Advanced Manufacturing of Chiral (R)-3-Aminobutanol: A Breakthrough in Dolutegravir Intermediate Production
The global pharmaceutical landscape is continuously evolving, driven by the urgent need for effective antiretroviral therapies to combat HIV/AIDS. Within this critical sector, Dolutegravir has emerged as a cornerstone second-generation integrase inhibitor, renowned for its high barrier to resistance and excellent tolerability profile. The manufacturing of this vital medication relies heavily on the consistent supply of high-quality chiral intermediates, specifically (R)-3-aminobutanol. A recent technological breakthrough documented in patent CN115594600A introduces a transformative synthesis method that addresses long-standing challenges in producing this key building block. This innovation not only streamlines the production workflow but also significantly enhances the safety and economic viability of the supply chain for major pharmaceutical manufacturers worldwide.
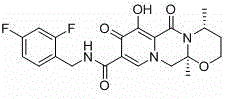
The structural complexity of Dolutegravir necessitates precise stereochemical control during the synthesis of its precursors. As illustrated in the molecular architecture below, the integration of the chiral side chain is fundamental to the drug's biological activity and therapeutic efficacy. Any deviation in optical purity or the presence of trace impurities can compromise the final API quality, making the choice of synthetic route a critical decision for R&D and production teams alike.
Historically, the synthesis of (R)-3-aminobutanol has been fraught with technical and logistical hurdles that impact both cost and scalability. Conventional approaches often rely on the use of highly reactive and dangerous reducing agents such as lithium aluminum hydride (LiAlH4) or borane complexes. These reagents, while effective in laboratory settings, present severe safety risks in large-scale industrial environments due to their pyrophoric nature and sensitivity to moisture. Furthermore, alternative strategies involving chemical resolution of racemic mixtures, such as those utilizing mandelic acid, suffer from inherent yield limitations, theoretically capping the maximum efficiency at 50% unless dynamic kinetic resolution is employed. Other methods involving Lewis acids like zinc chloride generate copious amounts of solid waste, creating significant environmental burdens and increasing the cost of waste disposal and regulatory compliance.
In stark contrast, the novel approach detailed in CN115594600A offers a robust and scalable alternative that circumvents these traditional pitfalls. By utilizing a combination of sodium or potassium borohydride alongside a methylating agent such as dimethyl sulfate or methyl iodide, the process achieves a direct, one-step reduction of (R)-3-aminobutyric acid. This methodology operates under relatively mild conditions, typically between 20°C and 60°C, and utilizes common organic solvents like tetrahydrofuran or ethylene glycol dimethyl ether. The result is a process that delivers yields exceeding 85% with chemical and optical purities greater than 99%, effectively eliminating the need for hazardous reagents and complex purification sequences associated with older technologies.
Mechanistic Insights into Borohydride-Mediated Reduction with In-Situ Activation
The core innovation of this synthesis lies in the unique interaction between the borohydride species and the methylating agent, which facilitates the reduction of the carboxylic acid moiety—a transformation that borohydride alone typically cannot achieve efficiently. In standard organic chemistry, carboxylic acids are resistant to reduction by sodium borohydride due to the formation of unreactive carboxylate salts. However, the presence of a methylating agent in this system likely promotes the in-situ formation of a reactive intermediate, such as a mixed anhydride or an activated ester species, which is susceptible to nucleophilic attack by the hydride ion. This mechanistic pathway allows for the smooth conversion of the acid to the corresponding alcohol without the need for pre-activation steps or harsh reaction conditions that could lead to racemization of the chiral center.
Furthermore, the control of impurities is a critical aspect of this mechanism, particularly regarding the prevention of aminomethylation side reactions. The patent data indicates that by carefully controlling the molar ratios of the reactants—specifically maintaining a ratio of acid to borohydride to methylating agent around 1:2.5:1.25—the formation of unwanted N-methylated byproducts is integrally suppressed. This precise stoichiometric balance ensures that the hydride source is available primarily for the reduction of the carbonyl group rather than engaging in side reactions with the amine functionality. The subsequent workup involving acid quenching and base adjustment further aids in separating the desired amino alcohol from inorganic salts, ensuring that the final distilled product meets the stringent purity specifications required for pharmaceutical applications.
