Optimized Synthetic Route for Quetiapine Hemifumarate: Enhancing Purity and Scalability for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust manufacturing processes that balance safety, efficiency, and regulatory compliance, particularly for high-volume atypical antipsychotic agents. Patent CN103772319A introduces a refined synthetic methodology for Quetiapine Hemifumarate, a critical active pharmaceutical ingredient (API) used globally for the treatment of schizophrenia and bipolar disorder. This technical disclosure outlines a strategic shift away from hazardous traditional reagents toward a more sustainable and operationally simple pathway. By initiating the synthesis with readily available commodities such as o-bromonitrobenzene and thiosalicylic acid, the process circumvents the severe safety risks associated with phosgene derivatives. For R&D directors and supply chain leaders, this represents a significant opportunity to optimize the production of high-purity pharmaceutical intermediates while mitigating environmental liabilities. The following analysis details the mechanistic advantages and commercial viability of this approach.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Quetiapine and its salts has relied heavily on starting materials like 2-diaminodiphenyl sulfide. While chemically feasible, these legacy routes impose substantial burdens on modern manufacturing facilities due to their reliance on aggressive reagents such as phenyl chloroformate or triphosgene. These substances are not only acutely toxic and corrosive but also necessitate specialized containment infrastructure and rigorous waste neutralization protocols, driving up operational expenditures. Furthermore, the esterification and cyclization steps in these traditional pathways often proceed under violent conditions that compromise the stability of intermediates. This instability frequently leads to the generation of difficult-to-remove impurities and inconsistent yields, creating bottlenecks in the supply chain for reliable API intermediate suppliers. The accumulation of urea-type byproducts and the difficulty in controlling exothermic reactions during scale-up further exacerbate the risk profile of these conventional methods.
The Novel Approach
In stark contrast, the methodology disclosed in CN103772319A constructs the dibenzothiazepine core through a convergent strategy that prioritizes mild reaction conditions and atom economy. The process initiates with a nucleophilic aromatic substitution between o-bromonitrobenzene and thiosalicylic acid, followed by a selective reduction and acid-catalyzed cyclization. This sequence effectively bypasses the need for toxic carbonylating agents entirely. As illustrated in the reaction scheme below, the pathway flows logically through stable intermediates (Z1, Z2, Z3) before the final piperazine attachment and salt formation. This structural assembly not only simplifies the purification workflow but also enhances the overall safety of the plant environment. By replacing hazardous reagents with safer alternatives like ferrous sulfate for reduction and phosphorus oxychloride for activation, the process achieves a level of operational simplicity that is ideal for commercial scale-up of complex pharmaceutical intermediates.
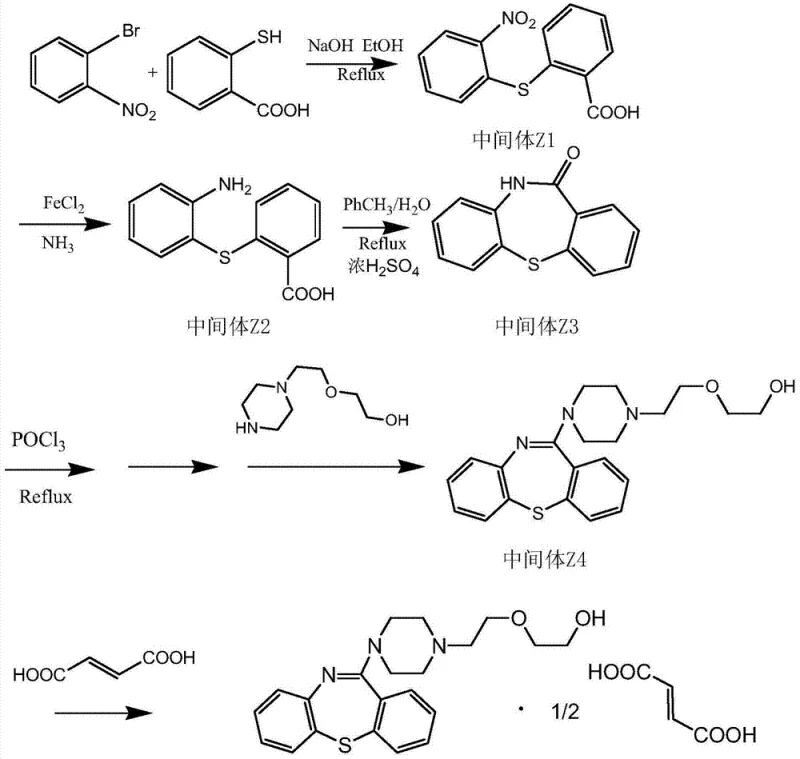
Mechanistic Insights into Fe-Catalyzed Reduction and Acid Cyclization
The core of this synthetic innovation lies in the efficient construction of the tricyclic dibenzothiazepine skeleton. The initial displacement reaction leverages the electron-withdrawing nature of the nitro group on the benzene ring to facilitate the attack of the thiolate anion derived from thiosalicylic acid. This step is critical as it establishes the diphenyl sulfide linkage with high regioselectivity. Following this, the reduction of the nitro group to an amine using ferrous sulfate in an ammoniacal medium is a classic yet highly effective transformation. Unlike catalytic hydrogenation which requires high-pressure equipment, this chemical reduction proceeds smoothly at atmospheric pressure, reducing capital expenditure requirements. The subsequent cyclization step utilizes concentrated sulfuric acid in a toluene-water biphasic system. The acid promotes the intramolecular nucleophilic attack of the amino group onto the carboxylic acid moiety (activated in situ), closing the seven-membered ring to form the ketone Intermediate Z3. This specific mechanism ensures that the ring closure occurs cleanly without the scrambling of substituents often seen in harsher thermal cyclizations.
