Advanced Ofloxacin Manufacturing: High-Yield Cyclization and Cost-Efficient Scale-Up
Introduction to Next-Generation Ofloxacin Synthesis
The global demand for high-quality fluoroquinolone antibiotics necessitates manufacturing processes that balance efficiency, purity, and environmental sustainability. Patent CN101648960A introduces a transformative methodology for the preparation of Ofloxacin, addressing critical bottlenecks found in legacy production techniques. This intellectual property outlines a streamlined synthetic pathway that initiates with tetrafluorobenzoyl chloride, bypassing the cumbersome multi-step sequences characteristic of older technologies. By integrating a direct compounding method with optimized cyclization conditions, the invention achieves a substantial elevation in both yield and product integrity. The strategic design of this route minimizes the generation of hazardous byproducts, aligning with modern green chemistry principles while ensuring a robust supply of this essential antimicrobial agent.
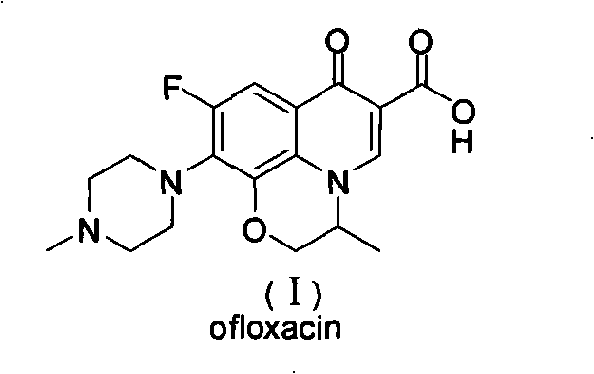
For procurement specialists and R&D directors, understanding the structural nuances of this synthesis is vital. The process targets the formation of the 9,10-difluoro-2,3-dihydro-3-methyl-7-oxo-7H-pyrido[1,2,3-de]-[1,4]benzoxazine-6-carboxylic acid core with exceptional precision. This intermediate serves as the pivotal scaffold upon which the final pharmacological activity is built. The patent emphasizes not just the chemical transformation but the operational simplicity, allowing for shorter production cycles that directly translate to improved throughput capabilities for manufacturing partners seeking reliable API intermediate supplier relationships.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Ofloxacin has been plagued by inefficiencies that drive up costs and complicate supply chain logistics. Traditional pathways, such as those described in US4777253, often commence with 2,3,4,5-tetrafluorobenzoic acid, necessitating a lengthy sequence involving chlorination, condensation with diethyl ethoxymethylenemalonate, and subsequent cyclization steps. These legacy routes are notoriously inefficient, with total recovery rates hovering around a mere 21%, meaning nearly 80% of valuable starting materials are lost to side reactions or purification losses. Alternatively, methods utilizing trifluoronitrobenzene derivatives, as seen in US4382892, present even steeper challenges with total yields dropping to approximately 14%. Beyond the economic drain of low yields, these conventional approaches generate excessive "three wastes"—wastewater, waste gas, and solid waste—creating significant environmental compliance hurdles and escalating disposal costs for manufacturers.
The Novel Approach
In stark contrast, the methodology disclosed in CN101648960A represents a paradigm shift towards lean manufacturing. By employing tetrafluorobenzoyl chloride as the primary electrophile and reacting it directly with a specialized 3-(2-R1-2-R2-4-methyl oxazolidinyl) acrylate derivative, the process eliminates several intermediate isolation steps. This convergence strategy allows for the direct formation of the quinolone skeleton with reported yields reaching 85-90% for the critical cyclization phase. The visual representation of this comprehensive synthetic pathway highlights the elegance of the design, where the construction of the heterocyclic ring and the installation of the fluorine substituents occur in a highly coordinated manner.
