Scalable Biocatalytic Production of High-Purity (R)-3-Aminobutanol for HIV Drug Intermediates
Introduction to Advanced Biocatalytic Synthesis
The pharmaceutical industry's relentless pursuit of efficient, sustainable, and scalable synthetic routes for critical chiral intermediates has led to significant breakthroughs in enzyme engineering. A pivotal development in this domain is documented in patent CN106754806B, which details an improved transaminase specifically designed for the preparation of (R)-3-aminobutanol. This compound serves as a vital chiral building block for the synthesis of Dolutegravir, a potent integrase inhibitor used in the treatment of HIV-1 infection. The patent discloses a genetically engineered variant derived from Aspergillus terreus NIH2624, exhibiting superior catalytic performance compared to its wild-type counterpart. By leveraging site-directed mutagenesis, specifically targeting residues such as position 215, the inventors have created a biocatalyst capable of operating at high substrate loadings while maintaining exceptional enantioselectivity. This technological leap addresses long-standing bottlenecks in the manufacturing of antiretroviral intermediates, offering a pathway that combines high yield with environmental sustainability.
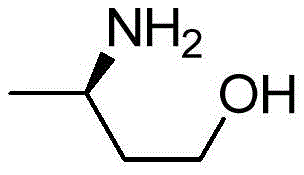
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (R)-3-aminobutanol has relied heavily on traditional chemical methodologies that present substantial challenges for industrial scale-up. One prevalent approach involves the kinetic resolution of racemic mixtures, a process inherently limited by a maximum theoretical yield of 50% and often requiring complex separation techniques to isolate the desired enantiomer. Furthermore, alternative chemical routes frequently employ hazardous reagents such as lithium aluminum hydride for reduction steps, introducing severe safety risks related to exothermic reactions and handling of pyrophoric materials on a large scale. Another strategy utilizes chiral pool starting materials like D-alanine, but this necessitates multi-step synthetic sequences that accumulate impurities and drive up the cost of goods due to low overall atom economy. These conventional methods also often struggle with waste generation, requiring extensive purification protocols to meet the stringent purity specifications demanded by regulatory bodies for pharmaceutical intermediates.
The Novel Approach
In stark contrast, the biocatalytic strategy outlined in the patent data introduces a streamlined, one-step asymmetric transamination process that fundamentally reshapes the production landscape. The improved transaminase facilitates the direct conversion of the prochiral ketone, 4-hydroxy-2-butanone, into the target chiral amine with remarkable efficiency. Unlike previous biological attempts that were restricted to low substrate concentrations (around 26.4g/L) and required the addition of organic cosolvents like DMSO to maintain enzyme solubility or activity, this novel enzyme operates robustly in aqueous media. The ability to tolerate substrate concentrations as high as 50g/L significantly enhances space-time yield, allowing manufacturers to produce more product per batch without expanding reactor footprint. Moreover, the elimination of organic cosolvents simplifies the downstream workup, reducing the burden on solvent recovery systems and minimizing the environmental impact associated with volatile organic compound (VOC) emissions.
Mechanistic Insights into Engineered Transaminase Catalysis
The core of this technological advancement lies in the precise molecular engineering of the transaminase active site to optimize substrate binding and catalytic turnover. The wild-type enzyme, while functional, suffers from steric hindrance or suboptimal electronic environments when confronted with high concentrations of the hydroxy-ketone substrate. The improved variant incorporates specific amino acid substitutions, most notably the mutation of Serine at position 215 to Proline (S215P), along with other potential modifications at positions 55 and 58 depending on the specific mutant library screened. These structural alterations likely rigidify the protein backbone or reshape the substrate binding pocket, thereby enhancing the affinity for 4-hydroxy-2-butanone and stabilizing the transition state during the pyridoxal-5'-phosphate (PLP) dependent reaction cycle. The result is a catalyst that not only accelerates the rate of amination but also maintains strict stereocontrol, ensuring the exclusive formation of the (R)-enantiomer which is critical for the biological activity of the final drug product.
From an impurity control perspective, the high enantioselectivity (ee value > 99.5%) of the improved transaminase is a decisive factor for pharmaceutical quality assurance. In chemical synthesis, the formation of the undesired (S)-enantiomer or over-reduced byproducts often necessitates costly recrystallization or chromatographic purification steps to achieve the required optical purity. The enzymatic route effectively bypasses these issues by leveraging the inherent chirality of the enzyme's active site, which acts as a molecular template that only accommodates the pro-chiral substrate in a specific orientation. This "chiral gatekeeping" mechanism ensures that side reactions are minimized, leading to a cleaner crude reaction profile. Consequently, the isolation of (R)-3-aminobutanol becomes more straightforward, typically involving simple extraction and distillation, which preserves the integrity of the heat-sensitive product and reduces the risk of racemization during processing.