From an impurity control perspective, this route offers distinct advantages over the triphosgene-mediated pathways. In traditional methods, the reaction of amines with phosgene equivalents can lead to over-carbonylation or the formation of symmetrical ureas if moisture control is not absolute. The current pathway avoids these carbonyl-transfer risks entirely until the very end, where the piperazine side chain is attached. Even then, the activation of the ketone (Z3) via Vilsmeier-Haack type conditions using POCl3 generates a reactive imidoyl chloride species that reacts selectively with the piperazine nitrogen. This selectivity minimizes the formation of bis-alkylated byproducts or O-alkylated impurities. Consequently, the final crude product requires less intensive chromatographic purification, relying instead on standard crystallization techniques to achieve the stringent purity specifications required for psychiatric medications. This mechanistic clarity provides R&D teams with a predictable profile for validation and regulatory filing.
How to Synthesize Quetiapine Hemifumarate Efficiently
Implementing this synthetic route requires precise control over reaction parameters, particularly temperature and stoichiometry, to maximize the yield of each intermediate. The process is designed to be telescoped where possible, although isolating the key intermediates Z1, Z2, and Z3 allows for rigorous quality control checkpoints. The initial displacement in ethanol requires careful pH management with sodium hydroxide to ensure complete deprotonation of the thiol without hydrolyzing the nitro group. The reduction step demands strict temperature control during the addition of ferrous sulfate to manage the exotherm effectively. For the final salt formation, the stoichiometric ratio of fumaric acid to the free base is critical to ensure the formation of the hemifumarate salt rather than other stoichiometric variants. Detailed standard operating procedures regarding mixing speeds, addition rates, and crystallization cooling profiles are essential for reproducibility. The standardized synthesis steps for this process are outlined in the guide below.
- Displacement Reaction: React o-bromonitrobenzene with thiosalicylic acid in ethanol using sodium hydroxide to form Intermediate Z1.
- Reduction: Reduce Intermediate Z1 using ferrous sulfate and ammonia to obtain Intermediate Z2 (2-amino-2'-carboxyl diphenyl sulfide).
- Cyclization: Cyclize Intermediate Z2 in toluene/water with concentrated sulfuric acid to form the dibenzo thiazepine ketone (Intermediate Z3).
- Condensation: React Intermediate Z3 with POCl3 followed by N-(2-hydroxyethyl)piperazine derivative to form Intermediate Z4.
- Salt Formation: Dissolve Intermediate Z4 in acetone and react with fumaric acid to crystallize Quetiapine Hemifumarate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this synthetic route offers tangible economic and logistical benefits beyond mere chemical elegance. The primary driver of value is the elimination of high-cost, high-risk reagents. Triphosgene and phenyl chloroformate are not only expensive to purchase but also incur significant costs related to storage, handling, and the disposal of hazardous waste streams. By substituting these with commodity chemicals like o-bromonitrobenzene and thiosalicylic acid, the raw material cost base is significantly lowered. Furthermore, the stability of the intermediates reduces the risk of batch failures due to degradation during storage or transport between processing stages. This reliability translates directly into more consistent lead times and a more resilient supply chain for cost reduction in API manufacturing. The simplified workup procedures, which rely largely on filtration and crystallization rather than complex distillations or extractions, also reduce utility consumption and cycle times.