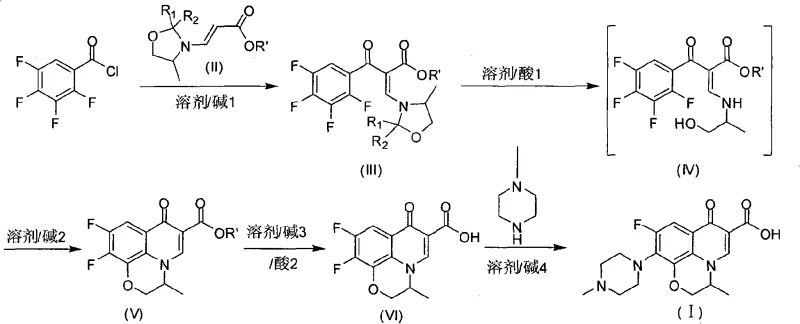
Furthermore, the physical characteristics of the intermediates produced via this novel route offer distinct processing advantages. While traditional methods often yield crude intermediates as yellow powders requiring rigorous and costly recrystallization to meet purity standards, this new approach consistently produces Compound V and Compound VI as white solids. This intrinsic high purity, with HPLC content exceeding 98.5%, significantly reduces the burden on downstream purification units. For a cost reduction in pharmaceutical intermediates manufacturing, this reduction in refining steps is a major value driver, enabling faster batch turnover and lower energy consumption per kilogram of finished product.
Mechanistic Insights into Oxazolidine-Mediated Cyclization
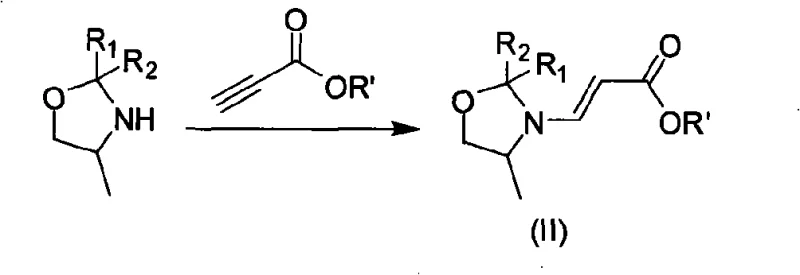
The cornerstone of this innovative synthesis lies in the preparation and utilization of the key building block, Compound II. The mechanism involves the condensation of a D/L-aminopropanol carbonyl analog derivative with a propiolate ester. This reaction creates a reactive enamine-like system within the oxazolidine ring that is perfectly poised for nucleophilic attack. When introduced to the tetrafluorobenzoyl chloride, the electron-rich nitrogen of the oxazolidine moiety facilitates an acylation event that sets the stage for the subsequent ring closure. The precise control of reaction parameters, specifically maintaining temperatures between 0-20°C during the initial formation of Compound II, is critical to preventing polymerization of the acrylate and ensuring the stereochemical integrity required for the final drug's efficacy.
Following the initial acylation to form Compound III, the process employs a sophisticated hydrolysis and cyclization sequence. The addition of acid triggers the hydrolysis of the oxazolidine protecting group, revealing the free amine necessary for the intramolecular nucleophilic aromatic substitution (SNAr) that closes the pyridone ring. This cascade reaction is meticulously managed by the addition of specific alkalis, such as potassium carbonate or triethylamine, which act as proton scavengers to drive the equilibrium towards the cyclized product. The result is the formation of the 9,10-difluoro-3-methyl-7-oxo-2,3-dihydro-7H-pyrido[1,2,3-de][1,4]benzoxazine core. This mechanistic pathway avoids the harsh conditions often required in nitro-reduction routes, thereby preserving the sensitive fluorine substituents that are essential for the antibiotic's broad-spectrum activity.
How to Synthesize Ofloxacin Efficiently
Implementing this synthesis route requires careful attention to solvent selection and stoichiometric ratios to maximize the benefits outlined in the patent. The process is designed to be operationally simple, utilizing common organic solvents like acetonitrile, toluene, or chlorobenzene which can be easily recovered and recycled, further enhancing the economic viability of the method. The following guide summarizes the critical operational phases derived from the patent examples, providing a framework for laboratory validation and pilot scale-up. Detailed standard operating procedures regarding specific agitation rates, cooling profiles, and filtration techniques should be developed based on these foundational steps to ensure reproducibility and safety in a GMP environment.
- Condense tetrafluorobenzoyl chloride with 3-(2-R1-2-R2-4-methyl oxazolidinyl) acrylate in the presence of an organic base at 50-90°C to form the acylated intermediate.