How to Synthesize (R)-3-Aminobutanol Efficiently
Implementing this biocatalytic route requires a systematic approach to fermentation, enzyme formulation, and reaction engineering to maximize the benefits of the improved transaminase. The process begins with the expression of the engineered gene in a suitable host, such as E. coli BL21(DE3), followed by the preparation of the biocatalyst in a stable form, such as freeze-dried enzyme powder or whole-cell lysate. The reaction itself is conducted under mild physiological conditions, utilizing isopropylamine as a cheap and effective amine donor to drive the equilibrium towards product formation. Detailed operational parameters, including precise pH control and temperature management, are critical to sustaining high enzyme activity throughout the conversion. For a comprehensive guide on the specific laboratory-scale protocols and optimization strategies derived from the patent examples, please refer to the standardized synthesis steps below.
- Prepare a reaction mixture containing phosphate buffer (pH 7.0-9.0), isopropylamine as the amine donor, and pyridoxal phosphate (PLP) as the cofactor.
- Add the substrate 4-hydroxy-2-butanone to a final concentration of up to 50g/L and introduce the improved transaminase enzyme powder or cell lysate.
- Incubate the reaction at 30-40°C with pH control, followed by heat denaturation of the enzyme and solvent extraction to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this improved transaminase technology translates into tangible strategic advantages that extend beyond mere technical feasibility. The shift from hazardous chemical synthesis to a benign enzymatic process mitigates significant operational risks associated with handling dangerous reagents, thereby lowering insurance premiums and safety compliance costs. Furthermore, the ability to run reactions at high substrate concentrations without organic cosolvents directly impacts the cost structure by reducing the volume of solvents that need to be purchased, recovered, or disposed of. This efficiency gain allows for a more compact manufacturing footprint, enabling existing facilities to increase throughput without capital-intensive expansion. The robustness of the enzyme also implies a more reliable supply chain, as the fermentation-based production of the biocatalyst is less susceptible to the raw material volatility that often plagues petrochemical-derived reagents.
- Cost Reduction in Manufacturing: The elimination of expensive chiral starting materials and hazardous reducing agents like lithium aluminum hydride drastically simplifies the bill of materials. By replacing multi-step chemical sequences with a single enzymatic transformation, manufacturers can significantly reduce labor, energy, and equipment utilization costs. The removal of organic cosolvents further decreases the expense associated with solvent procurement and waste treatment, leading to a leaner and more cost-effective production model that enhances overall margin potential for the final API intermediate.
- Enhanced Supply Chain Reliability: Relying on a fermentation-derived enzyme offers a more stable and scalable source of catalytic activity compared to precious metal catalysts which are subject to geopolitical supply constraints. The improved stability of the mutant enzyme ensures consistent batch-to-batch performance, reducing the likelihood of production delays caused by catalyst failure or inconsistent reaction rates. This reliability is crucial for meeting the rigorous delivery schedules of global pharmaceutical clients who require uninterrupted supply of critical HIV drug intermediates to maintain their own production timelines.
- Scalability and Environmental Compliance: The aqueous nature of the reaction medium aligns perfectly with green chemistry principles, facilitating easier regulatory approval and reducing the environmental footprint of the manufacturing site. Scaling up from laboratory to commercial production is streamlined because the process avoids the complex heat and mass transfer issues often encountered in heterogeneous chemical catalysis. The simplified workup procedure, which avoids complex quenching of reactive chemicals, ensures that the transition to multi-ton production can be achieved with minimal process re-engineering, securing long-term supply continuity.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the viability of this technology for their specific applications, we have compiled answers to common inquiries regarding the performance and implementation of the improved transaminase. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for assessing the process capabilities. Understanding these nuances is essential for R&D teams planning to integrate this biocatalytic step into their existing synthetic workflows for antiretroviral drug manufacturing.
Q: How does the improved transaminase in CN106754806B compare to wild-type enzymes?
A: The improved transaminase features specific amino acid mutations, such as S215P, which significantly enhance catalytic activity and substrate tolerance compared to the wild-type Aspergillus terreus enzyme, allowing for higher substrate concentrations up to 50g/L without organic cosolvents.
Q: What are the safety advantages of this enzymatic route over chemical synthesis?
A: Unlike traditional chemical methods that utilize hazardous reducing agents like lithium aluminum hydride or require multi-step chiral pool synthesis, this biocatalytic process operates under mild aqueous conditions (30-40°C), eliminating explosion risks and simplifying waste treatment.
Q: Is organic solvent required for this transamination reaction?
A: No, a key advantage of this improved enzyme is its ability to function efficiently in purely aqueous buffer systems, removing the need for toxic organic cosolvents like DMSO or acetonitrile often required in previous biocatalytic attempts, thereby reducing downstream processing costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-3-Aminobutanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the improved transaminase technology described in CN106754806B for the production of high-value pharmaceutical intermediates. As a seasoned CDMO partner, we possess the technical expertise to translate such innovative laboratory findings into robust, commercial-scale manufacturing processes. Our facilities are equipped to handle diverse biocatalytic pathways, with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch of (R)-3-aminobutanol meets the exacting standards required for HIV drug synthesis, guaranteeing consistency and quality for our global partners.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for chiral intermediates. By leveraging our advanced fermentation and downstream processing capabilities, we can help you realize the full economic and operational benefits of this enzymatic route. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing excellence can support your drug development and commercialization goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
