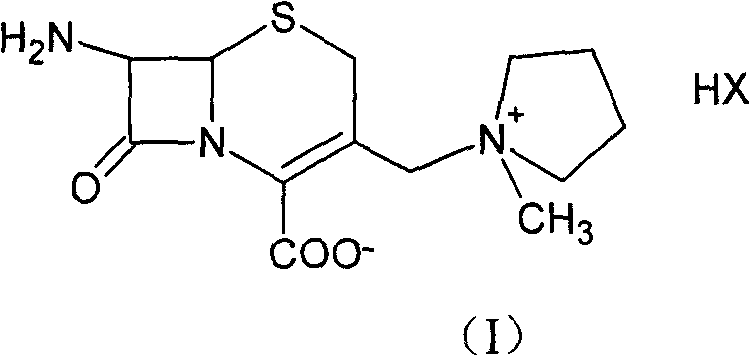
Advanced Manufacturing of Cefepime Intermediate 7-ACP via Optimized Mixed Solvent Systems
The pharmaceutical industry continuously seeks robust synthetic routes for fourth-generation cephalosporins, particularly for the critical intermediate known as 7-amino-3-[(1-methylpyrrolidinium)methyl]-3-cephem-4-carboxylate, commonly referred to as 7-ACP. Patent CN100453545C introduces a transformative methodology for synthesizing this key cefepime intermediate within a specialized mixed solvent system. This innovation addresses long-standing challenges in beta-lactam chemistry, specifically the suppression of the thermodynamically stable but pharmacologically inactive Δ2-isomer. By leveraging a strategic combination of dichloromethane and polar aprotic solvents such as N,N-dimethylformamide (DMF) or dimethyl sulfoxide (DMSO), the process achieves a remarkable molar yield of 80% based on the starting material 7-ACA. Furthermore, the resulting product demonstrates exceptional quality metrics, with a purity exceeding 99.5% and a color grade superior to No. 3, establishing a new benchmark for high-purity pharmaceutical intermediates in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 7-ACP has been plagued by significant technical and environmental hurdles that compromise both economic viability and product quality. Early methodologies, such as those disclosed in U.S. Patents 4868294 and 4714760, relied heavily on Freon TF (1,1,2-trichloro-1,2,2-trifluoroethane) as the primary reaction solvent. While effective in minimizing isomer formation, Freon TF is strictly prohibited globally due to its severe destructive impact on the stratospheric ozone layer, forcing manufacturers to seek compliant alternatives. Subsequent attempts to replace Freon with naphthenic hydrocarbons (C5-C8) resulted in inadequate solubility for the reaction intermediates, leading to prolonged reaction times and inferior product coloration. Moreover, when utilizing pure dichloromethane—a more common industrial solvent—without modification, the process suffers from a catastrophic generation of the Δ2-isomer, reaching levels as high as 50%, which drastically reduces the effective yield of the active pharmaceutical ingredient and complicates downstream purification efforts.
The Novel Approach
The methodology described in CN100453545C represents a paradigm shift by engineering a precise mixed solvent environment that optimizes both solubility and reaction kinetics. Instead of relying on a single solvent phase, this novel approach initiates the reaction in dichloromethane but strategically introduces a non-protonic polar solvent, such as DMF, DMAc, or DMSO, prior to the critical nucleophilic substitution step. This adjustment ensures that the 3-position iodo-intermediate remains in a uniform solution state even at the low temperatures required for isomer control. By maintaining homogeneity, the process facilitates a smoother interaction between the electrophilic iodo-species and the activated N-methylpyrrolidine nucleophile. This sophisticated solvent engineering effectively suppresses the rearrangement to the Δ2-isomer while simultaneously accelerating the reaction rate, thereby delivering a reliable cefepime intermediate supplier solution that balances regulatory compliance with superior chemical performance.
Mechanistic Insights into Silylation and Nucleophilic Substitution
The core of this synthetic strategy lies in the meticulous control of protecting group chemistry and the activation of the nucleophile. The process begins with the silylation of 7-amino-3-acetoxymethylcephalosporanic acid (7-ACA) using hexamethyldisilazane (HMDS) to protect both the amino and carboxyl functionalities. Following this, the 3-acetoxy group is converted into a highly reactive 3-iodo species using trimethylsilyl iodide (TMSI). It is mechanistically critical to note that an excess of TMSI, typically in a molar ratio of 1.15 to 1.25 relative to 7-ACA, is required to drive the iodination to completion. However, this excess TMSI must be carefully quenched using an ether compound, such as tetrahydrofuran (THF), to prevent interference with subsequent steps. The reaction scheme below illustrates this progression from the protected starting material to the reactive iodo-intermediate.
