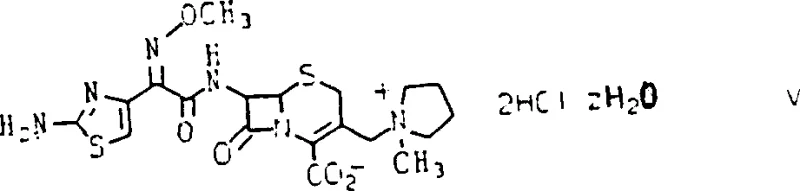
Following the preparation of the activated side chain, the acylation of the cephalosporin nucleus requires equally precise conditions to prevent the migration of the double bond from the Delta-3 to the Delta-2 position. The process involves the silylation of the 7-amino-cephalosporin carboxylate salt to form a soluble silylated derivative in situ. This solubilization is essential for the reaction to proceed in an organic medium. The acylation is then carried out under non-basic conditions to avoid base-catalyzed isomerization of the cephem nucleus. The patent emphasizes that the addition of the acid chloride and the base (such as N-methylmorpholine or triethylamine) must be controlled, often in portions, to maintain a pH that favors acylation over degradation. The reaction mixture is subsequently treated with water to induce phase separation, allowing the desired antibiotic to partition into the aqueous phase while organic impurities remain in the organic layer. This biphasic workup is a key feature that enables the direct crystallization of the final product as a stable dihydrochloride hydrate, bypassing the need to isolate unstable zwitterionic intermediates.
How to Synthesize Stable Cis-Cefepime Side Chain Acid Chloride Efficiently
The synthesis of this critical intermediate requires strict adherence to temperature and stoichiometry to ensure the high stereochemical purity demanded by regulatory standards. The process begins with the conversion of the free acid into its hydrochloride salt using gaseous HCl in a solvent like toluene or dichloromethane, followed by the critical chlorination step at cryogenic temperatures. Detailed operational parameters, including the specific order of addition for oxalyl chloride and DMF, are vital for reproducibility. The following guide outlines the standardized synthesis steps derived from the patent examples to assist process chemists in implementing this technology.
- Convert the cis-isomer of 2-(2-aminothiazol-4-yl)-2-methoxyiminoacetic acid into its corresponding hydrochloride salt using gaseous hydrogen chloride in an inert solvent like toluene or dichloromethane.
- Treat the resulting acid hydrochloride salt with a chlorinating agent mixture comprising oxalyl chloride and dimethylformamide at temperatures below -10°C, preferably between -15°C and -40°C.
- Maintain a molar ratio where dimethylformamide exceeds oxalyl chloride slightly to prevent isomerization, ensuring the final product is the stable crystalline cis-isomer substantially free of trans-impurities.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this anhydrous acylation technology offers substantial strategic benefits beyond mere chemical elegance. The elimination of protecting group chemistry significantly reduces the number of unit operations required, which directly correlates to lower capital expenditure on equipment and reduced utility consumption per kilogram of product. By removing the need for chromatographic purification, the process becomes inherently more scalable, allowing for the transition from pilot plant to commercial production with minimal technical risk. The stability of the crystalline intermediate also enhances supply chain resilience; unlike unstable oils or zwitterions that degrade rapidly, this solid intermediate can be stockpiled, decoupling the production of the side chain from the final coupling step. This flexibility allows manufacturers to optimize batch scheduling and respond more agilely to market demand fluctuations without compromising product quality. Furthermore, the use of non-toxic chloride leaving groups simplifies waste treatment protocols, aligning with increasingly stringent environmental regulations and reducing the cost of hazardous waste disposal.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the drastic simplification of the synthetic route. By avoiding the purchase and handling of expensive protecting group reagents and the associated solvents for their removal, the raw material cost profile is significantly improved. The high yield of the crystallization steps, as demonstrated in the patent examples, ensures that material throughput is maximized, reducing the cost of goods sold. Additionally, the avoidance of chromatography eliminates the need for large volumes of silica gel and elution solvents, which are major cost drivers in traditional fine chemical manufacturing. The overall process intensity is lowered, meaning less energy is required for heating, cooling, and solvent recovery, contributing to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by complex processes with multiple failure points. This technology mitigates such risks by establishing a robust, crystalline intermediate that serves as a quality gate. Because the intermediate is stable and easily characterized, suppliers can guarantee the purity of the starting material for the final step, reducing the likelihood of batch failures in the costly final acylation stage. The ability to store the intermediate allows for the creation of safety stocks, buffering against upstream supply disruptions. Moreover, the simplified workflow reduces the lead time from raw material intake to finished API, enabling faster turnaround times for customers and improving the overall responsiveness of the supply network to global pharmaceutical needs.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden complexities, but this anhydrous method is designed with manufacturability in mind. The use of common solvents like dichloromethane and acetone, combined with standard filtration and crystallization techniques, ensures that the process can be readily transferred to multi-ton reactors without specialized equipment. From an environmental perspective, the replacement of toxic leaving groups with chloride ions significantly reduces the ecological footprint of the manufacturing process. The reduction in solvent usage and the elimination of chromatographic waste streams contribute to a greener manufacturing profile, helping companies meet their sustainability goals and comply with international environmental standards such as REACH and TSCA without additional remediation costs.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this patented process is essential for stakeholders evaluating its implementation. The following questions address common inquiries regarding the stability, purity, and regulatory implications of using this specific acylation method. These answers are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for technical decision-makers.
Q: Why is the cis-isomer purity critical for Cefepime manufacturing?
A: The biological activity and stability of Cefepime depend heavily on the geometric configuration of the methoxyimino side chain. The trans-isomer is biologically inactive or less active and constitutes a difficult-to-remove impurity. The patented process ensures the intermediate is substantially free of the trans-isomer (<1%), eliminating the need for complex chromatographic purification later in the synthesis.
Q: How does this anhydrous acylation method improve supply chain reliability?
A: Traditional methods often require unstable zwitterionic forms or complex protecting group strategies that extend lead times. This method produces a stable, crystalline acid chloride hydrochloride salt that can be stored and transported. The elimination of chromatographic purification steps and the use of simple chloride leaving groups significantly streamline the manufacturing timeline, enhancing supply continuity for large-scale API production.
Q: What are the safety advantages of using this specific acid chloride intermediate?
A: Unlike prior art methods that utilize toxic leaving groups such as 2-mercaptobenzothiazole, this process employs a simple chloride ion as the leaving group. This reduces the toxicity profile of the waste stream and simplifies the purification of the final antibiotic, as there is no need for extensive measures to remove complex organic leaving group residues from the final pharmaceutical product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefepime Intermediate Supplier
As the global demand for high-quality antibiotics continues to rise, securing a supply of critical intermediates produced via advanced, compliant methodologies is paramount. NINGBO INNO PHARMCHEM stands ready to support your pharmaceutical development and commercialization goals with our expertise in complex organic synthesis. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the critical cis/trans isomer ratios essential for Cefepime efficacy. We understand that the reliability of your supply chain depends on the robustness of your intermediates, and we are committed to delivering products that meet the highest international standards for pharmaceutical manufacturing.
We invite you to engage with our technical team to discuss how this anhydrous acylation technology can be integrated into your production strategy. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and process constraints. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for this stable cis-isomer intermediate. Let us collaborate to optimize your antibiotic manufacturing process, ensuring both economic efficiency and uncompromised product quality for the global market.
