Advanced Synthetic Route for Quetiapine Hemifumarate Intermediates and Commercial Scale-Up
Introduction to the Novel Synthetic Pathway
The pharmaceutical industry continuously seeks robust and scalable methodologies for the production of atypical antipsychotic agents, with Quetiapine Hemifumarate standing as a cornerstone molecule in this therapeutic class. A significant technological advancement in this domain is detailed in patent CN103772319A, which discloses a refined production process that fundamentally alters the synthetic landscape for this critical API intermediate. Unlike legacy methods that rely on harsh conditions and unstable precursors, this innovation leverages a strategic sequence of displacement, reduction, condensation, and halogenation reactions starting from readily available raw materials like o-bromonitrobenzene and thiosalicylic acid. The technical breakthrough lies not merely in the chemical transformation itself, but in the holistic optimization of reaction conditions to ensure mildness and operational simplicity, thereby addressing long-standing challenges in impurity management and process safety. For R&D directors and process chemists, this represents a pivotal shift towards greener chemistry without compromising the structural integrity or pharmacological potential of the final dibenzo[b,f][1,4]thiazepine derivative.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Quetiapine Hemifumarate has been plagued by significant technical hurdles that impact both cost efficiency and environmental compliance. Traditional bibliographic methods typically commence with 2-diaminodiphenyl sulfide as the primary starting material, necessitating the use of highly toxic reagents such as phenyl chloroformate or triphosgene to effectuate esterification and subsequent cyclization. These reagents are not only hazardous to handle on a large scale but also introduce severe safety risks regarding operator exposure and containment requirements. Furthermore, the intermediates generated in these conventional pathways are notoriously unstable, leading to complex decomposition profiles that result in a multitude of difficult-to-remove impurities. This instability often forces manufacturers to accept lower overall yields and engage in extensive, costly purification protocols to meet stringent pharmacopeial standards, creating a bottleneck in the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology outlined in the referenced patent introduces a paradigm shift by utilizing o-bromonitrobenzene and thiosalicylic acid as the foundational building blocks. This novel approach circumvents the need for dangerous phosgene equivalents entirely, replacing them with a sequence of safer, more controllable reactions including nucleophilic aromatic substitution and iron-mediated reduction. The process is characterized by its mild reaction conditions, which significantly reduce the thermal stress on sensitive molecular structures, thereby preserving the integrity of the intermediates throughout the synthesis. By simplifying the reaction steps and employing solvents that are easier to recover and recycle, such as ethanol and toluene, this new route offers a streamlined pathway that is inherently more amenable to industrial batch production. The result is a synthetic strategy that not only enhances the safety profile of the manufacturing plant but also delivers a product with superior purity characteristics right from the crude stage.
Mechanistic Insights into FeCl2-Catalyzed Reduction and Cyclization
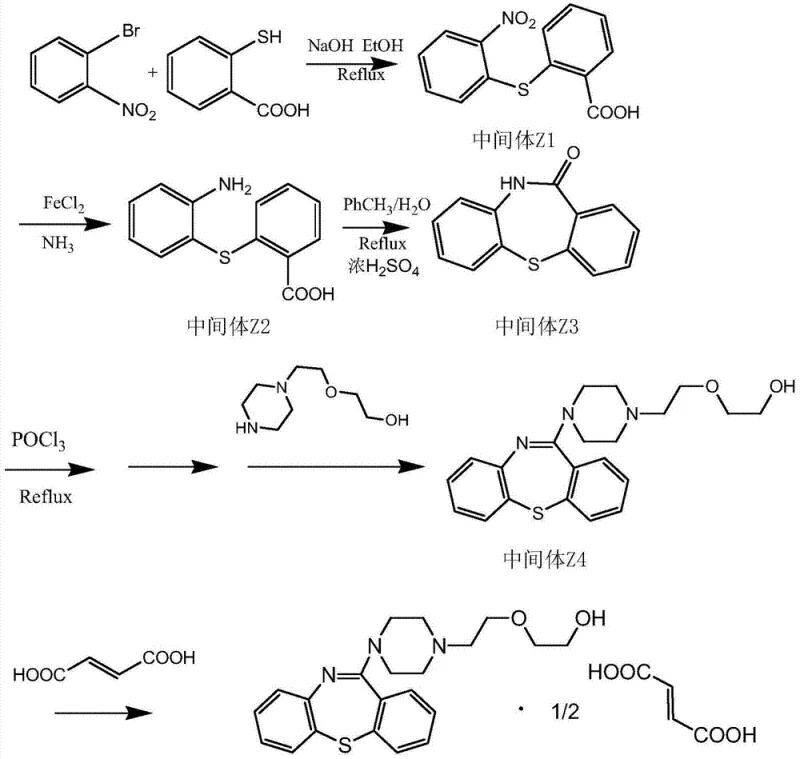