How to Synthesize (R)-3-Aminobutanol Efficiently
The implementation of this synthesis route requires careful attention to reaction parameters to maximize yield and safety. The process begins with the preparation of the reaction vessel under an inert nitrogen atmosphere to prevent moisture ingress, which could degrade the borohydride reagent. Following the addition of the solvent and reagents, temperature control during the addition of the methylating agent is paramount to manage the exothermic nature of the reaction. Once the addition is complete, heating the mixture to approximately 60°C drives the reduction to completion. For a comprehensive guide on the specific operational parameters and safety protocols, please refer to the standardized procedure outlined below.
- Prepare the reaction vessel under nitrogen protection with an organic solvent such as tetrahydrofuran or ethylene glycol dimethyl ether.
- Add sodium or potassium borohydride and (R)-3-aminobutyric acid to the solvent, cooling the mixture with ice water to control exothermic activity.
- Slowly drip a solution of the methylating agent (dimethyl sulfate or methyl iodide) while maintaining temperature between 20-30°C, then heat to 60°C for completion.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis method translates into tangible strategic benefits that extend beyond simple unit cost metrics. The shift away from hazardous reagents like lithium aluminum hydride and borane eliminates the need for specialized storage facilities and expensive safety monitoring equipment, thereby reducing capital expenditure and operational overhead. Additionally, the simplification of the workflow from a multi-step or resolution-based process to a direct one-step reduction significantly shortens the manufacturing cycle time. This efficiency gain enhances the agility of the supply chain, allowing for faster response to market demand fluctuations and reducing the inventory holding costs associated with work-in-progress materials.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the substitution of high-cost, high-risk reagents with commodity chemicals. Sodium borohydride is significantly less expensive than lithium aluminum hydride or specialized chiral catalysts, and its handling does not require the same level of costly safety infrastructure. Moreover, the high yield of over 85% minimizes raw material waste, ensuring that a greater proportion of the input mass is converted into valuable product. The elimination of heavy metal catalysts also removes the cost burden associated with metal scavenging and hazardous waste disposal, contributing to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on specialized or regulated reagents that may face availability constraints. By utilizing widely available starting materials such as (R)-3-aminobutyric acid and common solvents, this method diversifies the supply base and reduces dependency on single-source vendors for exotic chemicals. The robustness of the reaction conditions, which tolerate standard industrial equipment without requiring exotic alloys or extreme pressure ratings, further ensures that production can be maintained consistently across different manufacturing sites, mitigating the risk of supply disruptions due to equipment failure or maintenance downtime.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental sustainability intensify, the ability to demonstrate a green chemistry profile is a significant competitive advantage. This synthesis route generates minimal solid waste compared to Lewis acid-catalyzed methods, simplifying effluent treatment and reducing the environmental footprint of the manufacturing process. The absence of toxic heavy metals in the waste stream facilitates easier compliance with international environmental regulations, such as REACH or EPA guidelines. This clean profile not only future-proofs the supply chain against tightening regulations but also aligns with the corporate social responsibility goals of major pharmaceutical clients who prioritize sustainable sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a clear understanding of the process capabilities and limitations for potential adopters.
Q: Why is this new synthesis method safer than traditional lithium aluminum hydride reduction?
A: Traditional methods often utilize lithium aluminum hydride or borane, which are pyrophoric and pose significant fire and explosion risks requiring specialized equipment. This patented method utilizes sodium or potassium borohydride combined with a methylating agent, which are significantly more stable and manageable in standard industrial reactors, drastically reducing operational safety hazards.
Q: How does this process address environmental concerns regarding solid waste?
A: Previous methodologies, particularly those employing Lewis acids like anhydrous zinc chloride, generate substantial amounts of hazardous solid waste that complicate disposal and increase environmental compliance costs. The novel borohydride-based route described in CN115594600A eliminates the need for these heavy metal catalysts, resulting in a cleaner reaction profile and simplified waste treatment protocols.
Q: What represents the primary cost advantage for procurement teams adopting this route?
A: The primary cost driver is the replacement of expensive and hazardous reducing agents with commodity chemicals like sodium borohydride. Furthermore, the elimination of complex resolution steps or multi-step sequences reduces overall processing time and utility consumption, leading to a more economically efficient manufacturing process without compromising the high optical purity required for API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-3-Aminobutanol Supplier
The technical advancements described in patent CN115594600A represent a significant leap forward in the manufacturing of chiral pharmaceutical intermediates. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial realities. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of (R)-3-aminobutanol meets the exacting standards required for the synthesis of life-saving antiretroviral medications like Dolutegravir.
We invite global partners to collaborate with us to leverage this superior synthesis route for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your production of high-purity pharmaceutical intermediates is both economically optimized and securely supplied.
