Scaling High-Concentration Chlorinated Aromatic Dechlorination for Commercial Pharmaceutical Intermediates
Scaling High-Concentration Chlorinated Aromatic Dechlorination for Commercial Pharmaceutical Intermediates
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the need for greener, more efficient synthetic routes, particularly for persistent organic pollutants and valuable intermediates. A pivotal advancement in this domain is detailed in patent CN115928110A, which discloses a novel hydrogenation dechlorination method specifically designed for high-concentration chlorinated aromatic compounds. This technology represents a paradigm shift from traditional environmental remediation approaches, which typically handle dilute wastewater, to a robust synthetic methodology capable of processing reactant concentrations up to 300 g/L. By leveraging a specialized palladium-modified electrode prepared via cathode deposition, the process achieves exceptional metrics in current density, selectivity, and energy efficiency. For global procurement leaders and R&D directors, this innovation offers a compelling pathway to secure a reliable agrochemical intermediate supplier status while simultaneously addressing the critical challenges of catalyst cost and process scalability in the production of high-value dechlorinated aromatics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the electrochemical hydrodechlorination of chlorinated aromatic compounds (CAPs) has been predominantly researched and applied within the context of environmental water treatment, where the primary goal is the destruction of trace contaminants rather than the recovery of valuable products. Conventional methodologies often struggle with extremely low reactant concentrations, typically in the milligram-per-liter (mg/L) range, which necessitates massive reactor volumes to process meaningful quantities of material. Furthermore, these traditional systems frequently rely on high loadings of expensive palladium catalysts to drive the reaction, leading to prohibitive capital expenditures and operational costs that render the process economically unviable for commercial synthesis. The low current density and poor current efficiency associated with dilute systems result in sluggish reaction kinetics and extended processing times, creating bottlenecks that severely impact supply chain continuity and increase the overall carbon footprint of the manufacturing operation.
The Novel Approach
In stark contrast, the methodology outlined in patent CN115928110A introduces a high-concentration electrochemical system that fundamentally redefines the economic feasibility of dechlorination reactions. By operating with catholyte concentrations ranging from 5 g/L to an impressive 300 g/L, the process dramatically increases the space-time yield, allowing for significantly smaller reactor footprints and higher throughput capabilities. The core innovation lies in the utilization of a palladium-modified electrode fabricated through a specific cathode deposition technique involving polyvinyl pyrrolidone (PVP), which stabilizes the palladium nanoparticles and ensures a highly active catalytic surface with a loading as low as 0.5 to 5 g/m². This approach not only mitigates the reliance on bulk precious metals but also enables the system to sustain high current densities of up to 20 A/dm² while maintaining conversion rates exceeding 99% and selectivity above 98%, effectively turning a waste treatment challenge into a high-efficiency production asset for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Electrochemical Hydrogenation Dechlorination
The efficacy of this advanced dechlorination process is rooted in a sophisticated interplay of electrochemical reduction and surface catalysis mechanisms that occur at the modified cathode interface. The process initiates with the neutralization of the chlorinated aromatic acid in the alkaline catholyte, forming the corresponding carboxylate salt which serves as the active species for electrochemical reduction. This initial step is crucial for solubility and conductivity within the high-pH environment (pH 10-14), ensuring that the reactant molecules are readily available for transport to the electrode surface where the catalytic magic happens.

Following neutralization, the core transformation occurs at the cathode through a multi-electron transfer process where the carbon-chlorine bonds are cleaved and replaced by hydrogen atoms derived from water reduction. The presence of the palladium nanoparticles, stabilized by the PVP matrix, facilitates the adsorption of atomic hydrogen and the subsequent hydrodechlorination of the aromatic ring. This cathodic reaction is highly selective, preferentially removing chlorine atoms while preserving other functional groups such as carboxyl or amino groups, which is essential for maintaining the integrity of complex pharmaceutical intermediates. The generation of hydroxide ions as a byproduct helps maintain the alkalinity of the solution, creating a self-sustaining buffer system that supports continuous operation without the need for excessive pH adjustment.
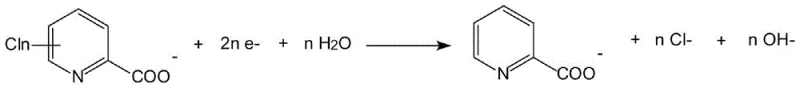
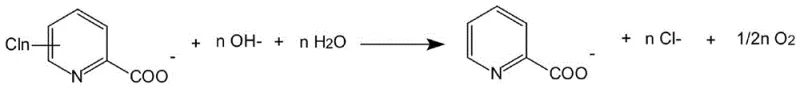
Complementing the cathodic reduction is the anodic reaction, typically involving the evolution of oxygen from the oxidation of hydroxide ions in the anolyte compartment. The use of an ion-exchange membrane effectively separates the anodic and cathodic chambers, preventing the re-oxidation of the dechlorinated product and ensuring high faradaic efficiency. The overall reaction stoichiometry demonstrates a clean conversion where the chlorinated substrate and water yield the dechlorinated product, chloride ions, and oxygen, with no hazardous reducing agents required. This mechanistic clarity allows for precise control over the reaction parameters, enabling operators to fine-tune current density and temperature to optimize the Specific Electrical Energy Consumption (SEEC), which has been demonstrated to be as low as 6.08 kW h kg⁻¹ for specific substrates like 3,6-dichloropicolinic acid.
