Advanced Manufacturing of (R)-3-Aminobutanol: A Strategic Breakthrough for API Intermediates
Advanced Manufacturing of (R)-3-Aminobutanol: A Strategic Breakthrough for API Intermediates
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective pathways for synthesizing critical chiral intermediates, particularly those serving high-value antiretroviral therapies. Patent CN110668958A introduces a transformative methodology for the preparation of (R)-3-aminobutanol, a pivotal building block in the synthesis of Dolutegravir, a leading HIV integrase inhibitor. This technology addresses long-standing challenges in the fine chemical sector by replacing complex, hazardous, or low-yield synthetic routes with a streamlined three-step process involving reductive amination and chiral resolution. For R&D directors and procurement strategists, this patent represents a significant opportunity to optimize the supply chain for anti-AIDS medications and other therapeutic areas, ensuring consistent quality while mitigating the risks associated with traditional chiral synthesis methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (R)-3-aminobutanol has been plagued by significant technical and economic hurdles that hinder efficient commercial manufacturing. Prior art methods, such as those utilizing morpholine as a solvent and base for resolution, often struggle with the complete removal of toxic amine impurities, compromising the purity profile required for API intermediates. Other approaches relying on chiral induction with organolithium reagents like n-butyllithium introduce severe safety hazards and require cryogenic conditions around -78°C, drastically increasing energy consumption and capital expenditure. Furthermore, enzymatic routes, while selective, frequently involve multi-step protection and deprotection sequences with expensive reagents like TBDMSCl and hydrazine hydrate, leading to cumbersome operations that are difficult to translate from the laboratory to kiloton-scale production facilities without substantial yield losses.
The Novel Approach
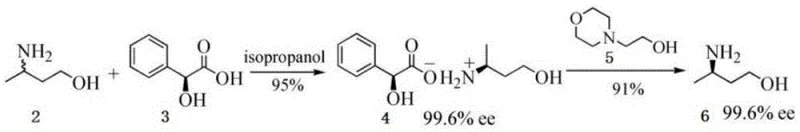
In stark contrast to these legacy technologies, the method disclosed in CN110668958A offers a remarkably direct and industrially friendly pathway. The core innovation lies in the initial reductive amination of 4-hydroxy-2-butanone with ammonia, utilizing robust heterogeneous catalysts like Raney Nickel or Palladium on Carbon under moderate hydrogen pressures of 1.2-1.5 MPa. This is followed by a highly selective resolution step using (S)-mandelic acid, which effectively separates the desired enantiomer through diastereomeric salt formation. The final liberation of the free amine is achieved through a mild alkalization process using sodium methoxide. This sequence eliminates the need for hazardous cryogenic reagents and complex enzymatic cascades, resulting in a process that is not only safer but also significantly more amenable to continuous improvement and scale-up in a GMP environment.
Mechanistic Insights into Reductive Amination and Chiral Resolution
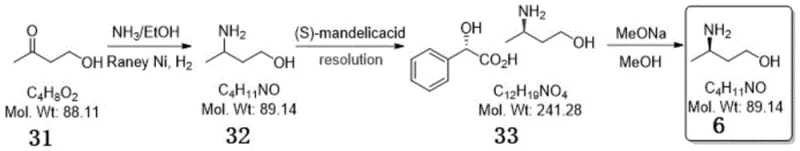
The success of this synthetic strategy hinges on the precise control of the reductive amination mechanism and the thermodynamics of the subsequent resolution. In the first step, the ketone group of 4-hydroxy-2-butanone reacts with ammonia to form an imine intermediate, which is immediately reduced in situ by hydrogen activated on the surface of the Raney Nickel catalyst. The use of a polar solvent system, such as ethanol, facilitates the solubility of reactants while maintaining the stability of the catalyst. Critical to the process is the maintenance of hydrogen pressure between 1.2 MPa and 1.5 MPa, which ensures complete conversion of the imine while minimizing the formation of secondary amine by-products. The reaction temperature is carefully controlled between 40°C and 50°C to balance reaction kinetics with selectivity, ensuring that the hydroxyl group remains intact and unreacted during the hydrogenation phase.
Following the formation of racemic 3-aminobutanol, the optical purity is established through a classical yet optimized resolution mechanism. The interaction between the racemic amine and (S)-mandelic acid relies on the formation of diastereomeric salts with distinct solubility profiles in polar solvents like ethanol or isopropanol. By meticulously controlling the molar ratio of the resolving agent to the racemate (optimized at 1:0.4 to 1:0.5) and managing the crystallization temperature gradient from 75°C down to 13°C, the process selectively precipitates the less soluble diastereomer. This solid-state separation is further enhanced by a slurry purification step, which washes away the mother liquor containing the unwanted enantiomer. The final alkalization with sodium methoxide simply reverses the salt formation, releasing the free (R)-amine with exceptional chemical purity of 99.9% and an enantiomeric excess exceeding 99.9%.