The core of this synthetic innovation relies on a sophisticated interplay of mechanistic steps that ensure high conversion rates and selectivity. The initial formation of Intermediate Z1 involves a nucleophilic aromatic substitution where the thiol group of thiosalicylic acid displaces the bromine atom on the nitrobenzene ring, facilitated by a strong base like sodium hydroxide in an alcoholic solvent. Following this, the reduction of the nitro group to an amine (Intermediate Z2) is achieved using ferrous chloride and ammonia, a classic yet highly effective green chemistry technique that avoids the use of catalytic hydrogenation which can be prone to over-reduction or catalyst poisoning. The subsequent cyclization step to form the tricyclic ketone (Intermediate Z3) is driven by concentrated sulfuric acid in a biphasic toluene-water system, promoting intramolecular condensation with high efficiency. Finally, the activation of the ketone with phosphorus oxychloride (POCl3) creates a reactive chloro-iminium species that undergoes nucleophilic attack by the piperazine derivative, locking in the side chain required for biological activity.
From an impurity control perspective, this mechanism offers distinct advantages over traditional routes. The stepwise nature of the synthesis allows for the isolation and purification of key intermediates like Z1 and Z2, effectively breaking the chain of impurity propagation that often occurs in one-pot telescoped reactions. The use of ferrous sulfate for reduction generates iron oxide byproducts that are easily filtered off, preventing metal contamination in the final API. Moreover, the specific choice of solvents and temperatures—such as refluxing in ethanol for the initial coupling and controlled cooling for crystallization—ensures that side reactions like polymerization or oxidative degradation are minimized. This rigorous control over the reaction environment translates directly into a cleaner crude product, reducing the burden on downstream purification units and ensuring that the final Quetiapine Hemifumarate meets the rigorous specifications required for clinical applications.
How to Synthesize Quetiapine Hemifumarate Efficiently
Implementing this advanced synthetic route requires precise adherence to the optimized parameters defined in the patent to maximize yield and purity. The process is divided into five distinct stages, beginning with the preparation of the diphenyl sulfide backbone and concluding with the final salt formation. Each step has been engineered to balance reaction kinetics with practical operability, ensuring that the transition from laboratory scale to commercial production is seamless. For process engineers looking to adopt this technology, understanding the critical control points—such as the temperature gradients during the reduction phase and the stoichiometry of the POCl3 activation—is essential for successful replication. The following guide outlines the standardized operational framework derived from the patent embodiments, serving as a blueprint for efficient manufacturing.
- Preparation of Intermediate Z1: React o-bromonitrobenzene with thiosalicylic acid in ethanol using sodium hydroxide under reflux to form 2-nitro-2'-carboxyl diphenyl sulfide.
- Reduction to Intermediate Z2: Reduce the nitro group of Z1 using ferrous sulfate and ammonia in water to obtain 2-amino-2'-carboxyl diphenyl sulfide.
- Cyclization to Intermediate Z3: Perform acid-catalyzed cyclization of Z2 using concentrated sulfuric acid in toluene/water to form the tricyclic ketone.
