Revolutionizing Lenalidomide Production: A High-Efficiency 3-Step Synthetic Route for Commercial Scale-Up
Revolutionizing Lenalidomide Production: A High-Efficiency 3-Step Synthetic Route for Commercial Scale-Up
The pharmaceutical industry is constantly seeking more efficient, cost-effective, and environmentally sustainable pathways for the production of critical oncology therapeutics. Patent CN103601717A introduces a groundbreaking methodology for the synthesis of Lenalidomide, a potent immunomodulatory drug used in the treatment of multiple myeloma and myelodysplastic syndromes. This novel preparation method fundamentally reimagines the construction of the piperidine-2,6-dione scaffold by utilizing glutarimide and 4-nitro-isoindolin-1-one as primary building blocks. Unlike legacy processes that suffer from excessive step counts and reliance on expensive chiral pool materials, this invention streamlines the synthesis into three high-yielding transformations. For R&D directors and supply chain leaders, this represents a significant opportunity to optimize the manufacturing of this high-value API intermediate, ensuring a more reliable lenalidomide intermediate supplier capability while adhering to stringent quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
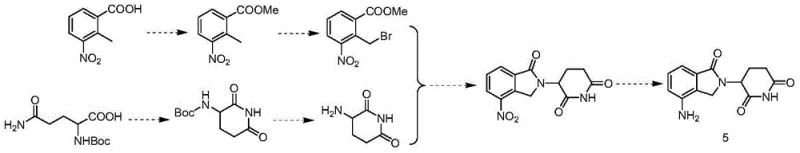
Historically, the industrial synthesis of Lenalidomide has been plagued by inefficiencies inherent in convergent strategies that rely on complex amino acid derivatives. As illustrated in prior art such as WO2010100476, traditional routes often commence with N-protected L-glutamine, necessitating a series of cumbersome intramolecular condensations and deprotection steps to establish the glutarimide ring. These methods frequently require the separate synthesis of functionalized aromatic fragments through multi-step esterification and benzylic bromination sequences. The cumulative effect of these operations is a prolonged production timeline, increased solvent consumption, and a significant erosion of overall yield due to the multiplication of losses at each stage. Furthermore, the reliance on chiral starting materials like N-Boc-L-glutamine introduces substantial raw material costs and supply chain vulnerabilities, making cost reduction in pharma manufacturing difficult to achieve without compromising purity.
The Novel Approach
In stark contrast, the methodology disclosed in CN103601717A adopts a divergent strategy that couples two readily available fragments late in the synthesis. By initiating the sequence with the direct alpha-halogenation of glutarimide, the process bypasses the need for amino acid protection entirely. This is followed by a robust nucleophilic substitution with 4-nitro-isoindolin-1-one, effectively stitching the two core heterocyclic systems together in a single operation. The final reduction of the nitro group unveils the active amine functionality, delivering the target molecule with exceptional efficiency. This approach not only simplifies the operational complexity but also enhances the commercial scale-up of complex pharmaceutical intermediates by utilizing commodity chemicals that are accessible globally. The elimination of chromatographic purifications in favor of crystallization and extraction further underscores the industrial viability of this route.
Mechanistic Insights into Alpha-Halogenation and Nucleophilic Coupling
The success of this synthetic route hinges on the precise control of the alpha-halogenation step, where glutarimide is converted into a reactive electrophile. Under acidic conditions, typically using bromine in acetic acid, the enol tautomer of glutarimide reacts selectively at the alpha-position to the carbonyl group. This transformation generates 3-halo-2,6-dioxopiperidine (Compound 3), a versatile synthon that retains the integrity of the imide ring while introducing a leaving group essential for the subsequent coupling. The choice of halogenating agent, ranging from molecular bromine to N-bromosuccinimide, allows for fine-tuning of the reaction kinetics to minimize di-halogenation byproducts. This step is critical for ensuring high-purity lenalidomide, as impurities formed here can propagate through the synthesis, complicating downstream purification efforts.
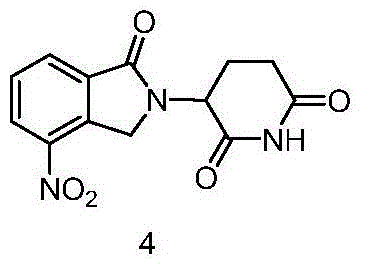
Following the activation of the glutarimide scaffold, the core carbon-nitrogen bond formation occurs via a nucleophilic substitution mechanism. The 4-nitro-isoindolin-1-one acts as the nucleophile, attacking the alpha-carbon of the halogenated intermediate under basic conditions. The presence of the electron-withdrawing nitro group on the isoindolinone ring enhances the acidity of the lactam nitrogen, facilitating deprotonation by mild bases such as potassium carbonate or triethylamine. This results in the formation of the key intermediate, 3-(4-nitro-1,3-dihydro-1-oxo-2H-isoindole-2-yl)-2,6-dioxopiperidine (Compound 4). The structural integrity of this coupled product is paramount, as it sets the stage for the final reduction. The ability to isolate this intermediate in high yield demonstrates the compatibility of the reaction conditions and the stability of the newly formed bond against hydrolysis or rearrangement.