How to Synthesize (R)-3-Aminobutanol Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters regarding pressure, temperature, and stoichiometry to maximize yield and purity. The process begins with the ammoniation of the ketone substrate, followed by high-pressure hydrogenation, and concludes with the resolution and free-base liberation steps. Operators must ensure rigorous gas置换 (purging) of the reactor with nitrogen and hydrogen to maintain a safe and active catalytic environment. The detailed standardized operating procedures, including specific work-up protocols like toluene extraction for water removal and precise distillation cuts, are essential for reproducing the high-quality results reported in the patent examples.
- Perform reductive amination of 4-hydroxy-2-butanone with ammonia using a heterogeneous catalyst like Raney Nickel at 1.2-1.5 MPa hydrogen pressure.
- React the resulting racemic 3-aminobutanol with (S)-mandelic acid in a polar solvent to form diastereomeric salts for resolution.
- Treat the resolved mandelate salt with an alkali metal alkoxide solution, such as sodium methoxide in methanol, to liberate the final high-purity (R)-3-aminobutanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers compelling strategic benefits that extend beyond mere technical feasibility. The shift away from exotic chiral catalysts and hazardous organometallic reagents towards commodity chemicals fundamentally alters the cost structure and risk profile of the supply chain. By utilizing widely available starting materials like 4-hydroxy-2-butanone and ammonia, manufacturers can secure raw material supplies more reliably and at a fraction of the cost associated with specialized chiral pool starting materials. This resilience against raw material volatility is crucial for maintaining consistent production schedules for high-demand antiviral medications.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents such as n-butyllithium or specialized enzymes leads to a substantial decrease in direct material costs. Furthermore, the use of heterogeneous catalysts like Raney Nickel allows for easier recovery and potential reuse, reducing waste disposal costs. The simplified downstream processing, which relies on standard crystallization and distillation rather than complex chromatographic separations, significantly lowers utility consumption and labor hours per kilogram of product, driving overall manufacturing efficiency.
- Enhanced Supply Chain Reliability: Dependence on single-source suppliers for niche chiral auxiliaries is a major vulnerability in pharmaceutical supply chains. This method mitigates that risk by relying on bulk chemicals that are produced by multiple global vendors. The robustness of the chemical steps, which tolerate standard industrial equipment and conditions, ensures that production can be easily transferred between different manufacturing sites or scaled up rapidly to meet surges in demand without requiring specialized infrastructure investments.
- Scalability and Environmental Compliance: The process is inherently designed for scale, avoiding the pitfalls of enzymatic methods that often face mass transfer limitations in large reactors. The absence of heavy metal contaminants from homogeneous catalysts simplifies the purification train and reduces the environmental burden of heavy metal waste streams. Additionally, the high atom economy of the reductive amination step and the recyclability of solvents like ethanol and toluene align with modern green chemistry principles, facilitating easier regulatory approval and reducing the carbon footprint of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy synthesis routes in terms of safety, purity, and operational simplicity.
Q: What is the primary advantage of this synthesis method over enzymatic routes?
A: Unlike enzymatic methods which often suffer from cumbersome processes, high reagent costs, and difficulties in industrial scale-up, this chemical route utilizes robust heterogeneous catalysis and standard crystallization techniques, ensuring better scalability and lower operational complexity.
Q: How does this method ensure high optical purity for API manufacturing?
A: The method employs a classical yet highly effective chiral resolution step using (S)-mandelic acid. By optimizing the molar ratios and crystallization temperatures, the process achieves an ee value greater than 99.9% and chemical purity of 99.9%, meeting stringent pharmaceutical standards.
Q: Are the raw materials for this process readily available for large-scale production?
A: Yes, the process relies on commodity chemicals such as 4-hydroxy-2-butanone, ammonia, and (S)-mandelic acid. This avoids the supply chain bottlenecks associated with specialized chiral catalysts or expensive bio-enzymes found in alternative synthetic pathways.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-3-Aminobutanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global fight against HIV and other diseases. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory patent data to industrial reality is seamless. We are committed to delivering (R)-3-aminobutanol with stringent purity specifications, leveraging our rigorous QC labs to verify every batch meets the >99.9% purity and ee standards required for next-generation API synthesis.
We invite pharmaceutical partners to collaborate with us to leverage this advanced manufacturing technology for their supply chains. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized process can enhance your production efficiency and reduce time-to-market for your critical therapeutic programs.