How to Synthesize Picolinic Acid Efficiently
The synthesis of high-purity picolinic acid from its chlorinated precursors via this electrochemical route involves a streamlined sequence of operations that integrates electrode preparation, electrolyte formulation, and controlled electrolysis. The process begins with the meticulous fabrication of the palladium-modified cathode, where a conductive substrate such as nickel foam is treated and subjected to cathodic deposition in a solution containing palladium salts and PVP to achieve the optimal catalyst loading. Once the electrode is prepared, the chlorinated starting material is dissolved in a concentrated sodium hydroxide solution to form the catholyte, which is then introduced into a membrane-separated electrolytic cell equipped with a stainless steel anode. Detailed standardized synthesis steps see the guide below.
- Prepare the catholyte by dissolving the chlorinated aromatic compound (e.g., 3,6-dichloropicolinic acid) in an alkaline solution (pH 10-14) to achieve a high concentration between 5 g/L and 300 g/L.
- Utilize a palladium-modified electrode prepared via cathode deposition with polyvinyl pyrrolidone (PVP) as the cathode, and a stainless steel plate as the anode within an ion-exchange membrane separated cell.
- Conduct electrolysis at a current density of 1-20 A/dm² and temperature of 0-100°C until the desired conversion is reached, then separate and purify the dechlorinated product via acid precipitation or extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this high-concentration electrochemical dechlorination technology presents a multitude of strategic advantages that directly impact the bottom line and operational resilience. The ability to process reactants at concentrations orders of magnitude higher than traditional methods translates directly into a substantial reduction in solvent usage and waste generation, simplifying downstream purification and minimizing the environmental burden associated with large-volume liquid handling. Furthermore, the high current density and rapid reaction kinetics enable a drastic simplification of the production schedule, allowing facilities to achieve higher output rates with existing infrastructure or to downsize capital equipment requirements for new installations, thereby optimizing capital allocation.
- Cost Reduction in Manufacturing: The economic model of this process is heavily favored by the significant reduction in precious metal catalyst requirements, as the specialized deposition technique ensures high utilization efficiency of palladium at loadings as low as 0.5 g/m². By eliminating the need for stoichiometric chemical reducing agents and minimizing energy consumption through optimized current efficiency, the operational expenditure per kilogram of product is markedly decreased compared to conventional catalytic hydrogenation or low-concentration electrochemical treatments. This structural cost advantage provides a buffer against raw material price volatility and enhances the overall margin profile for high-volume commodity intermediates.
- Enhanced Supply Chain Reliability: The robustness of the electrochemical system, capable of handling a wide variety of chlorinated aromatic substrates including phenols, benzoic acids, and pyridine derivatives, offers supply chain teams greater flexibility in sourcing raw materials. The continuous or semi-continuous nature of the electrolytic process, combined with the stability of the modified electrodes, ensures consistent production output and reduces the risk of batch-to-batch variability that can plague traditional chemical synthesis. This reliability is critical for maintaining just-in-time inventory levels and meeting the stringent delivery schedules demanded by downstream pharmaceutical and agrochemical manufacturers.
- Scalability and Environmental Compliance: From a sustainability perspective, the process aligns perfectly with modern green chemistry principles by utilizing electricity as the primary reagent and generating benign byproducts like chloride salts and oxygen. The absence of hazardous reducing agents simplifies safety protocols and waste disposal procedures, facilitating easier regulatory compliance across different jurisdictions. The modular nature of electrolytic cells allows for straightforward linear scale-up from pilot to commercial production, ensuring that the process performance observed in the laboratory can be faithfully replicated in multi-ton manufacturing facilities without the engineering uncertainties often associated with scaling complex catalytic reactions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical dechlorination technology, drawing directly from the experimental data and beneficial effects described in the patent literature. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing workflows or for procurement specialists assessing the quality and consistency of the resulting intermediates.
Q: How does this method improve upon traditional low-concentration dechlorination processes?
A: Unlike conventional methods that treat dilute wastewater (mg/L level) with high catalyst loading, this patent enables processing at high concentrations (up to 300 g/L) with significantly lower palladium loading (0.5-5 g/m²), drastically improving space-time yield and economic viability.
Q: What is the energy efficiency of this electrochemical dechlorination process?
A: The process achieves high current efficiency and low energy consumption, with Specific Electrical Energy Consumption (SEEC) values reported as low as 6.08 kW h kg⁻¹ for 3,6-dichloropicolinic acid, making it suitable for cost-sensitive industrial manufacturing.
Q: Can this technology be applied to substrates other than chloropicolinic acid?
A: Yes, the method demonstrates high selectivity and conversion for a broad range of chlorinated aromatic compounds including chlorophenols, chlorobenzoic acids, and chlorophenoxyacetic acids, converting them into their respective dechlorinated analogues with yields often exceeding 95%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Picolinic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced electrochemical technologies in reshaping the landscape of fine chemical production. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the one described in CN115928110A can be seamlessly transitioned into robust industrial operations. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the exacting standards required by the global pharmaceutical and agrochemical industries, guaranteeing that our clients receive high-purity OLED material and pharmaceutical intermediates that drive their own success.
We invite you to engage with our technical procurement team to explore how this cutting-edge dechlorination method can be tailored to your specific production needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits for your specific product portfolio. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a trusted partner in delivering cost-effective and sustainable chemical solutions.