- Hydrolyze the intermediate and induce cyclization using an inorganic or organic alkali under reflux conditions to generate the quinolone carboxylic acid ester core.
- Perform hydrolysis to obtain the carboxylic acid, followed by nucleophilic substitution with methyl piperazine in a solvent system to yield the final Ofloxacin API.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the adoption of this patented synthesis route offers compelling strategic advantages that extend beyond simple chemistry. The primary benefit lies in the drastic simplification of the production workflow, which inherently de-risks the supply of this critical antibiotic intermediate. By consolidating multiple reaction steps into a more direct sequence, manufacturers can reduce the number of unit operations required, leading to a significant decrease in labor costs and equipment occupancy time. This efficiency gain allows for greater flexibility in production scheduling, enabling suppliers to respond more rapidly to fluctuations in market demand without the need for massive capital investment in additional reactor capacity.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the elimination of expensive transition metal catalysts and the reduction of raw material waste. Traditional routes often require stoichiometric amounts of reagents that end up as waste, whereas this method maximizes atom economy by incorporating the majority of the starting material into the final product structure. Additionally, the ability to use a single solvent system throughout multiple steps facilitates efficient solvent recovery, drastically cutting down on the volume of fresh solvent required and minimizing waste disposal fees associated with mixed solvent streams.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of sourcing specialized reagents for multi-step syntheses. This novel route relies on readily available commodity chemicals such as tetrafluorobenzoyl chloride and methyl piperazine, which are produced at scale by numerous global chemical suppliers. This diversification of the raw material base reduces the risk of supply disruptions caused by vendor-specific issues. Furthermore, the high yield of the cyclization step ensures that less starting material is needed to produce the same amount of API, creating a buffer against raw material price volatility and securing long-term cost stability.
- Scalability and Environmental Compliance: As regulatory scrutiny on pharmaceutical manufacturing intensifies, the environmental profile of a synthesis route becomes a key differentiator. This method generates significantly less hazardous waste compared to nitrobenzene-based routes, simplifying the permitting process for new manufacturing lines. The production of white solid intermediates rather than colored, impure masses reduces the need for intensive chromatographic purification or repeated recrystallizations, which are often the most resource-intensive parts of API production. This streamlined approach supports sustainable manufacturing goals and ensures compliance with increasingly stringent environmental regulations globally.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Ofloxacin synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear picture of what potential partners can expect regarding performance and quality. Understanding these details is crucial for making informed decisions about technology transfer and long-term supply agreements.
Q: How does this new Ofloxacin synthesis route improve upon traditional nitrobenzene methods?
A: Traditional methods starting from trifluoronitrobenzene often suffer from total yields as low as 14% and generate significant hazardous waste. The patented route utilizing tetrafluorobenzoyl chloride achieves yields of 85-90% in the critical cyclization step, drastically reducing raw material consumption and waste treatment burdens.
Q: What are the purity advantages of the compound V and VI intermediates in this process?
A: Unlike additive methods that produce yellow powders requiring extensive refining, this novel process yields Compound V and Compound VI as white solids with HPLC content exceeding 98.5% and 99% respectively. This inherent high purity simplifies downstream processing and ensures a superior impurity profile for the final API.
Q: Can this synthesis method be scaled for commercial API production?
A: Yes, the process is designed for industrial scalability. It utilizes common organic solvents like acetonitrile or toluene which facilitate solvent recovery and recycling. Furthermore, the reduction in reaction steps and the ability to use single solvent systems significantly lowers the operational complexity required for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ofloxacin Supplier
At NINGBO INNO PHARMCHEM, we recognize that the theoretical advantages of a patent must be translated into tangible commercial reality through expert process engineering. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and purity specifications promised in CN101648960A are realized on an industrial scale. We utilize rigorous QC labs and state-of-the-art analytical instrumentation to monitor every critical parameter of the synthesis, guaranteeing that our Ofloxacin intermediates meet the stringent purity specifications required by top-tier pharmaceutical companies worldwide.
We invite you to collaborate with us to leverage this advanced manufacturing technology for your supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to evaluate how this optimized synthesis can enhance your portfolio of fluoroquinolone antibiotics.