- Cost Reduction in Manufacturing: The avoidance of precious metal catalysts or exotic reagents drastically simplifies the bill of materials. Traditional routes often require expensive scavengers to remove heavy metal residues from hydrogenation catalysts; this iron-mediated reduction leaves behind iron salts that are easily removed by aqueous washing and filtration. Additionally, the high yields reported in the patent embodiments (such as 93% for Z1 and 95% for the final salt) indicate a highly efficient use of raw materials, minimizing waste generation. This efficiency lowers the cost per kilogram of the active ingredient, allowing for more competitive pricing strategies in the generic pharmaceutical market without compromising margin.
- Enhanced Supply Chain Reliability: The starting materials for this process are bulk commodity chemicals produced by numerous global suppliers, reducing the risk of single-source dependency. In contrast, specialized reagents like triphosgene can face supply disruptions due to regulatory crackdowns on phosgene precursors. By utilizing a robust chemistry based on fundamental organic transformations (substitution, reduction, cyclization), manufacturers can secure long-term supply contracts with greater confidence. The mild reaction conditions also mean that the process can be run in a wider variety of reactor setups, increasing the flexibility of the manufacturing network to respond to demand surges.
- Scalability and Environmental Compliance: Environmental regulations regarding volatile organic compounds (VOCs) and hazardous waste are becoming increasingly stringent. This synthetic route addresses these concerns by utilizing solvents like ethanol and toluene, which are well-understood and easily recovered via distillation. The absence of phosgene-related waste streams simplifies the effluent treatment process, reducing the load on wastewater treatment plants. The process is inherently safer, with lower risks of runaway reactions, which facilitates easier permitting for capacity expansion. This alignment with green chemistry principles ensures long-term operational continuity and reduces the likelihood of regulatory fines or shutdowns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation. Understanding these nuances is critical for technical teams evaluating the feasibility of technology transfer. The focus is on practical implementation details that impact quality and throughput.
Q: What are the primary advantages of this synthetic route over traditional triphosgene methods?
A: This route avoids highly toxic reagents like phenyl chloroformate and triphosgene, utilizing milder conditions with o-bromonitrobenzene and thiosalicylic acid. This results in significantly reduced safety hazards, simpler waste treatment, and higher product purity by eliminating urea-type impurities common in phosgene-based routes.
Q: How does the process ensure high yield and scalability for commercial production?
A: The process utilizes robust reaction steps with high isolated yields (e.g., 93% for Z1, 92% for Z3, and 95% for the final salt). The use of common solvents like ethanol, toluene, and acetone, along with straightforward workup procedures like filtration and crystallization, makes the process highly amenable to large-scale batch production.
Q: What specific impurities are controlled by avoiding the 2-diaminodiphenyl sulfide starting material?
A: Traditional methods starting with 2-diaminodiphenyl sulfide often suffer from unstable intermediates and side reactions during esterification. By building the scaffold from o-bromonitrobenzene and thiosalicylic acid, the pathway minimizes the formation of complex byproducts associated with harsh esterification agents, leading to a cleaner impurity profile.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quetiapine Hemifumarate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex psychotropic agents depends on a partner who understands both the chemistry and the supply chain dynamics. Our technical team has extensively analyzed routes like CN103772319A to ensure that our manufacturing capabilities align with the highest standards of efficiency and safety. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met whether for clinical trials or full-scale market launch. Our facilities are equipped with rigorous QC labs capable of detecting trace impurities, ensuring that every batch meets stringent purity specifications required by global regulatory bodies. We are committed to delivering high-purity pharmaceutical intermediates that streamline your downstream formulation processes.
We invite you to engage with our technical procurement team to discuss how this optimized synthetic route can benefit your specific project requirements. By leveraging our expertise, you can access a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to this greener methodology. We encourage you to contact us to request specific COA data and route feasibility assessments tailored to your volume requirements. Let us collaborate to build a more resilient and cost-effective supply chain for Quetiapine Hemifumarate.