How to Synthesize Lenalidomide Efficiently
Implementing this novel synthesis requires careful attention to reaction parameters to maximize yield and minimize impurity profiles. The process begins with the halogenation of glutarimide, followed by the coupling with the nitro-isoindolinone fragment, and concludes with the reduction of the nitro group to the aniline. Each step has been optimized to use common industrial solvents and reagents, facilitating easy technology transfer from the laboratory to the pilot plant. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and workup procedures, are outlined below to guide process chemists in replicating this high-efficiency route.
- Perform alpha-halogenation of glutarimide using bromine or NBS in acetic acid to generate 3-halo-2,6-dioxopiperidine.
- Execute nucleophilic substitution by condensing the halo-intermediate with 4-nitro-isoindolin-1-one under alkaline conditions.
- Complete the synthesis by reducing the nitro group of the coupled intermediate using iron powder or catalytic hydrogenation to yield Lenalidomide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers transformative benefits that extend beyond simple yield improvements. By shifting the starting material base from specialized chiral amino acids to commodity glutarimide and nitro-phthalimide derivatives, the supply chain becomes significantly more resilient. The availability of these raw materials on a global scale mitigates the risk of shortages that often plague specialty chemical markets. Furthermore, the reduction in step count directly correlates to a reduction in manufacturing lead time, allowing for faster response to market demand fluctuations. This agility is crucial for maintaining the continuity of supply for life-saving oncology medications, ensuring that patients receive timely access to treatment without interruption due to production bottlenecks.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive protecting groups and chiral auxiliaries. Traditional routes incur high costs associated with the purchase and subsequent removal of Boc or Cbz groups, as well as the stoichiometric waste generated during deprotection. By circumventing these steps, the novel method drastically lowers the cost of goods sold (COGS). Additionally, the use of iron powder or standard catalytic hydrogenation for the final reduction avoids the need for precious metal catalysts that require complex recovery and recycling protocols. This simplification of the bill of materials translates into substantial cost savings that can be passed down the supply chain or reinvested into further R&D initiatives.
- Enhanced Supply Chain Reliability: The reliance on easily available raw materials such as glutarimide and simple nitro-aromatics ensures a stable supply base. Unlike complex chiral intermediates that may have limited suppliers, these commodity chemicals are produced by multiple manufacturers worldwide, reducing dependency on single-source vendors. This diversification of the supply base enhances negotiation leverage and protects against regional disruptions. Moreover, the robustness of the reaction conditions, which tolerate a range of temperatures and solvent qualities, reduces the likelihood of batch failures due to minor variations in raw material specifications, thereby improving overall supply reliability.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this route aligns well with green chemistry principles. The solvents employed, such as acetic acid, ethanol, and ethyl acetate, are generally regarded as safer and more environmentally benign than chlorinated solvents often used in older methodologies. The high atom economy of the coupling step and the avoidance of heavy metal waste streams simplify wastewater treatment and reduce the environmental footprint of the manufacturing facility. This compliance with increasingly stringent environmental regulations facilitates smoother regulatory approvals and reduces the operational costs associated with waste disposal, making the process highly attractive for large-scale commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Lenalidomide synthesis. These insights are derived directly from the patent data and are intended to clarify the feasibility and advantages of adopting this route for industrial applications. Understanding these details is essential for stakeholders evaluating the potential for technology transfer and commercial partnership.
Q: How does this new method improve upon traditional Lenalidomide synthesis?
A: Traditional methods often rely on expensive chiral amino acid derivatives and complex protection-deprotection sequences, leading to low overall yields and high waste. This novel approach utilizes commodity glutarimide and direct coupling, significantly shortening the route and eliminating costly chiral auxiliaries.
Q: What are the primary cost drivers eliminated in this process?
A: The process eliminates the need for Boc-protected glutamine derivatives and complex esterification/bromination sequences on the aromatic ring prior to coupling. By using pre-functionalized 4-nitro-isoindolin-1-one and direct alpha-halogenation of glutarimide, raw material costs and processing time are drastically reduced.
Q: Is this synthesis suitable for large-scale industrial production?
A: Yes, the reaction conditions utilize common solvents like acetic acid, DMF, and ethanol, and reagents like iron powder or palladium carbon which are standard in industrial settings. The high yields reported (up to 99% in early steps) indicate excellent scalability and robustness for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lenalidomide Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for high-value oncology intermediates like Lenalidomide. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering high-purity lenalidomide intermediates that meet the rigorous demands of the global pharmaceutical market. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, providing our partners with the confidence they need to advance their drug development pipelines.
We invite you to collaborate with us to leverage this innovative synthetic technology for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value and efficiency in your Lenalidomide manufacturing strategy.