- Formation of Intermediate Z4: Activate the ketone with POCl3 followed by nucleophilic substitution with N-piperazine ethoxy ethanol.
- Salt Formation: Dissolve Intermediate Z4 in acetone and react with fumaric acid to crystallize the final Quetiapine Hemifumarate product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthetic route presents a compelling value proposition centered on risk mitigation and cost optimization. By eliminating the reliance on highly regulated and expensive toxic reagents like triphosgene, the manufacturing process becomes significantly more robust against regulatory shifts and raw material price volatility. The simplified operational workflow reduces the complexity of the production schedule, allowing for faster batch turnover and improved responsiveness to market demand fluctuations. Furthermore, the enhanced stability of the intermediates minimizes the risk of batch failures due to decomposition, ensuring a more consistent and reliable supply of high-purity pharmaceutical intermediates for downstream API synthesis.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of costly and hazardous reagents with cheaper, commodity-grade chemicals such as ferrous sulfate and sodium hydroxide. Additionally, the ability to recover and recycle solvents like toluene and ethanol drastically lowers the variable costs associated with raw material consumption. The higher yields achieved through improved intermediate stability mean that less starting material is wasted, directly improving the cost-of-goods-sold (COGS) profile for the final product. This efficiency gain allows manufacturers to offer more competitive pricing without sacrificing margin, creating a sustainable advantage in the global supply market.
- Enhanced Supply Chain Reliability: The starting materials for this route, specifically o-bromonitrobenzene and thiosalicylic acid, are widely available commodities with established global supply chains, reducing the risk of sourcing bottlenecks. Unlike specialized reagents that may have single-source suppliers, these bulk chemicals ensure continuity of supply even during market disruptions. The robustness of the reaction conditions also means that production is less susceptible to minor variations in utility availability or environmental conditions, further stabilizing the delivery schedule for customers relying on just-in-time inventory models.
- Scalability and Environmental Compliance: From an environmental standpoint, this process significantly reduces the generation of hazardous waste, aligning with increasingly strict global environmental regulations. The avoidance of heavy metal catalysts and toxic chlorinating agents simplifies waste treatment protocols and lowers the cost of environmental compliance. This "green" profile not only mitigates regulatory risk but also enhances the brand reputation of the supply chain partners, making it an attractive option for pharmaceutical companies aiming to reduce their carbon footprint and meet sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic method. These insights are derived directly from the comparative data and beneficial effects described in the patent documentation, providing clarity on how this technology outperforms legacy methods in terms of safety, yield, and scalability. Understanding these nuances is critical for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this novel synthesis route improve safety compared to conventional methods?
A: Conventional methods often utilize highly toxic reagents such as phenyl chloroformate or triphosgene for esterification and cyclization. This patented process replaces those hazardous materials with milder reagents like ferrous sulfate for reduction and avoids the use of phosgene equivalents, significantly reducing operational risks and toxicity profiles.
Q: What are the key advantages regarding yield and impurity control?
A: The traditional pathway suffers from unstable intermediates which lead to high impurity levels and lower yields. By utilizing a stepwise approach starting from o-bromonitrobenzene and thiosalicylic acid, the reaction conditions are more controlled and mild, resulting in higher stability of intermediates and a cleaner final product profile.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is specifically designed for industrial scalability. It features simple operational steps, uses common solvents like ethanol and toluene which are easily recoverable, and generates significantly less 'three-waste' pollution compared to older technologies, making it economically and environmentally viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quetiapine Hemifumarate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is executed with precision and efficiency. We are committed to delivering high-purity Quetiapine Hemifumarate intermediates that adhere to stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our infrastructure is designed to handle complex chemistries safely, leveraging the very process improvements detailed in recent patents to drive value for our partners.
We invite you to collaborate with us to explore how this optimized synthetic route can enhance your supply chain resilience and cost structure. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments, allowing us to demonstrate concretely how our manufacturing excellence can support your long-term strategic goals in the production of atypical antipsychotic agents.