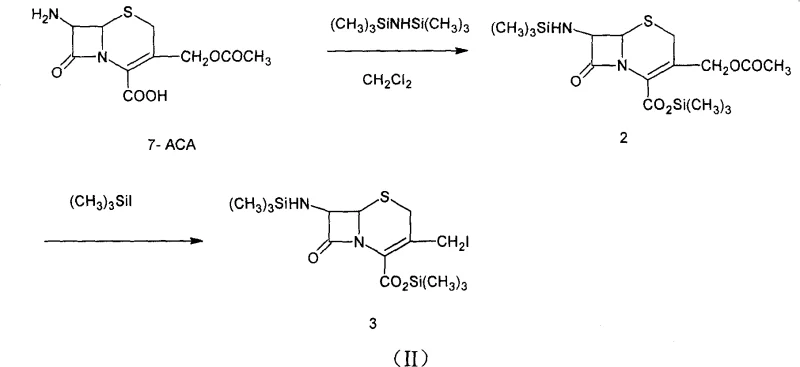
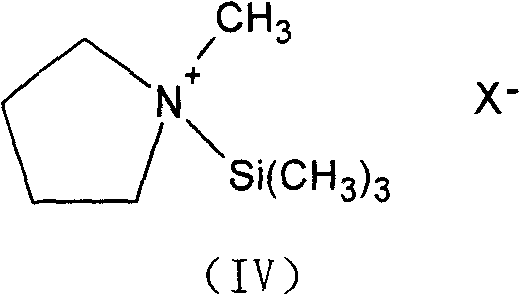
Following the formation of the iodo-intermediate, the process employs a unique activation strategy for the nitrogen nucleophile. N-methylpyrrolidine is activated using N,O-bis(trimethylsilyl)acetamide (BSA) to form a highly reactive silylated amine species, as depicted in Formula (IV). This activation enhances the nucleophilicity of the nitrogen atom, allowing the substitution reaction to proceed efficiently at significantly lower temperatures, specifically between -30°C and 10°C. Operating at these cryogenic conditions is the key mechanistic lever for inhibiting the formation of the Δ2-isomer, as higher temperatures would thermodynamically favor the migration of the double bond. The precise stoichiometry of the activator, maintained at a ratio of 1.10 to 1.20 relative to the starting material, ensures complete conversion without generating excessive byproducts, ultimately leading to the high-purity final salt after acid deprotection and crystallization.
How to Synthesize 7-ACP Efficiently
The execution of this synthesis requires strict adherence to temperature controls and solvent ratios to replicate the high yields reported in the patent literature. The procedure involves a multi-step sequence where the initial silylation and iodination are performed in dichloromethane, followed by a partial solvent recovery and the addition of the polar co-solvent before the amine addition. This specific sequence is designed to maximize the solubility of the intermediate while minimizing thermal stress on the beta-lactam ring. For detailed operational parameters, including specific addition rates and workup procedures, please refer to the standardized protocol outlined below.
- Protect 7-ACA using hexamethyldisilazane (HMDS) in dichloromethane, followed by iodination with trimethylsilyl iodide (TMSI).
- Partially recover dichloromethane under reduced pressure and introduce a polar aprotic solvent like DMF or DMSO to create a mixed solvent system.
- React the iodo-intermediate with BSA-activated N-methylpyrrolidine at low temperatures (-30 to 10°C), followed by acid deprotection and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this mixed solvent methodology offers profound advantages in terms of cost structure and operational reliability. The most significant benefit is the substantial increase in material efficiency, with molar yields jumping from the historical average of 50-60% to approximately 80%. This dramatic improvement directly translates to cost reduction in antibiotic manufacturing by reducing the consumption of expensive starting materials like 7-ACA per kilogram of final product. Furthermore, the elimination of ozone-depleting substances like Freon TF removes the regulatory burden and associated costs of handling restricted chemicals, simplifying the compliance landscape for production facilities. The use of common, commercially available solvents such as dichloromethane and DMF ensures that the supply chain remains resilient and unaffected by the volatility of specialty solvent markets.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive and environmentally hazardous solvents while significantly boosting yield. By achieving an 80% yield compared to the 50-60% of older methods, the effective cost per unit of active intermediate is drastically lowered. Additionally, the high purity (>99.5%) reduces the need for extensive recrystallization or chromatographic purification, further lowering processing costs and solvent waste disposal fees.
- Enhanced Supply Chain Reliability: The reagents utilized in this pathway, including HMDS, TMSI, and BSA, are commodity chemicals with stable global supply chains. Unlike processes dependent on restricted fluorinated solvents, this method relies on standard industrial solvents that are readily accessible from multiple vendors. This diversification of raw material sources mitigates the risk of supply disruptions and ensures consistent production scheduling for large-scale campaigns.
- Scalability and Environmental Compliance: The reaction conditions are well-suited for commercial scale-up of complex pharmaceutical intermediates. The workup involves straightforward crystallization techniques rather than complex separations, facilitating easy transfer from pilot plant to multi-ton production. Moreover, the absence of CFCs aligns the manufacturing process with modern environmental, social, and governance (ESG) goals, making the final product more attractive to sustainability-conscious pharmaceutical buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this specific synthetic route. These answers are derived directly from the experimental data and claims found within the patent documentation, providing clarity on the operational nuances of the mixed solvent system. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into their existing manufacturing infrastructure.
Q: Why is a mixed solvent system preferred over pure dichloromethane for this synthesis?
A: Pure dichloromethane leads to poor solubility of the iodo-intermediate at low temperatures and promotes the formation of the unwanted Δ2-isomer (up to 50%). A mixed solvent system containing polar aprotic solvents like DMF or DMSO ensures a homogeneous solution, significantly suppressing isomer formation and improving yield.
Q: What is the expected yield and purity of the 7-ACP produced via this method?
A: According to patent CN100453545C, this method achieves a molar yield of approximately 80% based on 7-ACA, which is substantially higher than conventional methods. The final product exhibits a color grade of less than No. 3 and a purity exceeding 99.5%.
Q: How does this process address environmental regulations regarding solvents?
A: Traditional methods often relied on Freon TF (1,1,2-trichloro-1,2,2-trifluoroethane), which is banned due to ozone depletion. This novel method utilizes dichloromethane and common polar aprotic solvents, eliminating the need for environmentally hazardous chlorofluorocarbons while maintaining high reaction efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-ACP Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving antibiotics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated mixed solvent chemistry described in CN100453545C can be executed with precision at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 7-ACP meets the >99.5% purity standard required for downstream API synthesis, providing our partners with a secure and compliant supply source.
We invite global pharmaceutical manufacturers to collaborate with us to optimize their supply chains for fourth-generation cephalosporins. By leveraging our expertise in this advanced synthetic route, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our optimized manufacturing capabilities can enhance your production efficiency.
